User:Maria Ochiai/Notebook/Biology 210 at AU
Zebra Fish
February 23, 2016
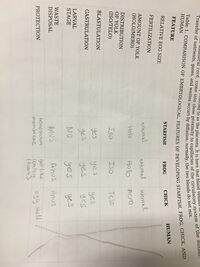 Table 1: Comparison of Embryological Features of Developing Starfish, Frog, Chick, and Human
Table 1: Comparison of Embryological Features of Developing Starfish, Frog, Chick, and Human
Day 2
CONTROL:
Dead eggs - 0
Living embryos still in egg cases - 9
living hatchlings (larvae) - 8
Dead hatchlings - 0
Empty - 3
 Figure 2: Hatched with embryo sac attached.
Figure 2: Hatched with embryo sac attached.
FLUORIDE:
Dead eggs - 1
Living embryos still in egg cases - 8
living hatchlings (larvae) - 6
Dead hatchlings - 0
Empty - 5
Day 5
CONTROL:
Dead eggs - 0
Living embryos still in egg cases - 0
living hatchlings (larvae) - 17
Dead hatchlings - 0
Empty - 3
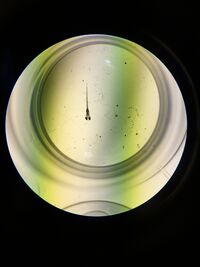 Figure 3: Hatched zebra fish without its yolk sac
Figure 3: Hatched zebra fish without its yolk sac
FLUORIDE:
Dead eggs - 1
Living embryos still in egg cases - 2
living hatchlings (larvae) - 12
Dead hatchlings - 0
Empty - 5
 Figure 4: Embryo/Hatched stage
Figure 4: Embryo/Hatched stage
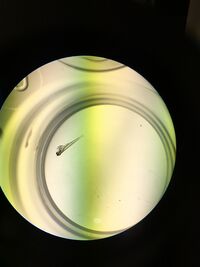 Figure 6: Hatched with yolk sac attached
Figure 6: Hatched with yolk sac attached
Day 7
CONTROL:
Dead eggs - 0
Living embryos still in egg cases - 0
living hatchlings (larvae) - 17
Dead hatchlings - 0
Empty - 3
Description 1: Total length: 15.5 micrometers at 4x. Tail length: 10 micrometers at 4x. Has pectoral fins, swim bladder, and protruding jaw
Description 2: Total length: 15 um at 4x. Eye movement, developing swim bladder, pectoral fin, protruding jaw. Active
Description 3: Total length: 14 um. Tail length: 10 um. Pectoral fins, developing swim bladder. Very active
FLUORIDE:
Dead eggs - 1
Living embryos still in egg cases - 0
living hatchlings (larvae) - 14
Dead hatchlings - 0
Empty - 5
Description 1: Total length: 13.5 um at 4x. Tail length: 10 um at 4x. Eye to eye length: 2 um at 4x. Has pectoral fin, developing swim bladder, opens and closes mouth. Very active, swims jerkily, tail undulates
Description 2: Total length: 18 um at 4x. Tail length: 11 um at 4x. Eye to eye: 1.9 um at 4x. Has pectoral fin, developing swim bladder. Very active, moves his fin very often
Day 9
CONTROL:
Dead eggs - 0
Living embryos still in egg cases - 0
living hatchlings (larvae) - 17
Dead hatchlings - 0
Empty - 3
Description 1: Total length: 0.3 cm. Tail length: 0.2 cm
Description 2: Eyes are very close together, underdeveloped swim bladder, very skinny, swims with only one fin
Description 3: Total length: 0.35 cm. Tail length: 0.25 cm
Description 4: Total length: 0.35 cm. Tail length: 0.25 cm
Description 5: Total length: 0.3 cm. Tail length: 0.2
FLUORIDE:
Dead eggs - 1
Living embryos still in egg cases - 0
living hatchlings (larvae) - 12
Dead hatchlings - 2
Empty - 5
Description 1: Heartbeat: 1 beat/second. Underdeveloped swim bladder
Description 2: small. total length: 0.2 cm. tail length: 0.15 cm
Description 3: total length: 0.25 cm. tail length: 0.2 cm. continued to sink headfirst to the bottom, kept jerking to try and right himself
Description 4: underdeveloped swim bladder, seems to only have the use of one fin
Description 5: total length: 0.3 cm. tail length: 0.25 cm. underdeveloped swim bladder, head keeps sinking to the bottom involuntarily so that it vertically collapses on itself
Day 12
CONTROL:
Dead eggs - 0
Living embryos still in egg cases - 0
living hatchlings (larvae) - 7
Dead hatchlings - 10
Empty - 3
Description 1: total length: 15 um. tail length: 12 um. heart beat: 27 beats in 30 sec. 2.1 um eye-to-eye length. 1.3 um eye diameter
Description 2: total length: 15 um. tail length: 11 um. heart beat: 22 beats in 30 sec. 2.5 um eye-to-eye length. 1.4 um eye diameter. bladder is bigger compared to fluoride
FLUORIDE:
Dead eggs - 1
Living embryos still in egg cases - 0
living hatchlings (larvae) - 5
Dead hatchlings - 9
Empty - 5
Description 1: total length: 14um. heart beat: 28 beats in 30 sec.
