User:Nicole L Yergler/Notebook/Biology 210 at AU
3/22/2014 EMBRYOLOGY LAB: The effect of constant light exposure on zebrafish embryo development
Purpose/Objective
Embryology is a valuable field in the realm of biology because it explores the development of an embryo of a specific organism, from fertilization to the fetal stage. Embryology can be used to test and determine the effects of certain environmental changes on the development of the embryo. Because animals share distinct similarities, embryology can be used to develop theories regarding environmental (including chemical, physical, etc.) manipulations and their effect on the embryos' growth stages. Zebrafish are oftentimes used in biological research as a 'model' for research, due to their fast development, and transparent shell characteristic. Also, in terms of abnormal light rearing, zebrafish are invaluable in retinal research, since they show a high degree of retinal immaturity, which allows researchers to assess the embryological effects without interrupting development. The purpose of the lab is to gain understanding in the complex stages of normal zebrafish growth, and test the effect of constant light exposure on zebrafish embryological development, using a control group and a group exposed to the independent variable of permanent light.
Hypothesis and Prediction
Based on the article "Effects of Abnormal Light-rearing Condition on Retinal Physiology in Larvae Zebrafish", the effects that are seen in zebrafish exposed to constant light show a severe deficit in the UV and short-wavelength areas and only a slight deficit in the middle- and long-wavelength areas. The deficits in this study were not permanent in the zebrafish (showing signs of successful regeneration). The main questions in this lab are: how is the behavior of the zebrafish raised in constant light different from those raised in normal conditions?, and what are the differences in physical developments seen in both the independent and dependent variable group? The hypothesis is that the zebrafish in the constant light environment will demonstrate a retarded eye development and disorderly swimming, compared to the control group.
Methods
Both a control and treated group were included in the lab, in order to successfully monitor the differences between the groups. In total, 40 zebrafish of similar ages were chosen at random; 20 were put in one petri dish with 20mL distilled water, and 20 were put in another with 20mL distilled water. For the next 2 weeks, the control group was exposed to normal light (natural day-night cycle) and the independent variable group was exposed to constant light under a lamp. Over the next two weeks, the zebrafish were observed 7 times (Mondays, Wednesdays, and Fridays), and their qualitative and quantitative characteristics were recorded, including: hatching, viability, swimming/movement, timing/yolk absorption, eye pigmentation development, eye movement, development of body/tail pigmentation, heart rate, structural development, tail and total length, eye diameter, and developmental delay. The quantitative measurements were taken for the zebrafish that were preserved in fermeldahyde and then set on two specific dates. The water was changed every three days, and dead zebrafish/hatched eggs were removed from the petri dishes, so as to prevent contamination.
Results
Table 1: Raw Data for Zebrafish Exposed to Constant Light and Control Zebrafish
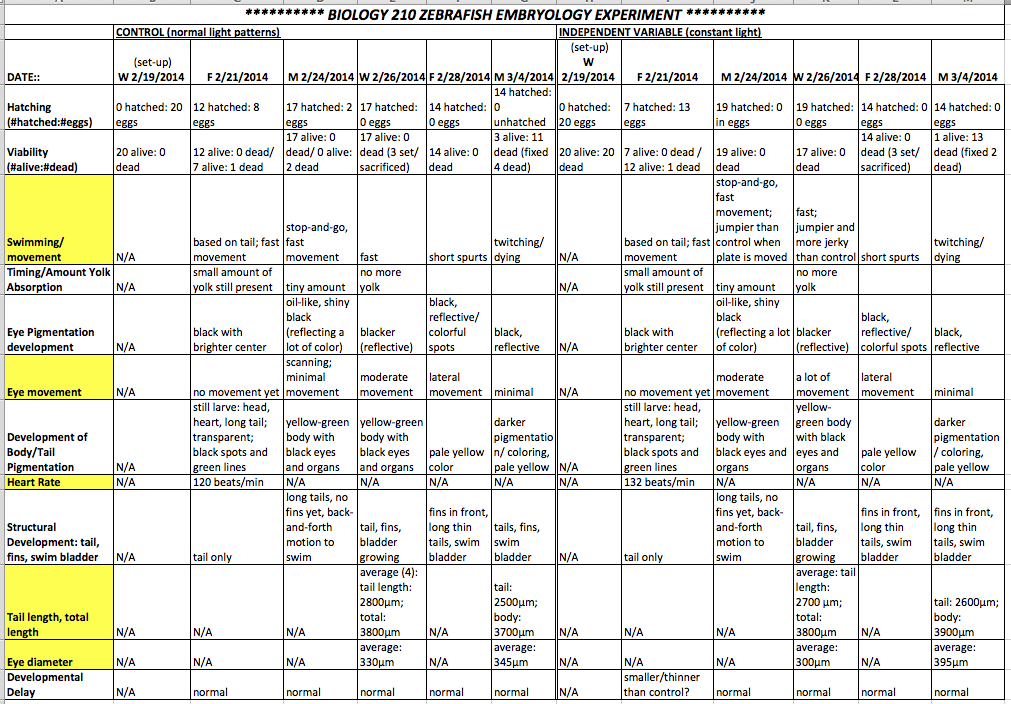
The above chart shows the raw data for the control and constant light exposure group. The N/A cells correspond to measurements not taken (for values of heart rate or tail/body length), since these were only taken when the fish were easy to measure and/or set in fermeldahyde. The dates the data was taken are along the top, and the characteristics that show notable differences between the control and independent variable group are highlighted in yellow.
