User:Leo G. Brody/Notebook/Biology 210 at AU
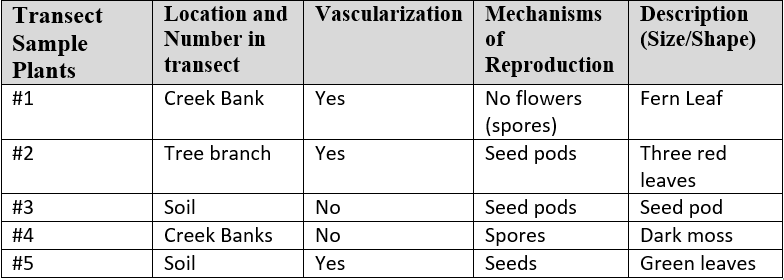
Lab 1-Transect Analysis (1/13/2016): The 20 X 20 transect that our group (group 3) was assigned is located next to the amphitheater. The transect is a small creek, about a meter across and five inches deep, with foliage surrounding it. Abiotic Components: 1.) Rocks- in the creek and soil 2.) Water- in the creek 3.) Clay- near the banks of the creek 4.) Litter- scattered throughout the transect 5.) Concrete- blocks of it in the creek. Biotic Components: 1.) Ivy- located throughout the transect 2.) wild onion- around the edge of the creek 3.) flowers- near the sidewalk 4.) birds- nesting in the trees within the transect 5.) insects- within the dirt and on the surface of the water.
