User:Nicolas Lubitz/Notebook/Biology 210 at AU
Embryology lab: 3/22/14
Introduction Embryology is a huge field in biology and it is often used to determine relationships between groups of organism. During the development of embryos a lot of similarities between reptiles, fish, birds and mammals can be observed. In this embryology lab our purpose was to understand the different stages of embryonic development and to come up with an experimental set up to study embryonic development when different environmental conditions act on the embryos. In our case we used Zebrafish, model organisms commonly used in biological studies. First we had to read a published scientific paper about zebrafish development and based on the reading a hypothesis had to be made. The independent variable "permanent light" was assigned to my group. So our goal was to examine the effects of permanent light on embryonic development in zebrafish.
Hypothesis: In our assigned Zebrafish paper, the results showed that zebrafish exposed to permanent light and darkness show developmental delays or missstructures in their retinas. Although, these damages or delays will be repaired after exposure to normal light conditions. Based on these findings our hypothesis is the following: Permanent light will affect eye movement as well as eye diameter and will also affect behavioral patterns in zebrafish embryos.
Experimental setup/procedures In order to establish an accurate experiment it was important to have a control group in addition to our treated group. To do so, we were given 40 zebrafish in total, 20 for the untreated control group and 20 for our treated group. The control group, 20 fish in a petri dish with 20ml water, was exposed to a normal day-night rhythm for light. Our treated group, also 20 fish with 20ml water, was exposed to permanent light under a lamp. The main part of the experiment was to observe both groups every three days for the following criteria: hatched animals, animals still in egg, alive animals, dead animals, movement, amount of yolk, eye-pigmentation and eye movement, morphological development, heart rate, body structures, tail length and total body length. Then both groups are to be compared. Additionally, the water had to be changed every three days in order to prevent contamination. Also, hatched egg cases had to be removed because they would have grown molds. Any dead embryos found over the course of the experiment had to be preserved in formaldeyhde for future obersavation. Furthermore, week 2 was the time when we sacrificed three individuals from both groups to fix them on slides for better microscopic observations.
Results:
Conclusion/discussion:
Based on our results a general conclusion can be made:
We could not find any significant difference between the group other than the fact that the permanent light group was faster and more active than the control group. A possible explanation is that these animals might be more active during the day time and because our experimental group has been constantly exposed to light they stayed in the active-day time state.
Eye diameter and total length are basically the same and no significant difference could be observed. Both groups developed normally and no delay could be observed.
A possible reason for that might have been the lamp we used. In other words, the light might have been to weak or our control group was exposed to light too long as well, so they developed the same way our experimental group did.
The only time we were able to observe the heart beat in both groups was day 2 of observations. The control group showed a heart rate of 120/min and the light group showed 132/mins. But, it is unlikely that this small difference is due to any experimental values.
At the end of the experiment virtually all individuals died in both groups (this was common amongst other groups as well). One explanation might be the room in which we conducted the experiment, the temperature might have been to high for the zebrafish, which means the developed faster and therefore used all of their yolk, which without additional feeding, resulted in starving.
Our hypothesis could not be succesfully tested. Based on the results we found, the null hypothesis seems to be true. For future experiments it will be helpful to focus more on the method of permanent light exposure. A different lamp and a different room should be used and the temperature should be kept at a natural value to prevent starving or additional feeding should be part of the experiment.
Lab 6: 3/16/2014
Mini - Lab: DNA sequencing based on microbiology PCR This labs purpose was to analyze a DNA sample from out bacterial colonies from lab3 (microbiology) via PCR and Gel-electrophoresis.
Steps:
The DNA sample was heated to 100degrees celsius in order to breake hydrogen bonds, it was then cooled down up to 50 degrees Celsius to allow primers to bind to DNA and then heated again to 72 Degrees Celsius in order to allow DNA Polymerase to work on the DNA strand. After that we seperated DNA strands of different length via gel electrophoresis, the DNA material was then sent to a laboratory to sequence it.
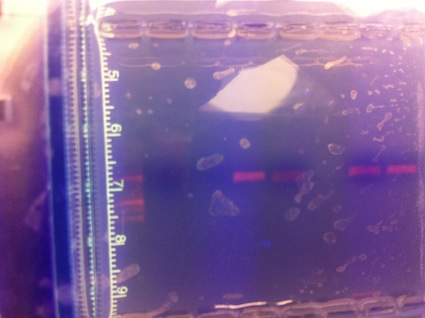
After we received the results from the laboratory, we used the given sequence to find out which bacterial species we encountered on our petri dish. To do so, we visited a specific online database, which tells you the organisms name when you type in the DNA sequence.