Conclusions & Future Steps
The purpose of the experiment was to determine the effect of constant light exposure on zebrafish embryological development. This experiment was a learning opportunity in many ways, since many of the challenges were unpredictable, and the data in the latter part of the experiment was relatively useless because the zebrafish died suddenly and in mass. The temperature in the laboratory, where the zebrafish were kept, was unnaturally warm, which led to the quickened development of the embryos and yolk absorption, and they essentially starved to death before accurate observations could be made and viable living fish could be set for successful measurements. The development of the control group versus that of the group exposed to constant light were not strikingly dissimilar. As highlighted in the table, there are indeed differences in a number of the measurements and behaviors of the fish, which would have probably been even more stark if the fish had not all died before data was taken in the last few days. Also, the results that were seen in the published paper that the hypotheses were based on were dependent on highly sophisticated data collection. The most notable differences include: faster heart rate in the light group; size of the tail and body, as well as eye diameter (though the measurement differences were not consistent); and the fact that those fish exposed to constant light were definitely more active and jumpy, and demonstrated more eye movement, than the controls when the petri dishes were moved in any way. This could be because of abnormal eye development. The differences in eye development and behavior between these two zebrafish groups was not convincing enough to form a viable conclusion. In the future, the same experiment should be conducted, in a thermo-regulated room; furthermore, preservation and set samples should be made more than twice, so that the differences in each and every developmental stage could be recorded. Because the objective was not addressed successfully, it is important to conduct more studies, especially since they could be beneficial to further research in other species.
NLY
2/26/2014 DNA sequence lab
Introduction: The purpose of this experiment is to find a specific strain of bacteria based on the DNA sequences of the bacteria found in the tall bushes transect (number 3). The PCR reactions did not work for our group, so we will use the sequences from other groups that were assigned the same transect. The data all comes full circle in the comparison of the laboratory data with the DNA sequences of the actual bacteria found in the transect, the data all comes together full-circle. The NCBI Blast website is useful in identifying DNA sequences from PCR reactions.
Methods: Please refer to the PCR reaction procedure in the Bacteria portion of the transect reports for the PCR Procedure. After the PCR was completed, the materials were sent to Genewiz Inc. for sequencing purposes. The DNA sequences were then identified using the NCBI Blast Database.
Raw Data:
Table 1: PCR Results and NCBI Blast Bacteria Identification
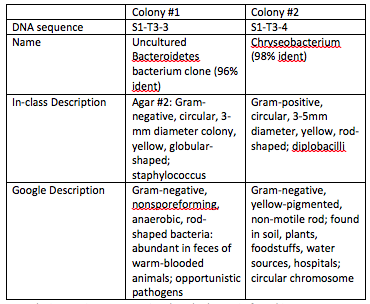
Discussion: It is not a surprise that the bacteria found in our transect are not the same bacteria that another group found in our transect, since there is a plethora of bacteria that is undoubtedly inhabiting the 20-by-20 foot transect. One gram-negative bacteria was found and identified in our group, but this was unlike the two gram-negative bacteria whose sequences were found by another Biology 210 lab group. NLY
2/25/2014
LAB Five
Objective: The objective of this experiment is to understand the importance of invertebrates and learn how simple systems (including specialized cells and overall body plan) evolved into more complex systems. In this experiment, we observed the three types of worms with different body structures/cross-sections, in terms of their ectoderm, mesoderm, and endoderm. Then the invertebrates that were exposed using the Berlese funnel and that are therefore present in the transect were identified. All of the information acquired during the past weeks can be used to hypothesize which types of vertebrates are most likely coming in contact with the transect/ecosystem, and also construct a food web based on these organisms. The hypothesis of the lab is that the invertebrates found in the transect will be mites and spiders, especially since spiders are the organisms that are most present in leaf litter, and there was also soil in our sample.