Picture of aerial view of transect:
Picture of transect:
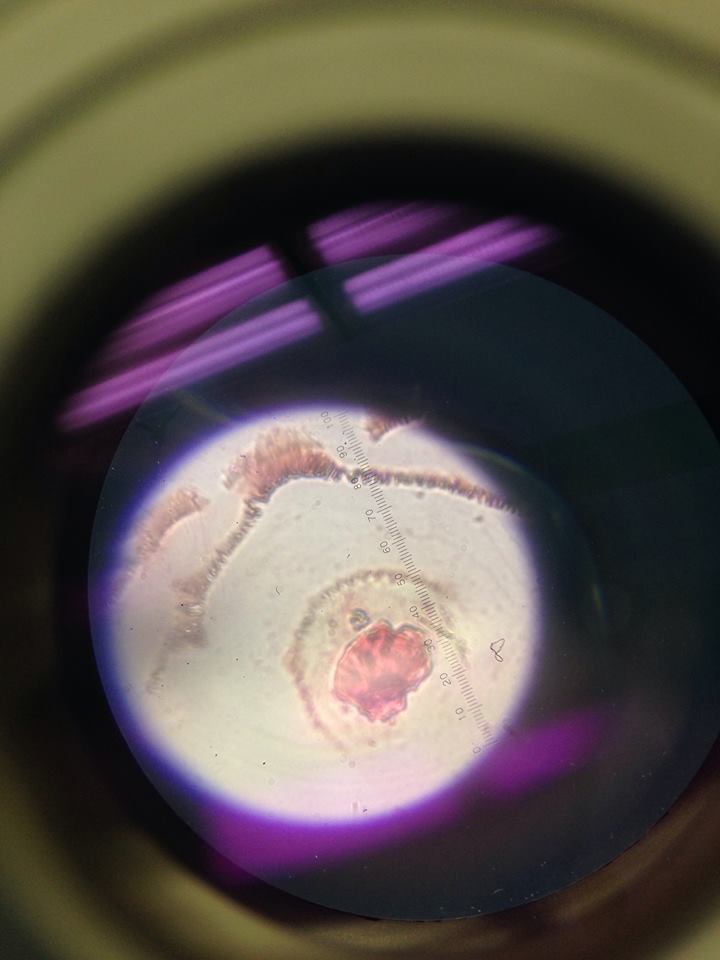
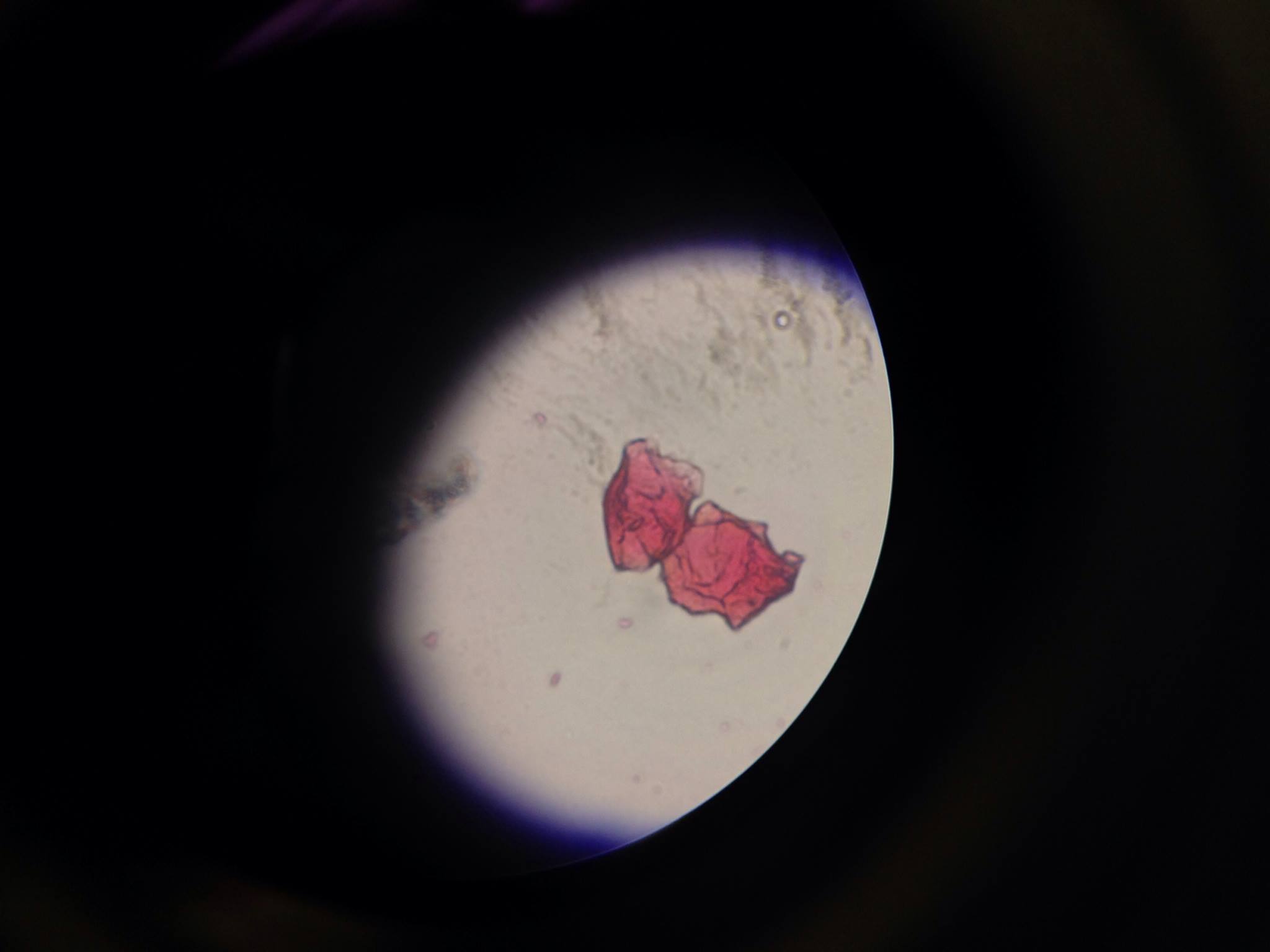
Lab 2 "Protists" (1/27/2016:The hay infusion culture had an acrid smell and the liquid within was black with a filmy surface. The plant matter within the jar were far decayed, and had turned shades of brown and black. The rotting onion root likely contributed to most of the foul smell. Mold was prevalent throughout the sample, especially on the surface. The mold growing on the plant matter is likely different than the mold that fed on the dried milk solution. It was hypothesized that the life at the bottom of the jar would be more diverse and numerous.