Description 2: total length: 13.8um heart beat: 33 beats in 30 seconds.
Day 14
CONTROL:
Dead eggs - 0
Living embryos still in egg cases - 0
living hatchlings (larvae) - 1
Dead hatchlings - 16
Empty - 3
Description : upper body: 5.5 um. total length:15.5um . tail length: 11 um. eye-to-eye: 2.5 um. eye diameter: 1.2 um. heart beat: 23 beats in 30 sec. the bladder is bigger
FLUORIDE:
Dead eggs - 1
Living embryos still in egg cases - 0
living hatchlings (larvae) - 0
Dead hatchlings - 14
Empty - 5
NOTE: Fluoride fish had a yellow-ish tint on their clear bodies. Fluoride fish were more sluggish compared to the control, but had agitated eye movement.
Purpose: Understand embryology and development starting from the egg and into adulthood.
Conclusion: The fluoride zebrafish had slower growth and were shorter in body length. The fluoride ones had faster heart beats compared to the control. The control developed faster that by Day 5, all the eggs were hatched and gotten rid of their yolk sacs. The fluoride fish had more twitchy-ness in their eyes and had smaller gall bladder compared to the control.
MO
Invertebrates
February 16, 2016
Berlese Funnel Set Up: First, put a piece of screening material in the bottom of the funnel -- this is to catch leaf little sample so it will not interfere with the observation for invertebrates. Then secure the screening material by the corner using tape. Then pour 25mL of 50:50 ethanol and water into a 50mL conical tube. Attach the conical tube to the funnel using parafilm and regular tape so the solution will not evaporate. Add the leaf little sample into the funnel. Then, using a ring stand, set the funnel and place it under a 40 watt lamp and leave it for a week.
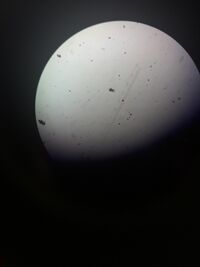 Figure 1: Picture of flea at 4X objective.
Figure 1: Picture of flea at 4X objective.
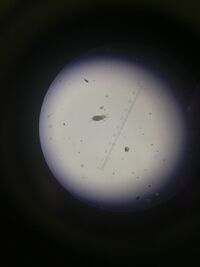 Figure 2: Picture of lice at 10X objective.
Figure 2: Picture of lice at 10X objective.
 Figure 3: Table of invertebrates found in the Berlese Funnel.
Figure 3: Table of invertebrates found in the Berlese Funnel.
 Figure 4: Pictures of vertebrates and their food web.
Figure 4: Pictures of vertebrates and their food web.
These organisms are a community because they share the safe diet and live in the same transect. The carrying capacity will not be as large because the transect is small and if the population increases in the transect, then the species will leave the transect to find new homes. House sparrows are herbivores. and the rest of the vertebrates listed are omnivores.
Purpose: Observe different invertebrates using the Berlese Funnel method and understand the process. Identify the organisms using dichotomous key.