Specific Steps: Procedure I: 1. Observe the a coelomate, Planaria, with the dissecting scope. 2. Look at the cross-sectional slide of the Planaria with the microscope. 3. Observe the nematodes and a cross-sectional slide of their pseudocoelomate structure. 4. Observe the coelomate, Annelida. 5. Describe the movements of these three types of worms and how the movement relates to their body structure in your notebook. Procedure II: 1. Break down the Berlese setup and transfer the preservative solution to a Petri dish. Examine under a dissecting microscope. 2. Identify the main groups of invertebrates. 3. Randomly select 5 organisms in the petri dish and identify them as closely as you can. Use the West Virginia Petri dishes if there are not enough organisms present. Procedure III: 1. Consider the vertebrates who may inhabit and pass through the transect. 2. Look through the textbook and identify five who might inhabit the transect. 3. Determine the classification of each and biotic/abiotic benefits. 4. Construct a food web based on the organisms observed in the transect.
Raw Data: Table 1: Acoelomates, Pseudocoelomates, and Coelomates

Table 2: Invertebrates
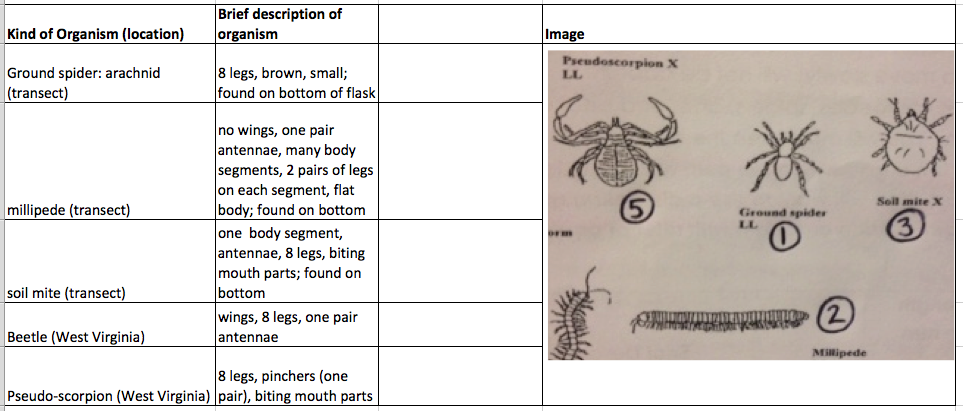
Size range: a few mm (soil mite) to a few centimeters (beetle). The spiders are most common in leaf litter, as well as millipedes, centipedes, and pseudoscorpions.
Table 3: Vertebrates
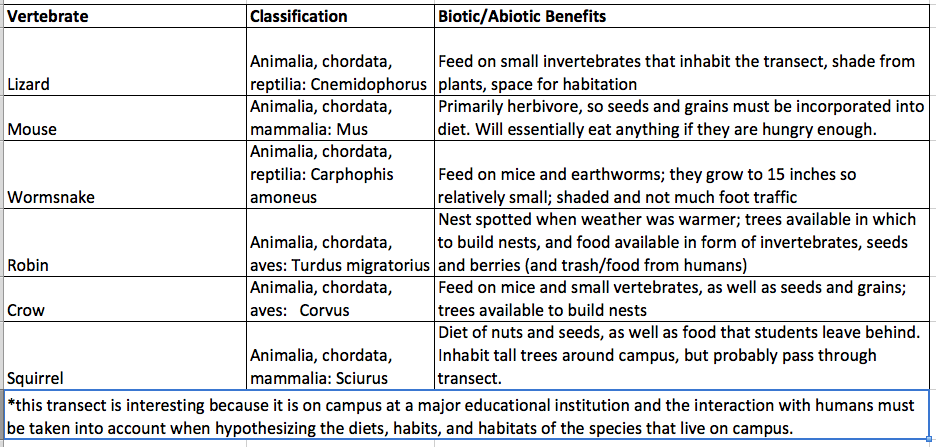
Table 4: Food Web
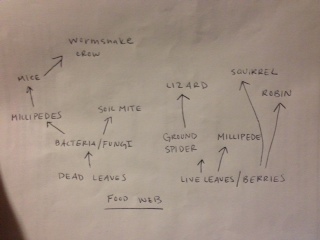
Conclusions/Future Labs:
This lab incorporates invertebrates, vertebrates, and the web of organisms that inhabit the 20x20 transect on the AU campus. The hypothesis was confirmed to an extent, since a small spider and soil mites were found in the samples that were incorporated into the Burlese funnel setup, as well as a small millipede. This lab brought everything together, in that the invertebrates were observed and the types of vertebrates that would be present in the transect were hypothesized.
NY
2/25/2014
LAB Four
Objective: The objective of this experiment is to understand the characteristics and diversity of plants, and to appreciate the function and importance of fungi. The evolution of Bryophytes, Tracheophytes, and Angiosperms was explored, and the existence of these plant types in the AU transect was studied. The characteristics of a bryophyte moss and an angiosperm were observed, and five plant samples from the transect were collected and categorized using a dichotomous key. The plant types were examined in terms of three general features: (1) vascularization, (2) specialized structures, and (3) reproduction. A hypothesis for the types of plants that will be found in the transect is: all three types of plants will be present in the transect, but the most plentiful will be the angiosperms. Because we are studying and identifying the plants in the AU transect , we will have a better idea of the interaction between all of the organisms present in the 20x20ft ecosystem.
Specific Steps: Procedure I: 1. Bring three bags and proceed to transect. 2. Obtain a leaf litter sample from a site with soft soil and dead leaves or ground cover. Dig only into the top layer of the soil and plant matter above. Place 500g of litter into one bag. 3. Take representative samples from five plants in a minimally-damaging manner. For trees, take a picture of the whole tree and identify the genus by identifying old leaves or branches on the ground. 4. Find any seeds, pine cones, flowers, etc. and put them into another bag. Procedure II: 1. Observe moss and examine lily stem cross-section slide. 2. Describe the vascularization of each plant found in transect. Procedure III: 1. Observe leaves of angiosperm and moss. 2. Describe the shape, size, and cluster arrangement of leaves from transect. Procedure IV: 1. Observe the polytrichum moss 2. Assert monocot or dicot for plants found in transect Procedure V: 1. Examine black bread mold under a dissecting microscope Procedure VI: 1. Pour 25mL of 50:50 ethanol:water solution into a flask. 2. Tape screening material over the bottom of a large plastic funnel and place funnel into neck of flask 3. Place leaf-litter sample in the top of the funnel, with leaves on the bottom and soil on the top layer 4. Place a 40 Watt lamp above the funnel with the bulb 1-2 inches from the top layer. Cover everything with foil.
Raw Data: The plant vascularization, specialization, and reproduction are all included in the following table.