After counting (estimating) the number of colonies on the week-old dilution plates, the results were recorded and found to be surprising. The nutrient plates not containing tetracycline had colonies, but they were not numerous and clustered enough to be visible. The diluted nutrient plates containing tetracycline starkly contrasted the others in that their colonies were large, colorful, and numbered in the dozens to even hundreds. The following trend in the number of colonies on the tetracycline (tet) plates was noticed- (10^-3): hundreds, (10^-5): hundreds, (10^-7): ~50, (10^-9): ~40. We expected the number to colonies to decrease with increasing dilution, however we did not expect to see less colonies on the plate that did not contain the antibiotic. The bacteria in the 10^-3 and 10^-5 tet dilutions were likely able to survive the antibiotics because they were able to spread out and develop a resistance. As for the nutrient plates lacking tet, finding a hypothesis for their small numbers proved more difficult. It may be that the lack of antibiotic led to very large colonies of bacteria that began to compete for food, and over time sustained large losses in population due to scarcity and natural selection. Simply put, the tet plate colonies were not compelled by their environment to compete for space and food as much.
Research on tetracycline resistant bacteria has discovered that tet resistance is often the result of bacteria acquiring new genes which code for proteins that protect essential ribosomes from the antibiotic. Bacteria may also develop resistance from random mutation. The way in which tet operates is that it inhibits protein synthesis within bacterial ribosomes.
Materials/Methods: For the gram stain, four slides were prepared with bacteria samples from the diluted dishes. The samples were dried over a flame, then crystal violet was used to cover each sample. The stain was decolorized using 95% alcohol for 10-20 seconds. Then, the samples were covered with safrannin stain for 20-30 seconds. For PCR, two tubes were prepared with 20 microliters of primer/water mixture. A toothpick was used to transfer some of the bacterial colony into the tubes.
Source: Chopra, I., & Roberts, M. (2001). Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiology and Molecular Biology Reviews, 65(2), 232–260
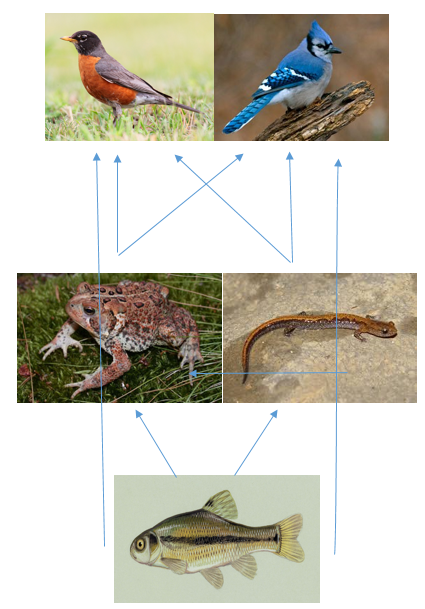
Invertebrates Lab:Our transect sample yielded no invertebrates! So we used another group's sample from the same transect, however there was only one identifiable species:
 Using the dichotomous key, we identified the insect as a mayfly. Other vertebrates that may inhabit the aquatic transect could be 1.) American Robins (Chordata, Aves, Passeriformes, Turdidade, Turdus, T. migratorius. 2.) Bluejays (Chordata, Aves, Passeriformes, Corvidae, Cyanocitta, C. cristata. 3.) American Toad (Chordata, Amphibia, Anura, Bufonidae, Anaxyrus, and A. americanus). ( 4.) Northern zigzag salamander (Chordata, Vertebrata, Amphibia, Caudata, Plethodontinae, Plethdeon, Plethedon borsalis). and 5.) Fathead Minnows (Chordata, Actinopterygii, Cypriniformes, Cyprinidae, Pimpephales, P. prompilaes).