Conclusion: We only found 2 invertebrates in the Berlese Funnel: possibly an error because invertebrates live in the soil so we might not gotten the deep soil. So next time, digging to get the soil and not just get the top soil. Since only 2 were found, it goes against the hypothesis of biodiversity in transect 3. Overall, the experiment showed the evolution from the microorganisms (protists and bacteria) to the macroorganisms (plants) . The transect contains all kinds of organisms and if such transect is gone, then the organisms will lose their habitats can affect the ecosystem.
MO
Plants and Fungi
February 13, 2016
Characteristics of Plants Collected from the Transect
1. Located in the Southeast side of the transect. The plant was found close to the ground. The leaves are paired and grows in even intervals on the stem of the plant. The leaves are oval-shaped and have rounded tips and net-like veins. This plant performs vascularization looking at the whiter spots on Figure 1a possibly a xylem. Also, the inside of the leaf is spaced out and there are clear, thick veins. The edge also has yellow spots possibly indicating that there are stomata. The plant has a reproductive mechanism of alternation of generations based off of angiosperms since this plant is an angiosperm so the sporophyte stage dominates the cycle.
 Figure 1: Picture of 1. plant with oval-shaped leaves.
Figure 1: Picture of 1. plant with oval-shaped leaves.
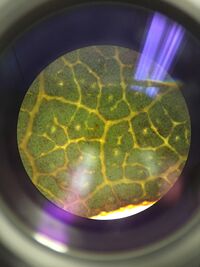 Figure 1a: Picture of plant 1. through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
Figure 1a: Picture of plant 1. through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
2. Located in the Northeast side of the transect. The leaves are furry (almost have itchy feeling to touch) and have pointed tips and net-like veins. The leaves grow in pairs. Vascularization is evident from Figure 2a. The leaves possibly have xylem and phloem because of the circular bundles that are everywhere on the leaf. The picture also shows how some parts of the leaf have space where there are more light streaming from it. The plant has a reproductive mechanism of alternation of generations based off of angiosperms since this plant is an angiosperm so the sporophyte stage dominates the cycle.
 Figure 2: Picture of 2. plant with furry leaves.
Figure 2: Picture of 2. plant with furry leaves.
 Figure 2a: Picture of 2. plant through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
Figure 2a: Picture of 2. plant through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
3. Located in the Northern edge of the transect. The plant bears red berries, which grows between two leaves at the tip of the stem. The leaves are deep green in color and have net-like veins. The leaves also have nice tough, hard, smooth layer like wax coating over them. The leaves grow in pairs. From Figure 3a. it looks like this plant has vascularization because of phloem, which are the small, dark circles. The plant has a reproductive mechanism of alternation of generations based off of angiosperms since this plant is an angiosperm so the sporophyte stage dominates the cycle.
 Figure 3: Pictures of 3. plant with red berries.
Figure 3: Pictures of 3. plant with red berries.
 Figure 3a: Picture of plant 3. through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
Figure 3a: Picture of plant 3. through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
4. Located in the Western side of the transect. The leaves are pointy all around on the upper part of the leave. It almost looks like mistletoe leaves. The leaves have net-like veins. The leaves grow in pairs. From Figure 4a, the plant performs vascularization because the small, circular bundles are likely to be phloem. The plant has a reproductive mechanism of alternation of generations based off of angiosperms since this plant is an angiosperm so the sporophyte stage dominates the cycle.
 Figure 4: Picture of 4. plant with pointy leaves.
Figure 4: Picture of 4. plant with pointy leaves.
 Figure 4a: Picture of 4. plant through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
Figure 4a: Picture of 4. plant through the microscope. This shows the vascularization in the leaves of the plant.
_____________________________________________________________________________________________________________________________________________________________
5. Located in the Southwestern side of the transect. the branch has a grey color. There is a seed or flower bud growing on the tip of the branch. The leaves have net-like veins and grow in pairs. From Figure 5a, this plant has vascular bundles arranged in a ring. The plant has a reproductive mechanism of alternation of generations based off of angiosperms since this plant is an angiosperm so the sporophyte stage dominates the cycle.