Results: Our DNA sample did not show any results.
Conclusion: Based on errors during the experiment could be possible reasons: Mistakes during collection of DNA sample from petri dish. Mistakes during PCR.
Lab 5: 2/27/14
Introduction In this lab our goal was to understand the importance of invertebrates within an ecosystem. It was also key to understand what they are made of, specialized cells and body parts and how simple systems evolved into more complex systems. In addition, this lab was a great opportunity to get an insight into the great diversity of invertebrates.
Procedures:
Procedure 1: observe three different types of worm under the miscrocope
- Planaria
- Nematodes
- Annelida => Describe movement in correlation with body structure
Procedure 2:
- Examine invertebrates collected with the Berlese Funnel. (Berlese Funnel was setup the week before. Soil and leaves from our transect were put into a funnel that was placed above a petri dish of ethanol Invertebrates living in the soil are supposed to react negatively to light and will therefore move downward the funnel until they fall into the dish.
- The ethanol will then preserve them for later observation) Measure length and identify and describe organisms as best as possible.
Procedure 3:
- Observe, describe and identify vertebrates on your transect
- Discuss abiotic and biotic factors on your transects, influencing the vertebrates niches.
- create food web
Results:
Results for procedure 1: Planaria Flat Worm: ----------------------------- Acoelomate tissue layer of Planaria
Acoelomate organisms have no body cavity. Because of their body structure these flatworms move wave-like. The head moves forward and the rest of the body follows by contractions
Nematodes (Groundworms): ------------------------------------------------------------ Pseudoacoelomate tissue layer (no picture)
structure provided by cuticula. "Snake-like" movements
Ringed Worm "Annelida:" ---------------------------------------------------------------------- Acoelomate tissue layer of Annelida
Annelida moves with contractions. Head regions moves forward while other body parts will be pulled following the head.
Results for procedure 2:
Insects appear to be the most abundant organisms in our soil sample. Diplura and Insects seem to be the largest organisms.
Results for procedure 3:
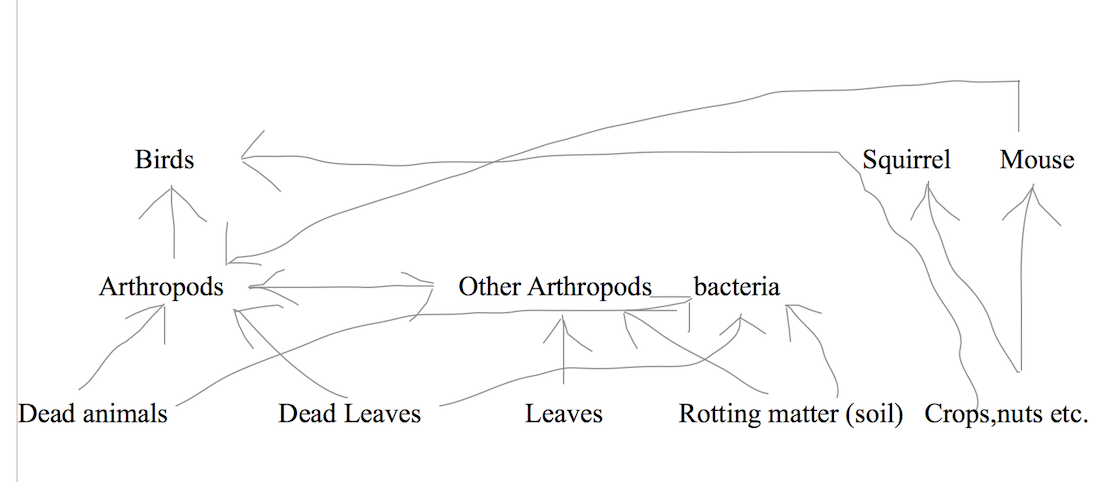
Species of Vertebrates identified on the transect:
Spizella arborea - American Tree Sparrow
Progne subis - Purple Martin
Black Squirrel
Eastern Grey Squirrel - Sciurus carolinensis
House Mouse - Mus musculus
The vertebrates found on the transect are chordates of the class Aves or Mammalia, birds and mammals.
Possible biotic and abiotic factors are:
trees and branches for nesting sites (squirrels and birds)
food availability (all vertebrates on transect)
temperature (all vertebrates found on transect)
soil consistens for borrowing holes and nutrients (all vertebrates found on transect)
trees that provide shelter and protection from wind and temperature (all vertebrates found on transect)
other predatory animals (all vertebrates on transect)
Food web:
Lab 4: 2/28/14
Introduction: This lab's purpose was to learn more about the biodiversity of plants and fungi. We also wanted to examine their structures and the different functions of their parts.