Using the dichotomous key, we identified the insect as a mayfly. Other vertebrates that may inhabit the aquatic transect could be 1.) American Robins (Chordata, Aves, Passeriformes, Turdidade, Turdus, T. migratorius. 2.) Bluejays (Chordata, Aves, Passeriformes, Corvidae, Cyanocitta, C. cristata. 3.) American Toad (Chordata, Amphibia, Anura, Bufonidae, Anaxyrus, and A. americanus). ( 4.) Northern zigzag salamander (Chordata, Vertebrata, Amphibia, Caudata, Plethodontinae, Plethdeon, Plethedon borsalis). and 5.) Fathead Minnows (Chordata, Actinopterygii, Cypriniformes, Cyprinidae, Pimpephales, P. prompilaes).
Embryology Lab:The developmental stages of frog, chicken, and starfish embryos were observed under microscopes in the lab. The sizes of the embryos varied greatly, and the blastuals and morulas were highly visible. Preparations for the zebra-fish experiment began. Our zebra-fish embryos will be brought up in two environments: one of variable light (control group) and one in constant darkness. We hypothesized that those exposed to variable light will have a lower visual acuity than those kept in the dark. Based on readings we had done before lab, we knew that exposure to too much light can hinder physiological development of the fish's retina. In all other ways, the two groups of zebrafish are in identical conditions: two trays filled with water and twenty eggs per tray. Each tray is fed food at the same intervals. The only control is lighting. I am excited to see how the fish develop over time!
DNA Sequence Analysis: Nucleotide Sequence: GCNTACNCATGCAAGTCGAGCGGAGATGAGGTGCTTGCACCTTATCTTAGCGGCGGACGGGTGAGTA ATGCTTAGGAATCTGCCTATTAGTGGGGGACAACATTTCGAAAGGAATGCTAATACCGCATACGCCCTACGGGGGAAAGC AGGGGATCTTCGGACCTTGCGCTAATAGATGAGCCTAAGTCGGATTAGCTAGTTGGTGGGGTAAAGGCCTACCAAGGCGA CGATCTGTAGCGGGTCTGAGAGGATGATCCGCCACACTGGGACTGAGACACGGCCCAGACTCCTACGGGAGGCAGCAGTG GGGAATATTGGACAATGGGGGGAACCCTGATCCAGCCATGCCGCGTGTGTGAAGAAGGCCTTTTGGTTGTAAAGCACTTT AAGCGAGGAGGAGGAGCTCTAGATTAATACTCTAGATGCTTGGACGTTACTCGCAGAATAAGCACCGGCTAACTCTGTGC CAGCAGCCGCGGTAATACAGAGGGTGCAAGCGTTAATCGGATTTACTGGGCGTAAAGCGTGCGTAGGTGGTCTTTTAAGT CGGATGTGAAATCCCTGAGCTTAACTTAGGAATTGCATTCGATACTGGGAGACTAGAGTATGGGAGAGGATGGTAGAATT CCAGGTGTAGCGGTGAAATGCGTAGAGATCTGGAGGAATACCGATGGCGAAGGCNGCCATCTGGCCNNNTACTGACACTG AGGTACGAAAGCATGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCATGCCGTANACGATGTCTACTAGCCGTTGGGG CCTTTGAGGCTTTAGTGGCGCANCTAACGCGATAAGTAGACCGCCTGGGGAGTACGGTCGCAAGACTAAAACTCAAATGA ATTGACNGGGGCCCGCACAANCGGTGGANCATGTGGTTTAATTCNATGCAACGCNAANACCTTACCTGCTCTTGACGTA
Species identified: Acinetobacter, a genus of Gram-negative bacteria that is ubiquitous and is important for soil health.