 Figure 5: Picture of 5. plant with buds at the tip.
Figure 5: Picture of 5. plant with buds at the tip.
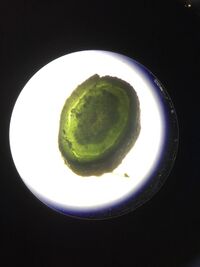 Figure 5a: Picture of 5. plant through the microscope. This shows the stem of the plant to observe for vascularization and mechanism.
_____________________________________________________________________________________________________________________________________________________________
Figure 5a: Picture of 5. plant through the microscope. This shows the stem of the plant to observe for vascularization and mechanism.
_____________________________________________________________________________________________________________________________________________________________
There were two seeds from the transect: the red berry from one of the plants collected and a black seed. The red berry was a dicot because it had two cotyledons. The black seed was a monocot because it had one cotyledon.
 Figure 6: Inside of red berry. It shows two cotyledons in the middle of the seed.
Figure 6: Inside of red berry. It shows two cotyledons in the middle of the seed.
 Figure 7: Inside of the black seed. Although the picture is not distinct, the cotyledon is the white part of the inside. When it is monocot, the cotyledon is usually shown on one side/corner of the seed on the inside.
_____________________________________________________________________________________________________________________________________________________________
Figure 7: Inside of the black seed. Although the picture is not distinct, the cotyledon is the white part of the inside. When it is monocot, the cotyledon is usually shown on one side/corner of the seed on the inside.
_____________________________________________________________________________________________________________________________________________________________
 Figure 8: Table of the Characteristics of Plants Collected from the Transect. More detailed descriptions are written above.
Figure 8: Table of the Characteristics of Plants Collected from the Transect. More detailed descriptions are written above.
Fungi sporangia is when a fungi grows hyphae (filamentous structure) upwards and forms small, black sporangia (contains spores which are releases when sporangia opens).
PCR amplification did not work because there was too much DNA in the sample that was put in the agarose gel. This resulted in blurry, undefined bar lines instead of clear bar lines that was supposed to appear when amplified in electrophoresis. Since the PCR 16 S Sequence did not work for us, I used Spring 2014 Lab, Brittney Nalty's amplification since we both had the same transect, the 16 S sequence would be the same.
Sequence 1: TGNTCAGTTTGAACGNTGGCGGCATGCCTTACACATGCAAGTCGAACGGCAGCACGGAGCTTGCTCTGGTGGCGAGTGGCGAACGGGTGAGTAATATATCGGAACGTACCCTGGAGTGGGGGATAACGTAGCGAAAGYTACGMTAATACCGCATACGATCTAAGGATGAAAGTGGGGGATCGCAAGACCTCATGCTYSTGGAGCGGCCGATATYTGATTAGCTAGTTGGTAGGGTAAAAGCCTACCMAGGCATCGATCAGTAGCTGGTNTGAGAGGACGACCAGCCACACTGGAACTGAGACACGGTCCAGACTCCTACGGGAGGCAGCAGTGGRGAATTTTGGACAATGGGCGAAAGCCTGATCCAGCAATGCCGCSYGAGTGAAGAAGGCCTTNNGGTYGTAAAGCTCTTTTGTCANGGAAGAAACGGTGNARRGCTAATTTNCTTTGCTAATGACGGWACCWGAAGAATAANCANNNGCTAANN Sequence 2: AGCGGTAGAGATTCTTCNGAWTCTKRAKAGCGGMGTRCRGRTKMRGAACACGKRWGCAASCTGSCTTTRTSGSGGGGATARCCTTTCKAAAGGAAGATTAATMCCCCATAATATATTAARNNNNATCASKKGAYMTTNNNGMMAASTCCGGTGGRWAAWGATGGGCWCGSRCAAGATTAGWKAGWTGGTAAKGTRRCGGCWANCCAAGTYMGTGATCTTTATGGGGCMTGAKAGGGTGATCCCCCACWCTGGTAMYGAGACMCKGACCAGACTCSTACRGKASGCAGCAGTGAGGAATATTGGACWATGGGTGARAGCCTGATCCMGCCATCCCGCGTGAASGATGACGGCCCTATGRGTTGTAATCTTCTTNTGTATWTGGATAAACCTTNNCNACKGT Sequence 3: TCNAAACAGCAAAGTATTAATTTACTGCCCTTCCTCCCAACTTAAAGTGCTTTACAATCCGAAGACCTTCTTCACACACGCGGCATGGCTGGATCAGGCTTTCGCCCATTGTCCAATATTCCCCACTGCTGCCTCCCGTAGGAGTCTGGACCGTGTCTCAGTTCCAGTGTGACTGATCATCCTCTCAGACCAGTTACGGATCGTCGCCTTGGTGAGCCATTACCTCACCAACTAGCTAATCCGACCTAGGCTCATCTGATAGCGCAAGGCCCGAAGGTCCCCTGCTTTCTCCCGTAGGACGTATGCGGTATTAGCGTTCCTTTCGAAACGTTGTCCCCCACTACCAGGCAGATTCCTAGGCATTACTCACCCGTCCGCCGCTGAATCAAGGAGCAAGCTCCTCTCATCC
Bacterias identified were: pseudomona, Sphingobacterium, and the last one was unidentifiable.