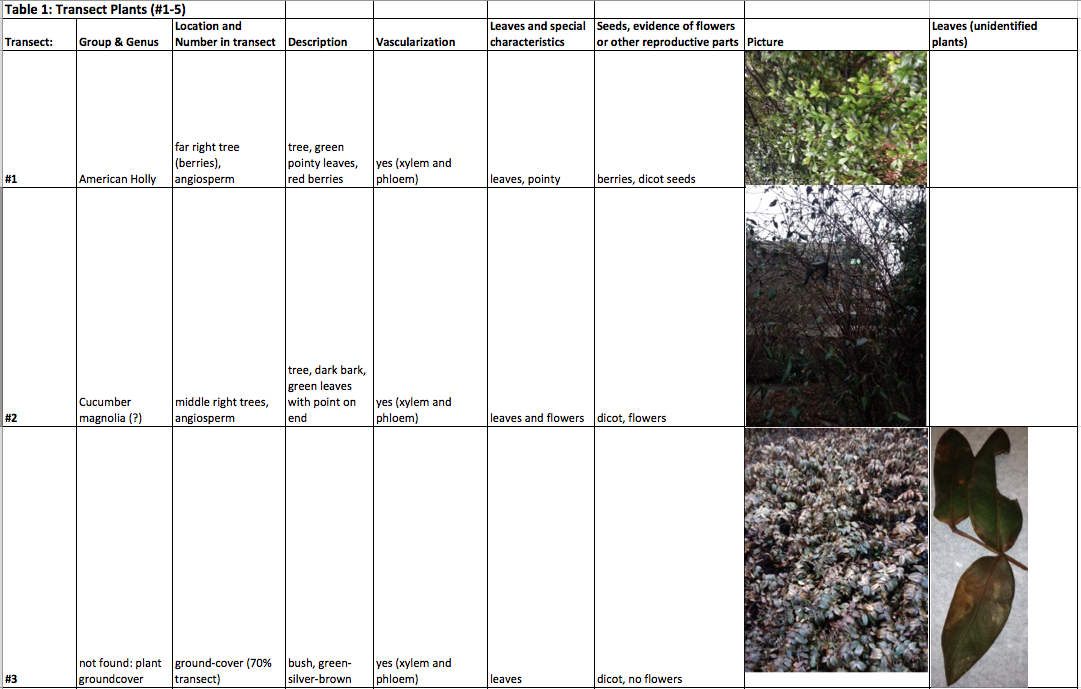

Plant specimen location map

Comparison between moss and lily: the moss (inches) is much shorter than the lily (feet), showing first-hand the benefits of vascularization and the special structures that go along with it, and allow it to grow taller. All of the specific structures of vascular plants (xylem and phloem, etc) gives rise to a plethora of variability in vascular plants.
Fungi: The sporangia cells that bud from the mycelia/hyphae as a whole are visible with and without the microscope. Asexual reproduction is made possible in these molds by the sporangia.
Image 1: Black Bread Mold drawing
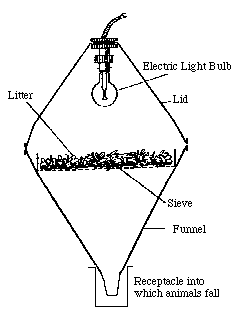
The Berlese funnel contained the leaf litter over the soil, as well as the top layer of soil. No invertebrates were directly seen during collection or manipulation of the sample.
Image 2: Berlese funnel
Conclusions/Future Labs:
Only three out of the five plants in the transect were able to be appropriately categorized, which is probably due to the fact that the leaves are dead and it is difficult to identify a plant this way. Furthermore, the color and possible flowering plants on these specimens were not present, so all of the information needed to categorize them was not available. The vascularization portion of the lab was interesting because, firstly, it asserted the hypothesis that our plants were all vascular, and it also brought into perspective the variability of the plants in the transect. The construction of the Berlese funnel was a learning experience and the invertebrates present in the leaf litter sample, and therefore the ecosystem, will be interesting to observe next week.
NY
2/16/2014
LAB Three
Objective: The objective of this experiment is to understand the characteristics of bacteria, to actively observe antibiotic resistance and explore its implications, as well as to understand how DNA sequences are used to identify different species. The study of the prokaryotes grouped in the domains Bacteria and Archaea--the latter of which tends to grow in the most extreme environments. Cell morphology (shape, size, movement) and cell metabolism in species of these prokaryotes ranges greatly, but polymerase chain reactions with primer sequences specific for coding regions can be utilized to identify species. A hypothesis for the types of prokaryotes that will be found in the normal agar plates and agar plates plus the antibiotic, tetracycline, is: there will be both fungi and bacteria on the agar plate, and the few bacteria that are resistant to tetracycline will be seen on the treated agar plates. Furthermore, more colonies (and probably a lawn) of each species will be seen on the plates that have the more concentrated (less diluted) bacteria sample. Tetracycline is an antibiotic that works against both gram-positive and gram-negative bacteria like E. coli, H. influenzae, and Mycobacterium tuberculosis. It inhibits many of the enzymatic reactions that are essential to vital processes of bacterial cells, including protein synthesis. it can also alter the cytoplasmic membrane, causing leakage of nucleotides and overall inhibition. Because we are studying and identifying the prokaryotes in the Hay Infusion culture , we will know what types of bacteria are present in our transect. There are mesophilic archaea species that live in soil, but there are probably no archaea cells present in the transect, and none that will have grown on both the agar and treated agar plates, since archaea species usually live in the most extreme environments. The classification of microorganisms in this lab is performed in many ways, including by colony morphology, cell morphology, motility, cell wall structure (gram staining), and by using the PCR reaction.
Specific Steps: Part I 1. Check the Hay Infusion Culture and note any changes in smell or appearance. 2. Obtain the handout describing how to characterize colony morphology, and count the total number of colonies on each plate. 3. Choose two bacterial colonies from nutrient agar and one from nutrient agar+tetracycline to observe as wet mounts and gram stains. Part II 1. Study prepared slides of cell morphology. 2. Make wet mounts and cover slip of three samples from three identified colonies, observe at 10x and 40x. 3. Prepare oil immersion slides for samples and observe under the microscope using the oil immersion magnification. 4. Prepare gram stains of the three samples and observe cell shape and motility under 40x and oil objectives.
a) Label the slides and smear one type of bacteria on each.
b) Heat fix the dried slide by passing it through a frame three times, bacterial side up.
b) Cover the smear with crystal violet for one minute, rinse.
c) Cover the smear with Gram's iodine mordant for one minute, rinse.
d) Flood the smear with 95% alcohol for 10-20 seconds, rinse.
e) Cover the smear with safranin stain for 20-30 seconds, rinse.
f) Blot excess water with paper towel and air dry.
5. PCR reaction.
a) Select one bacteria from each of the two plates that have the best characterization.
b) Transfer a single colony to 100 micro liters of water; incubate at 100 degrees Celcius for 10min and centrifuge.
c) Use 5 micro liters of the supernatant in the PCR reaction.
Raw Data: The smell and appearance of the culture: murky green-brown water with growth on the surface and on the bottom of the container. Plant material dead and decomposing, sludge on bottom, most solid settled on bottom, while a darker brown film settles on top with a few floating plant pieces. The smell is less putrid than last week. Transparent, green-brown water.
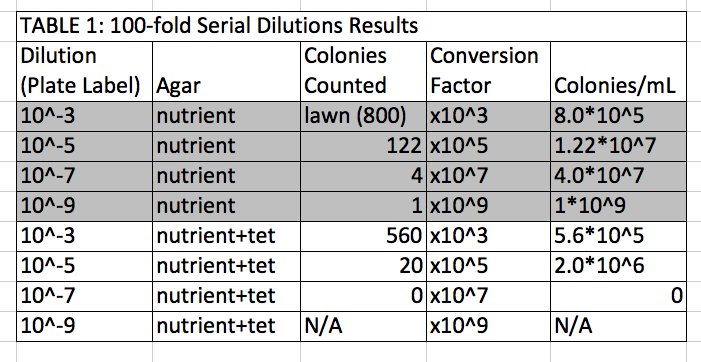
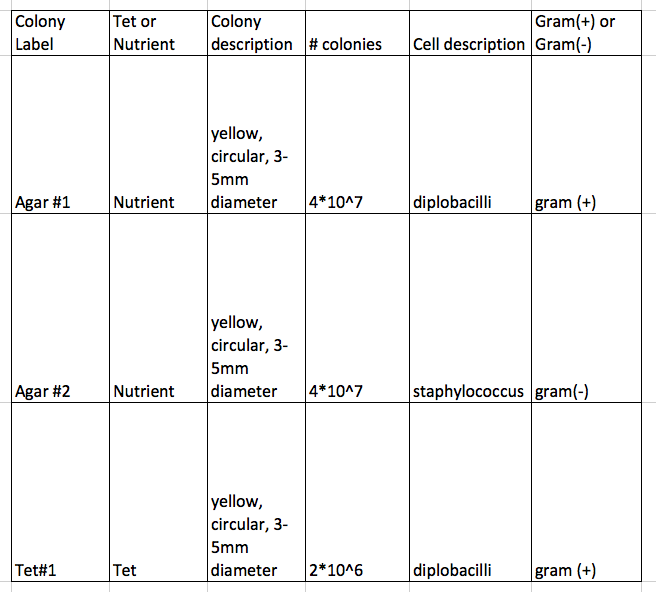
Agar colonies are white and yellow; tetracycline colonies are only yellow (one white on 10^-5 dilution)
- 10^-3 colonies are smaller, more plentiful
- tetracycline kills funghi; bacteria is resistant
- decrease in # of colonies for tetracycline, but they are bigger in tetra plates
- one species unaffected by tetracycline
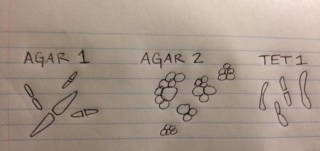
Conclusions/Future Plans:
The purpose of this lab was to get a grasp on the types of bacteria in our hay infusion culture, understand antibiotic resistance and its implications, and explore how DNA sequences are used to identify species of microorganisms. This lab allowed us to observe types of bacteria that are present in our transects, and further study the specific characteristics of these species. The less-strong smell of the hay infusion culture was counterintuitive since the development and growth of microorganisms and the ecosystem in the Hay Infusion culture would be expected to yield a stronger, musty scent. It was expected that no archaea species were found on the cultures, and it was also expected that both bacteria and fungus were found on the plates (both the nutrient agar and tetracycline-treated plates). The objective was addressed in the experiment. Furthermore, the last part of the lab included the preparation of the PCR reaction for DNA sequence identification.
NY
2/9/2014
LAB TWO
Objective: The objective of this experiment is to learn how to use a dichotomous key, as well as understand the characteristics of single-celled Prokaryotes and Eukaryotes, the former of which is grouped into either the Bacteria or Archaea domains. Algae and protists are classified under the Eukarya domain, which is composed of seven lineages. The observation, study, and identification of these single-celled eukaryotes is made easier using a dichotomous key; Ward's Free-Living Protozoa key was used in this experiment. A hypothesis for the types of unicellular eukaryotes that will be found in the Hay Infusion culture from the transect is: a paramecium species will probably be present in the culture, as well as green algae species, since the transect is covered in plants and trees and is also a moist environment, given the winter weather. Since we are studying and identifying the organisms in the Hay Infusion culture under a microscope, we will know what types of unicellular eukaryotes are present in our transect. The dichotomous key will be used in the process, and the niches of the ecosystem created with the jar of matter from the transect will be explored.
Specific Steps:
1. Make a wet mount of a sample with known organisms and observe with the microscope at 4x and 10x.
2. Focus on one organism, characterize it and identify it; repeat for another.
3. Bring the Hay Infusion culture to the work area and take two samples--one from the top floating material and one from the material that has settled to the bottom of the culture.
4. Characterize, identify, and describe two organisms from each of the samples from the two niches.
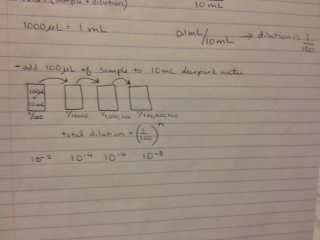
5. Make serial 100-fold dilutions of the culture in four nutrient agar and four agar plus tetracycline plates, which will be 10^-3, 10^-5, 10^-7, and 10^-9 dilutions.
6. Incubate the agar plates at room temperature over the next week.
Raw Data:
The smell and appearance of the culture: murky green-brown water with growth on the surface as well as on the bottom of the container. The top growth is composed of green leaves and brown/dead plant matter, while the bottom portion is brown and sludge-like.
If the hay infusion culture were to be observed over the next two months, I would predict that the strong, putrid smell would get stronger, and that the mold/algae would grow even more, and that all of the organisms would be more plentiful, since they would have had more time to reproduce. The selective pressures that probably affected the compositions of the sample most likely include the access to sufficient food and space, as well as access to enough light for the photosynthesizing microorganisms that depend on it for energy. Some organisms found in the hay infusion culture, like paramecia, feed on bacteria, algae, and yeasts; the fact that these cells may compete for resources and/or feed on each other must be taken into account when assessing the various cells that were identified from the culture.
The two samples were taken directly from the top growth and from near the bottom sludgy matter. The organisms that would appear near to and away from the plant matter would differ, depending upon what they eat and how they metabolize and grow. The algae and protists are plant-like and animal-like, respectively, so they will live in different environments; for this reason, it is important that the two samples varied.