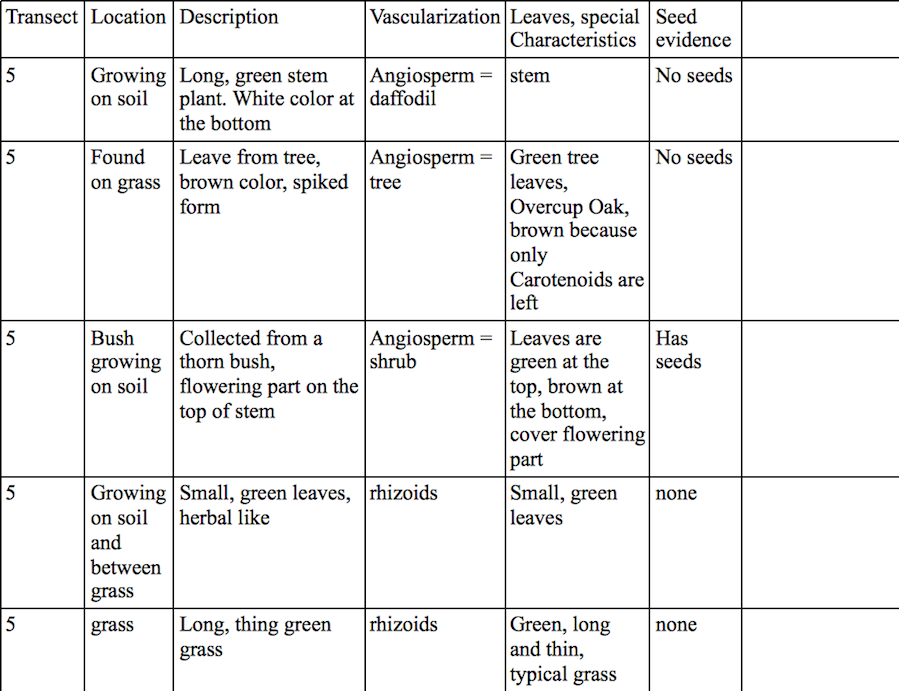
Procedure: For this lab we collected five plant samples from our transect and described and analyzed them for structure, vascularization, characteristics and seeds. We also analyzed fungi and their structures.
Results:
Most plants from our transect were flowering angiosperms. The green grass and the herbal-like plant were rhizoids. Only the thornbush had seeds, which appeared to be monocot. Leaves are either attached to the wood part of the plant, as in trees, or grow out of the soil and are connected to the roots, as in grass.
Results for fungi analysis:
In fungi Sporangia are important for the production of spores, units of asexual reproduction, and can be either multicellular or unicellular. They are important because without them no fungi could reproduce.
The fungi we examined under the microscope looked like this: It shows the clear cellular connection between all individual groups of fungi cells. They appear in a fibre, common in most fungi species. Each group of cells is able to move and other cells will follow enabling the fungi to move to new environments.
Lab 3: 2/16/14
Introduction: In this microbiology lab we tried to understand characteristics of bacteria and their ecology. We also tried to observe antiobiotic resistance in the bacteria colonies from our delutions (see lab 2). In order to do so, we compared bacteria growing on nutrient and bacteria growing on nutrient + antibiotic. The third part of this lab was to use DNA sequencing to identify species of bacteria from our colonies with the help of PCR. Referring back to our hay fusion, we observed a change in smell and appearance. Ongoing decompostation by microorganisms seems to be the reason.
Procedures:
Procedure 1: Observing the bacteria colonies on the petri dishes:
- observe all petri dishes ( nutrient and nutrient + antiobiotic)
- estimate number of bacteria colonies
Procedure 2: compare 2 colonies from nutrient argar plate and 1 nutrient + antibiotic argar plate (total of 3 colonies)
- extract a small part of each colony
- place on microscope slide
- observe under the microscope
procedure 3: Produce gram stains for the three bacteria colonies. Gram stains help to make bacteria more visible by incorporating dye into the bacterias cell wall. Depending on the components of the cell wall, species of bacteria will be either gram positive or negative ( dark purple or light pink).
- place wet mount of each petri dish sample ( 3 in total, see above) on a slide
- heat slide in open flame
- cover sample with crystal violet (1 minute)
- wash slide with water
- cover slide with gram's iodine mordant (1 minute)
- was slide with water
- decolorize by treating slide with 95% alcohol
- treat slide with safranin stain
- wash slide and dry with paper towel
- observe under microscope and describe the bacteria's morphology
procedure 4: prepare samples for PCR (Polymerase chain reaction) to sequence DNA samples in order to identify a species
- put bacterial colony into 100microliters of water ( sterile tube)
- heat the tube at around 95c for ten minutes
- centrifuge the sample
- fill 5 microliters of supernatant into the reaction
Results/Data:
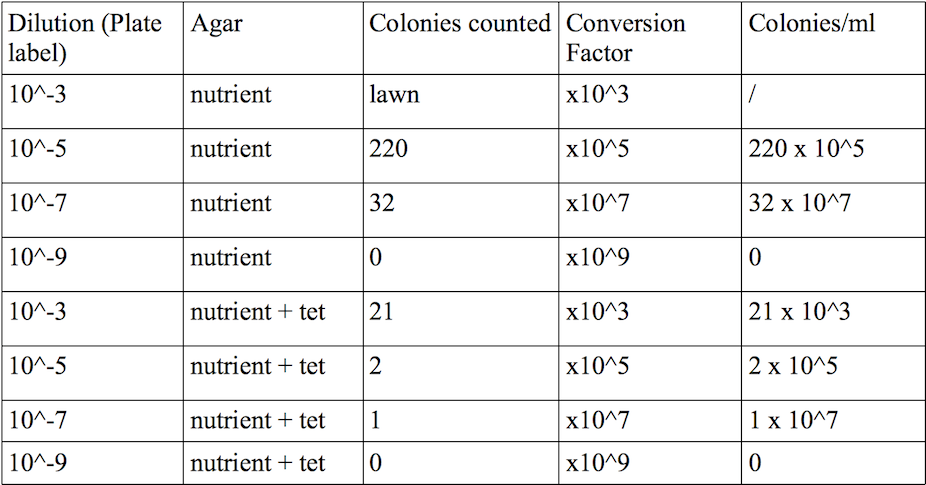
1.Results for procedure 1: (Lawn: too many bacterial colonies, so counting is impossible
Results for procedure 2: As we can see the number of bacteria growing on nutrient without the antibiotic is much larger than the number of bacteria growing on nutrient + antiobiotic. Usually, antibiotics attack the bacteria's cell wall. This disrupts the entire bacterium. The few bacteria that actually grow on the plate with the antibiotic have to have a mutatin, that helps them to fight the antibiotic, so it is not longer able to attack their cell walls.
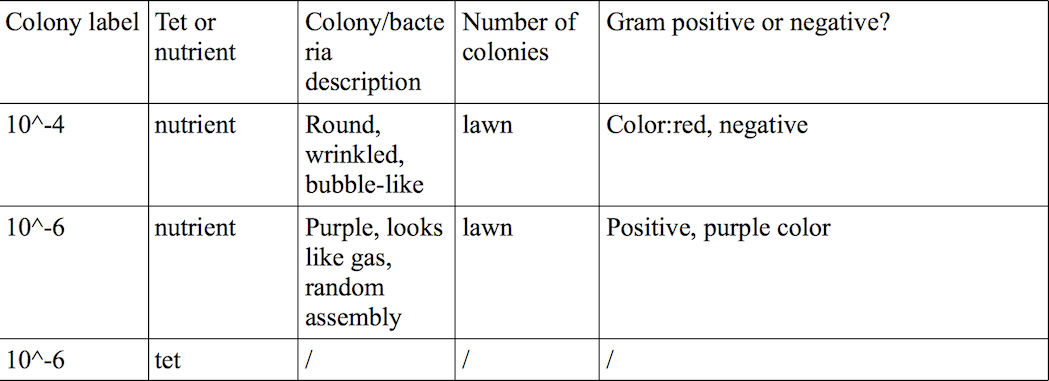
Results for procedure 3:
For our third sample we could not find any results. Mistakes during our gram staining could be the reason.
Lab 2: 2/7/14
Introduction: This lab we examine the different protist and algae species from our hay fusion, which we prepared last lab. The main goal is to define different organisms and their ecological niche within our hay fusion sample. Our hay fusion represents a whole ecosystem with several different species taking up distinct ecological niches.
Procedures: In order to define different organisms of different niches, we take samples from our hay fusion from different parts. Looking at the fusion, we can see that a layer of soil has developed on the bottom of the beaker, other matter like plants, green molds, algae and grass grow up to the surface of the water. Describing the overall appearance we can say the smell reminds of a pond. It also smells rotten. A slight layer of soil on the bottom provides the bases for some vegetation to grow throughout the fusion.