pseudomona - Gram +, 244 x 10‐3 # of colonies of bacteria/mL culture, not motile
Sphingobacterium - Gram -, 5 x 10‐5 # of colonies of bacteria/mL culture, not motile
unidentifiable - Gram -, lawn, motile
Purpose: Identifying the vascularization, mechanism of reproduction, and characteristics of the plants using cross section of the leaves and stems and look them under the microscope to identify the mechanism for vascularization.
Conclusion: All 5 plants performed vascularization using stomata (the first one). The other four definitely had spnogy mesophyll and intercellular spaces. Knowing that all 5 plants are angiosperms (not byrophytes like moss that does not have vascularization), they all perform vascularization.
MO
Microbiology
February 9, 2016
In the Hay Infusion Culture, water evaporated, the film of mold that was growing on the surface covered only half of what it originally did, and the smell is not as strong as before.

From the table, the agar-filled petri dishes that included the nutrient and the antibiotics (tetracycline) have larger number of colonies growing on the plates. On the other hand, agar-filled petri dishes with no tet have fewer number of colonies on the plates. This indicates that the bacteria culture adapted to the antibiotic and therefore, grew out in large numbers as the bacteria reproduced in larger amount to counter the antibiotic. This could also mean that tetracycline becomes another food source or a stimulant that increase the reproduction of the bacteria culture. For both bacteria and fungi, they became resistant to the antibiotic, and so almost all the bacteria were unaffected by the tetracycline.
Tetracycline is used to treat human and animal infections, as growth promoters in animal feeds, and in prophlylaxis (Chopra). Tetracycline fight against gram-stain positive and negative bacteria, chlamydiae, mycoplasmas, rickettsiae, and protozoan parasites (Chopra).
Citation:
Chopra, I., & Roberts, M. (2001, June). Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Retrieved February 09, 2016, from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC99026/
Gram stain is performed by placing the bacteria onto the microscopic slide, creating a bacterial smear, and adding a drop of water. Heat the slide with a bunsen burner by waving the slide over the flame to evaporate the water. After it dries, place it slide on the staining tray and cover the bacterial smear with crystal violet for 1 minute. Then rinse the stain off using distilled water. Then, stain the bacterial smear with Gram's iodine mordant for 1 minute, and rinse off with distilled water. Then, discolor the bacterial smear using 95% alcohol for 30 seconds. Rinse off the alcohol using distilled water. After that, cover the smear with safranin stain for 30 seconds, and rinse it off using distilled water. Finally, using a wipe specific for microscopic slides, wipe off any excess water around the bacterial smear. Then the gram-stained slide is looked under the microscope (without cover slips) using 40X and 100X oil immersion objective lenses. The purpose of the gram stain is to see differences in the DNA sequence that encodes for an enzyme, peptidoglycan. Using gram stain is a plymerase chain reaction (PCR) that uses primer sequences specific for such genes that produce peptidoglycan and other non-coding areas. This method also provides information on how Archaea are different from other prokaryotes.
The PCR for 16S amplification was set up using 2 PCR tubes (labeled with our transect number and colony). 20µl of primer/water mixture was added to each tubes. Then, using toothpicks, we picked a small amount of bacteria from 10^(-5) tet and 10(-7) (using one toothpick each) and mixed them into the PCR tubes (one for each tube). Then, after discarding the toothpicks and the tubes are mixed by flicking the tube, the tubes are placed in the PCR machine. The PCR amplification on agarose gel will be observed next week.
Table 2: Bacteria Characterization _____________________________________________________________________________________________________________________________________________________________
10^(-7) tet: The colony is filamentous and has a color of pink. It does not exhibit motility and the colony is single or in clomps together. This bacteria is gram+ and it was measured 15µm at 40X magnification. _____________________________________________________________________________________________________________________________________________________________
10^(-7) no tet: The colony is irregular in shape and the edge is categorized as lobate. The colony is purple meaning that it is gram+. The bacteria is not motile, and they are not in clusters (cells all clomped together in a group) but separated as individuals. The cells measured from 4µm to 20µm at 40X magnification. The cells differs in size because all the cells are irregular in shape and size. _____________________________________________________________________________________________________________________________________________________________
10^(-9) tet: The colony is filamentous and coccus. The cells have no motility. The coccus parts of the cells are in groups, but the filamentous are tied together. This bacteria is gram+. The coccus measures 2µm and the filamentous varies from 20µm to 100µm both at 40X magnification. _____________________________________________________________________________________________________________________________________________________________
10^(-5) no tet: The colony is filamentous and are all in clomps together, as if there is not a lot of space for the cells to be by themselves. The cells are gram+ and measures 10µm at 40X magnification. _____________________________________________________________________________________________________________________________________________________________
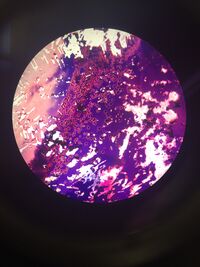
Figure 1: Picture of 10^(-7) no tet. Measures from 4µm to 20µm at 40X magnification
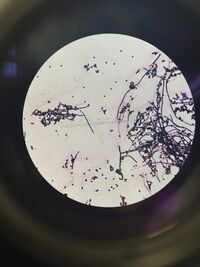
Figure 2: Picture of 10^(-9) tet. The cells include both coccus - 2µm at 40X - and 20µm to 100µm at 40X magnification

Figure 3: Picture of 10^(-5) no tet. Measures 10µm at 40X magnification.
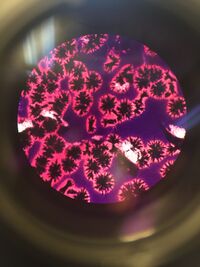
Figure 4: Picture of 10^(-7) tet. Measures from 15µm at 40X magnification.
Purpose: How serial dilution works. How antibiotics affect the bacteria culture in the agar-filled petri dish (is there competition, adaptation). Understand the importance of gram-stain which explains how some organisms are immune to certain antibiotics because of the extra outer layer of peptidoglycan. Identify different characteristics and motility of the bacteria.
Conclusion: All the bacteria that was used to test the gram-stains were all Gram +. That means that all of them have an outer layer of peptidoglycan that makes the antibiotics harder to reach the core of the bacteria. Some of them were similar in shape like the filamentous. But there were irregular and coccus shaped ones. From this, not a lot of biodiversity is seen because all of them were Gram +, so they must have been similar or same organisms.
MO
Protists and Algae
February 2, 2016
The Hay Fusion was set up using about 12 grams of the biotic and abiotic components collected from the transect, .1 grams of milk powder, and 500mL of water; all put into a plastic jar and mixed. Then the Hay Fusion was left for a week, to grow the protists. For the initial observation, there were layers of mold on the surface of the Hay Fusion, shaped like a surface of a leaf. The Hay Fusion also smelt like a porter potty and soil from decomposed things like leaves. The heavy objects like the wood chip, soil and leaves were at the bottom of the jar.
The protists might have come from the biotic components that was picked up from the transect. and possibly from frost because the frost might have trapped the protists as the water froze and turned into frost on the soil's surface . From the top layer of the Hay Fusion, there were two main protists that inhabited that layer. The protists were identified using the dichotomous key. One of them was Arcella. The protist was colorless and it was creeping or floating on the surface (no apparent movement by flagella or cilia) when viewed by the microscope. The protist was not spherical in shape. Although the picture looks as if the protist is spherical, according to the dichotomous key, if the protist is indeed spherical, then it would have radiating spines coming out of the protist. The shape of the protist remained constant meaning that it did not shape-shift. The protist did not have attached materials and had flat shell. So dichotomous key lead to Arcella, and the found Arcella was 50µm in diameter, which is within the range of 45-100µm in diameter of Arcella. From the dichotomous key, it is likely that Arcella is an amoeba since id does not use cilia or flagella to move around.
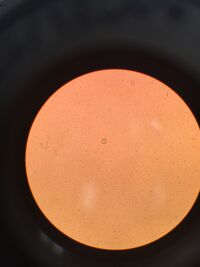
The other protist was of Paramecium Species, and the top layer particularly had Paramecium bursaria. This protist was moving around a lot; it span in circles and up and down. It was also colorless and it exhibited motions using hair-like structure: cilia, which covers the entire body. The protist had an elongated body with rounded ends, swam in corkscrew fashion, and was not attached to a substrate. The dichotomous key lead to Paramecium and by the measurement of 70µm and the shape of the protist, which was small and had a little hook-like or dent-like shape, told that the protist was Paramecium bursaria. Since this leaveing up on the deadline of summer break. Based on the motility and the dichotomous key, it looks like Paramecium bursaria is a protozoan. Also, since there is no color, it does not likely that they photosynthesize.

When the bottom layer of the Hay Fusion was observed under the microscope, the same protists that were identified on the top layer was the only protists that were identified. So in the Hay Fusion, the same protists appeared on both the top layer and the bottom layer of the jar.
The protists found describe how these species meets the needs of life. For example, arcella gets energy by feeding on the food, in this Hay Fusion the food was powdered milk and biotic components in the jar. Arcella is unicellular so it is made up of cell. It is encoded in units called genes to pass on the heriditary location, and so replication (protists are reproduced asexually) and evolution still occurs for Arcella.
If the Hay Fusion culture grow for another two months, then there will surely be a competition for food because food is not being added to the growing population. There will also be some kind of natural selection where one type of gene favors the environment compared to others.
Purpose: How does Hay Fusion work and what does it do? (Grow culture in an aquatic environment with food). Is there growth of certain algae and bacteria? Identifying protists using dichotomous key.
Conclusion: There were only 2 protists found on both the top and bottom layers of the Hay Fusion. This does not show any biodiversity and may have had an error by not picking out healthy, live biotic factors. Also, it's autumn and winter so the plants are dead, dying and since it's a deciduous forest, the leaves fall down, already dead.
MO
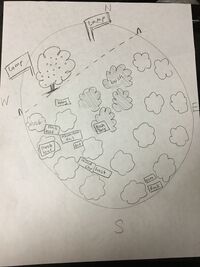
Transect Description
January 18, 2016
The transect was located near the Amphitheater on American University's campus. The transect had a circular shape and there were two lamp posts at each North and Northwest side of the circle. The transect had lots of shrubs, bushes, wood chips and dead leaves. The shrubs had dying leaves but the bushes grew healthy leaves. The ground was covered in dead leaves and wood chips and it was moist on the inside but was hard on the top soil with frost on top. The leaves were decomposed and so the soil created a layer of dead leaves and soil combinations. There was a berry-bearing tree right beside the transect and some berries dropped over to the transect. The transect is a deciduous forest.
Biotic: leaf from the shrub, wood chip, twig from the bush, berry, and black leaf collected from the ground
Abiotic: frost, aluminum foil, dirt, rock, and piece of gum
Purpose: Importance of ecosystem in the transect. Understanding evolution as we keep going with the lab.
Conclusion: The transect contains different organisms including plants and microorganisms. It'll be interesting to see what kind of organisms live in the transect as we go through the labs.
MO