Organisms from bottom sample:
1) mobile (fast), colorless, 120 micrometers - unidentified
2) motionless/rotating/turning, greenish color, spherical, 30-35 micrometers: PANDORINA ALGAE, photosynthesizing
Organisms from top sample:
1) motionless/rotating/turning, greenish color, spherical: 15-20 micrometers: EUDORINA ALGAE, photosynthesizing
2) oval-shaped, 2 flagella, motile, green: CHLAMYDOMONAS ALGAE, photosynthesizing
Organisms share a suite of 5 fundamental characteristics, of which include: energy, cells, information, replication, and evolution. The Chlamydomonas Algae acquires and uses energy from the sun, as most species are obligate phototrophs. They are motile, unicellular flagellates that are found in stagnant water or damp soil. Chlamydomonas reproduces both sexually and asexually, and their lineage diverged from land plants over one billion years ago. Their DNA genome sequence is used to further study the use and biogenesis of chloroplasts and eukaryotic flagella.
Figure 1a/!b: Hay Infusion Culture
Figure 2: Unidentified Cell
Figure 3: Pandorina
Figure 4: Eudorina
Figure 5: Chlamydomonas
Figure 6: Serial Dilutions






Conclusions/Future Plans: In conclusion, this experiment taught the value in describing and identifying unicellular eukaryotes in the hay infusion culture. Understanding the relationship between and among the environmental niches--on the surface and settled onto the bottom--is an important part of analyzing and studying an entire ecosystem. On the whole, three species of algae were found in these niches, and it was difficult to find and/or identify other types of microorganisms. The objective was definitely addressed in the experiment. Furthermore, the last part of the lab included the preparation of serial dilutions in nutrient agar plates with and without the antibiotic, tetracycline.
NLY
2/6/14, lab 1 notes
Great job!!! Make sure you include pics from lab 1 and lab 2 by Sunday. Also, start working on building a map of your transect to detail your land and where your samples are taken from. We will talk about this more Wednesday.
AP
1/31/2014
LAB ONE
Objective: The objective of this experiment is to identify and experimentally explore a 20 by 20 foot niche on the American University campus, in order to explore a community of organisms inhabiting a specified ecosystem. Some questions to explore concern the components of an ecosystem (biotic and abiotic), and what types of factors must be included in the study of an ecosystem. Because the assigned transect of land is characterized by plants and small animals, and also located on a University campus, it is hypothesized that the ecosystem will be composed of both natural (organic) and synthetic materials. The microorganisms that will be studied and identified using microscope in lab will probably range from bacteria like staphylococcus and e.coli to certain species of fungi. This experiment will give insight into what types of organisms inhabit this transect, which types can be assumed to inhabit other spaces on campus, and will also help in the understanding of the structure, characteristics, and dynamics of an ecosystem and its living (and non-living) parts.
Specific Steps: 1. A 20 by 20 foot transect was assigned to the group, and the types of biotic and abiotic components were identified. 2. A 50 mL sterile conical tube was filled with a sample of soil and surface plant matter that best represented the entirety of the transect. 3. 12 grams of the sample was placed in a plastic jar and filled with 500mL deer park water. Next, 0.1g dried milk was added and the entire mixture was agitated for 10 seconds. The open jar was placed in a safe place in the laboratory.
Raw Data: The transect is located near the American University Bender Arena tunnel, and is surrounded by cement sidewalks (which are not included in the transect). It is clearly a man-made garden due to its location, but it appears to be both healthy and well-kept. It is wintertime so the plants/trees have lost their leaves and buds, and the dead leaves are decomposing on the ground. A list of biotic and abiotic components is as follows: Biotic: bushes, small trees, soil, topsoil, bare trees, berries, bird nest, small male cardinal, tall grasses Abiotic: steel light, soda cup
Figure 1:

Conclusions/Future Plans: In conclusion, the AU transect project will be ongoing, and many components of the ecosystem will be explored in more depth: microorganisms will be identified under a microscope, and specific gene sequences will be amplified. Furthermore, with the passing of time (and weather), the transect will be monitored by the group. Both the biotic and abiotic aspects of the ecosystem are factors; the plants, animals, invertebrates, bacteria, and protists interact, and the fact that humans are constantly exposed to the ecosystem--and vice versa--introduces a unique dimension to the study of this environment. More will be learned about our multifaceted ecosystem, and our area will eventually be compared with the assigned transects of other groups.
NLY
1/22/2014
I have successfully logged in.
NLY