User:Liam L. Bond/Notebook/Biology 210 at AU
I was finally able to find this!
January 26 2015: Lab Data from Lab 1
This is a good start. While you have all the requirements, this information will be more difficult to base your final project off of. Having a more detailed diagram and description will be helpful in the long run. Good job. ML
Our group's transect is located in the gardens by the statue of the eagle outside of Bender Arena at American University. It includes concrete pathways that wind around tall and low bushes and some low trees.
The biotic factors of the transect are as follows: -Tall Bushes -Short Bushes -Trees -Low Plants -Leaves The abiotic factors are as follows: -2 x Lampost -Concrete -Snow/water -Soil -Rocks -Embedded sprinkler system
January 27 2015: Lab Data from Lab 2
After viewing the culture inside of the jar, it smelled rancid and had a little bit of mold growing on the surface of the water. Samples were separately taken from the top and bottom of the jar and a wet mount was prepared. After viewing different organisms under the microscope, the following protists were identified:
Top
-Chlamydomonas
-Colpidium
-Eudorina
Chlamydomonas and Eudorina are types of algae, while Colpidium is a protozoa
Bottom
-Colpidium
-Actinosphaerium
Colpidium and Actinosphaerium are both protozoa

Using samples taken from the hay infusion culture, 8 bacteria plates were prepared. All were given a nutrient agar for growing, while 4 of them were also given tetracycline, an antibiotic.
This is not enough information. In order to be sure of what you are looking at, you should document everything with drawings or photos. Documentation is key! Good observation of your hay infusion though. ML
February 3 2015: Lab Data from Lab 3
After allowing the hay infusion culture to sit for another week, there were noticeable changes. First off, no archaea were found because they grow in extreme environments, and this is not an extreme environment. The noticeable changes were that the water level was lower, there was mold and algae growing on the leaves, and that the leaves had become mushy in the hay infusion culture.
The 8 bacteria plates were then viewed after 1 week. The results are as follows.
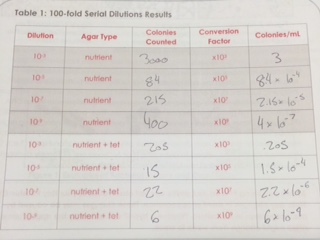 There are obvious differences between the tet and non-tet plates. The plates with tetracycline had far less growth on them. Although none of the species of bacteria were killed off by the tetracycline, it had an obvious impact on each species due to the lower amount of growth that is seen.
There are obvious differences between the tet and non-tet plates. The plates with tetracycline had far less growth on them. Although none of the species of bacteria were killed off by the tetracycline, it had an obvious impact on each species due to the lower amount of growth that is seen.
According to the National Center for Biotechnology Information, tetracycline is a protein synthesis inhibitor that affects the attachment of tRNA to the ribosomal acceptor site. Since this inhibits protein synthesis, the bacteria would not be able to produce protein leading to cell death. Therefore, this would obviously affect all types of bacteria, unless the colonies had evolved to become tet resistant.
Different types of bacteria found in the transect were then observed under the microscope. Samples were taken from the -3 plate, the -5 plate, the -3 tet plate and the -5 tet plate. A gram stain was also performed on each colony.

Make sure you include pictures to help in the future! ML
February 11 2015: Lab Data from Lab 4
The transect was revisited, and 5 plant samples were taken along with leaf litter. The plant samples were observed while the leaf litter was used to create a Berlese Funnel.
The five different plant samples were observed and recorded:




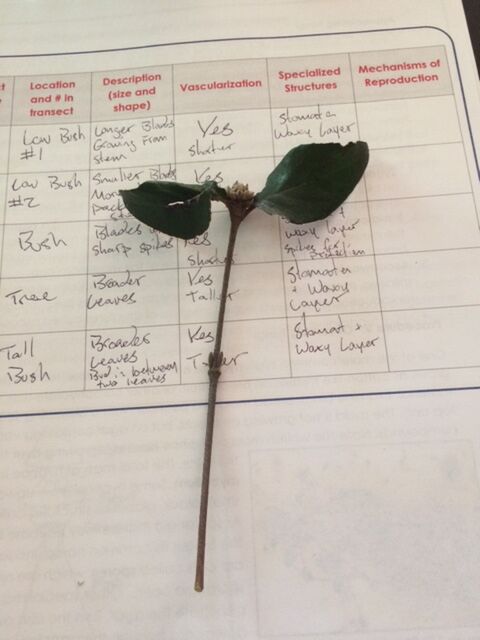
Out of all the samples, three of the samples were monocots and two were dicots. The two dicot samples were the tree sample and the tall bush sample.

Fungi grown in the culture dishes were then observed. Fungi are distinguishable by their sporangia, which are black, globelike structures that grow upwards. The fungi that we observed that were Ascomycotas, which most commonly include yeasts and molds. Due to the presence of mold in our hay infusion culture, we determined that the fungi that was found were ascomycotas.
February 15 2015: Lab Data from Lab 5
This week, we looked at invertebrates that were collected in from our transect in a Berlese funnel. We began by viewing three different types of worms. We viewed the cross sections of one worms, which have a completely lined body cavity. We also looked at nematodes, which was very interesting to watch them swim around. We then looked at common earth worms, which had moved under soil and coiled themselves around each other.
In our analysis of the Berlese funnel, we identified four different organisms.