Taking Organisms from different niches: Different parts of the sample ( for example areas close to the plants, areas on the soil) provide different niches for different organisms. By carefully taking a sample of water closer to the plants and closer to the soil, we should be able do observe different organisms under the microscope. After taking a sample, we use a dropper to carefully place a drop of liquid onto a microscope slide.
By taking organisms from the top, the middle part and the bottom of the sample we can identify the following organisms:
Top film: no organisms
middle part (plant matter): Chlamydomonas, size: very small - 5-10 nanometers
Peranema, size:medium - 30 nanometers
bottom part: Chlamydomonas, size: very small - 5 nanometers
Spirostonum, size: large - 55 nanometers
Eudorina, size: small - 10 nanometers
Didinium Cyst, size: small - 12 nanometers
Paramecium Aurelia, size: very large - 110 nanometers
Chlamydomonas for example are unicellular, motile green algae that photosynthesize. Being autotroph they do not feed on other organisms, but are indeed prey to other organisms which digest unicecullar organisms.
The protist Peranema is a eukaryotic organism, not capable of photosynthesis. Instead these unicellular organisms prey on other cells, like bacteria and yeast.
Additional Information/discussion
I choose the organism Peranema to describe how this species meets all the needs of life according to our textbook. Peranema consists of cells, in fact it is a unicellular eukaryote. Additionally, this organism ingests other cells to provide itself with energy. Peranema uses asexual reproduction to establish new cells. By giving identical genetic information to the next generation, Peranema process hereditary. Furthermore these organisms undergo evolution. By filling out a niche within an ecosystem Peranema are exposed to environmental conditions and natural selection.
After two month our hay fusion sample would look different. Being a enclosed ecosystem without any influence from outside (no new individuals enter, no individuals leave) the hay fusion provides limited space and resources as an ecosystem. In addition the populations of organisms cannot grow much larger because they space is limited. Most of the plant matter would be used after a while.
Possible selective pressures that affect the composition of our sample are: 1. environmental conditions like temperature, water consistens, plant matter and soil. Different individual organisms are better adapted to certain conditions because of their allels. On a larger scale, our sample provided the possibility of establishing ecological niches. Nutrients in the soil make it possible for organisms who live off the soil to establish a population. The plant matter in our sample provided living space and nutrients for organisms making a living off using plants as an energy source. 2. predator - prey relations establish a food web/chain in our sample. Autotrophs are under selective pressure because of other organisms praying on them.
conclusion/future plans:
As we could observe, the biodiversity in even such a small sample "ecosystem" is huge. Different niches are occupied by highly specialized multicellular and unicecullar organisms.
For the future we prepare petri dishes with nutrient and petri dishes with an antibiotic to grow different bacteria and fungi on it. We prepare some samples of our hay fusion and dilute them. (Diagram below)
DILUSION:
d= Volume of samples/ total(sample+dilution)
Tube 1---------------------------------------------------------------------Tube 2-----------------------------------------------------------------Tube3---------------------------- ----------------------------------------------------------------------------Tube 4
1/100--------------------------------------------------------------------1/100-----------------------------------------------------------------1/100-----------------------------------------------------------------------------1/100
In total: 1/100 => 1/10.000 => 1/1.000.000 => 1/100.000.000
2/6/14, lab 1 notes
Great job, Nico! Some notes:
-Make sure you date each lab
-Sign each lab at the end with initials
-Make sure you include a conclusion for each entry. Refer to email I sent with an attachment about lab notebook organization
Good work!
AP
Lab 1 1/31/2014
Introduction/purpose'
Our planet provides an astonishing biodiversity. Millions of organisms inhabiting different ecological niches, billions of years of evolution have created a sheer limitless number of organisms. In this lab we examined how organisms interact with other organisms and with their environment. Each research group was given a different 20x20 ft transect on the AU campus. Our goal was to examine and compare the different organisms living on the different transect by taking samples of the biomass that is most representing the transect.
GENERAL INFORMATION ABOUT OUR TRANSECT:
-location: Washington D.C, American University campus, quad. Area called Eric. A Friedheim Quadrangle
-Topography: Flat, grass and concrete, thornbushes on soil next to the grass, stone bench on the concrete.
-Biotic Factors: grass, bushes, humans, organisms (squirrels, insects, micro organisms)
-Abiotic Factors: concrete, bench, soil, litter
PROCEDURES: 1.Collect a sample that is representative for the whole transect with a sterile 50ml conical 2.For lab 2, create a hay fusion, on which the collected organisms can grow and live: 3.weigh 12 grams of your sample, mix with 500ml deer park water in plastic jar and add 0.1gm milk. Shake, place safely and wait one week