Make sure you include a description of the Berlese Funnel and the other methods that you used to determine the invertebrates in your transect. ML
February 25 2015: Results from PCR
10^-3 Sequence: NNNNNNNNNNGNNGCNNNNNTGCAGTCGAACGGGGTANGCGTAANGCTGCTNCTAGAGGGGGAGAGGGGTGCGTGAGGGC NNAGCGTCTGACTGACCGCGGGAGAGGGGTTGAAANAGGAACGCTAAAACCATGAACGTCACAGAGGAAAAGGCGGGAGA CGGTGGCCTTTTGCTCTATTTAATGATNATGGGTGGGAGCCCGAGANGGTGGTGGTNNTCNCCGTAANGGNAAGATGTAT ATGGGGTCAGAGAANAAGCTNAGACNCAGAGAAACTGAGACAGCGTCCAGACTCATACGGGGGGNGGGTGTGGAATTTGG GGCAATGGGGGAAAGGGCCATATCCAGCCGTGCCGCGTGTGCGAAGAACGCCTGAGTTTTGCCCTGCTTTTTGTCTTGNG ATGAAGGCCAGGAATAAAATATTCTACNCTTGTGATTCTGACTGTACAAAAAGAATAAGGACCGGCTAACTCCGTGCCCG CAGCCGCGGTAATACGGAGGATGCTAGCGTTATTCAGATTTATTGGGTTTAACGCGTGCGTATGCGGTCTTTTAATTCAA TGGTCAAATACTCTACCTCAACTGTCGCATTGCCTTTGATACTGTAGTACTTGAGTCTGTTTGANGTGNGTGCAATTACA CGTGTAACGGTGATATNCATANATATGTCNNGGANCTCCNATTGCGNNNGCCTCTCCCTANNCTNNCACTGACGCTNATG CACGAANGNNNNNGGATCGAACAGGATTACATACNCTCNNNNNCCACNCCCTANACAATTATAACTTGATGTTTGNNAAG ACAAAANNCTNCCCNCCCNCCCCAANCNTTTNNTTNNTNNNCCTNNGGAGGAGGNNCNCCCNGGTGAANACTCNNNNGNN TTTNNNNNCNGNCNNCACNCNCNCANNANNNGNNNNNNNNNNNNNNTNATACNNNAANNAACCNTGCNNCCCCTCCTTNN NNNNNGGGGGNNGNGGANAANNANNNNNNNNNNGNTNNNNNNNNNNANGANGNAANNNNNNNNNGGNGGGGNNNNNNNNN NNNNNNNTNTTNNTANNNNNANNNGGNGNNNNNANCNNCNNNNNGTNNNNN
10^-3 tet Sequence: NNNNNNNNNCNTANNNTGCAGTCGTANCAANGTAGCCNNNNNNTCCTGGCGGCGAGTGGCGAACGGGTGAGTAATACATC GGAACGTGCCCAATCGTGGGGGATAACGCAGCGAAAGCTGTGCTAATACCGCATACGATCTACGGATGAAAGCAGGGGAT CGCAAGACCTTGCGCGAATGGAGCGGCCGATGGCAGATTAGGTAGTTGGTGAGGTAAAGGCTCACCAAGCCTTCGATCTG TAGCTGGTCTGAGAGGACGACCAGCCACACTGGGACTGAGACACGGCCCAAACTCCTACGGGAGGCAGCAGTGGGGAATT TTGGACAATGGGCGAAAGCCTGATCCAGCCATGCCGCGTGCAGGATGAAGGCCTTCGGGTTGTAAACTGCTTTTGTACGG AACGAAACGGCCTTTTCTAATAAAGAGGGCTAATGACGGTACCGTAAGAATAAGCACCGGCTAACTACGTGCCAGCAGCC GCGGTAATACGTAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGTGCGCAGGCGGTTATGTAAGACAGTTGTG AAATCCCCGGGCTCAACCTGGGAACTGCATCTGTGACTGCATAGCTAGAGTACGGTAGAGGGGGATGGAATTCCGCGTGT AGCAGTGAAATGCGTAGATATGCGGAGGAACACCGATGGCGAAGGCAATCCCCTGGACCTGTACTGACGCTCATGCACGA AAGCGTGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCACGCCCTAAACGATGTCAACTGGTTGTTGGGTCTTCACTG ACTCANTAACNAAGCTAACGCGTGAAGTTGACCGCCTGGGGAGTACGGCCGCAAGGTTGAAACTCAAGGAATTGACGGGG ACCCGCACNAACGGTGGATGATGTGGTTTAATTCNATGCAACNCNAAAAACCTTACCCACCTTTGACATGTACGGAATTC NNCAGANNTNGCTTANGGCTCNAANNANAACCGTTACACNGNTGCTGCATGGNNGTCNTCAGCTCGTGTCNNGAGNNNNT NGGGNTAANTCCCGNAACNAGCNCNNACCCNNTGNNN
The 10^-3 sequence was identified to be an uncultured bacterium. This means that the bacteria was unidentifiable. This is most likely due to the a mistake during the PCR process that would return poor results.
The 10^-3 tet sequence was identified to be the bacteria variovorex paradoxus. This bacteria does not match the description of what we found. Variovorax paradoxus is gram negative, while what we found is gram positive. This again is most likely due to errors during the PCR process.
February 18 2015: Lab 5
This week, the lab focus shifted from transects to zebra fish. In our experiment, we made three separate groups of zebra fish with 20 eggs in each group. The first group was placed in water while the second group was exposed to fluoride and the third group was exposed to a neon chemical.
February 25 2015: Lab 6
This week, the zebra fish had been hatched and alive for one week. The findings are as follows:


