User:Keanan K. McDaniel/Notebook/Biology 210 at AU
Zebrafish final week 3/4/16 K. McDaniel DNA sequence: BAM3-16s_Forward_H11.ab1 NNNNNNNNNNNNNCNNNNTTACNNTGCAGTCGTAACAGNGNGTNANCCNNNNNTGCTCTGGTGGCGAGTGGCGAACGGGT GAGTAATATATCGGAACGTACCCTGGAGTGGGGGATAACGTAGCGAAAGTTACGCTAATACCGCATACGATCTACGGATG AAAGTGGGGGATCGCAAGACCTCATGCTCGTGGAGCGGCCGATATCTGATTAGCTAGTTGGTAGGGTAAAAGCCTACCAA GGCATCGATCAGTAGCTGGTCTGAGAGGACGACCAGCCACACTGGAACTGAGACACGGTCCAGACTCCTACGGGAGGCAG CAGTGGGGAATTTTGGACAATGGGCGAAAGCCTGATCCAGCAATGCCGCGTGAGTGAAGAAGGCCTTCGGGTTGTAAAGC TCTTTTGTCAGGGAAGAAACGGTGAGAGCTAATATCTCTTGCTAATGACGGTACCTGAAGAATAAGCACCGGCTAACTAC GTGCCAGCAGCCGCGGTAATACGTAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGTGCGCAGGCGGTTTTGT AAGTCTGATGTGAAATCCCCGGGCTCAACCTGGGAATTGCATTGGAGACTGCAAGGCTAGAATCTGGCAGAGGGGGGTAG AATTCCACGTGTAGCAGTGAAATGCGTAGATATGTGGAGGAACACCGATGGCGAANGCAGCCCCCTGGGTCAAGATTGAC GCTCATGCACGAAAGCGTGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCACGCCCTAAACGATGTCTACTAGTTGTC GGGTCTTAATTGACTTGGTAACGCAGCTAACGCGTGAAGTAGACCGCCTGGGGAGTACNGTCGCAAGATTAAAACTCAAA GGAATTGACGGGGACCCGCACAAGCGGTGGATGATGTGGATTAATTCGATGCAACGCGAAAAACCTTACCTACCCTTGAC ATGGCTNGNANTCCCNCGAGAGATTGGGGAGTGCTCGAAAGANAACCAGTACACNNNGCTN This is the sequnce for MB11: 5MWP-16s_Forward_G11.ab1 NNNNNNNNNNNNNNNNTNGCNNNGCAGTCGNNNNNNNNNGNNAGGNNNNNNNCTTGCTGCTTCGCTGACGAGTGGCGGAC GGGTGAGTAATGTCTGGGAAACTGCCTGATGGAGGGGGATAACTACTGGAAACGGTAGCTAATACCGCATAACGTCGCAA GACCAAAGAGGGGGACCTTCGGGCCTCTTGCCATCAGATGTGCCCAGATGGGATTAGCTAGTAGGTGGGGTAACGGCTCA CCTAGGCGACGATCCCTAGCTGGTCTGAGAGGATGACCAGCCACACTGGAACTGAGACACGGTCCAGACTCCTACGGGAG GCAGCAGTGGGGAATATTGCACAATGGGCGCAAGCCTGATGCAGCCATGCCGCGTGTATGAAGAAGGCCTTCGGGTTGTA AAGTACTTTCAGCGGGGAGGAAGGCGATGTGGTTAATAACCACGTCGATTGACGTTACCCGCAGAAGAAGCACCGGCTAA CTCCGTGCCAGCAGCCGCGGTAATACGGAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGCACGCAGGCGGTC TGTCAAGTCGGATGTGAAATCCCCGGGCTCAACCTGGGAACTGCATCCGAAACTGGCAGGCTTGAGTCTCGTAGAGGGGG GTAGAATTCCAGGTGTAGCGGTGAAATGCGTAGAGATCTGGAGGAATACCGGTGGCGAANGCGGCCCCCTGGACGAAGAC TGACGCTCANGTGCGAAAGCGTGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCACGCCGTAAACGATGTCGACTTGG AGGTTGTGCCCTTGAGGCGTGGCTTCCGGAGCTAACGCGTTAAGTCGACCGCCTGGGGAGTACGGCCGCAANGTTAAAAC TCNNTGAATTGACGGGGGCCCGCACAAGCGGTGGAGCATGTGGTTTAATTCNATGCAACGCGAAGAACCTTACCTGGTCT TGACATCCACAGAACTTNNNCAGANATNGNNNNGGTGCCNTTNNGGAACTGTGANACAGGTGCTGCATGGNTNNCGTCAG CTCGTGNTNNTGAAANNNNN
Observation: the Zebrafish were feed every day until they died. they grew up to 30mm before they died. the zebrafish that were in the APAP solution, were hard to find because the solution turned from clear to a dark brown  . it is unknown why the solution turned this color because the solution in the wells were changed daily.
. it is unknown why the solution turned this color because the solution in the wells were changed daily.

- Keanan K. McDaniel 00:44, 14 March 2016 (EDT):
Zebrafish Week1 2/26/16 K.McDaniel Purpose:
To perform an controlled experiment with dependent and independent variables. Using zebrafish and observing the delevopmental stages of growth and see how it is effected by Acetaminophen. Will be comparing physical changes in the organs and overall size of the fish. The Zebrafish will be affected by APAP because the drug causes liver toxicity and will have a major effect on the size of their organisms and how they function especially the liver.
Method:
Once the mg of APAP were calculated for a litter of water. One 325mg tablet was crushed using a mortar and pestle and added to the beaker of 1 litter of Deerpark water. Once the solution was shaken and labeled 2x, 235ml of the solution were added to a separate beaker along with 265ml of water to get it at the correct concentration for the Zebrafish. Once, both solutions were labeled, 2ml of the solution were put into 20 wells and 2ml of water was put in 20 different separate wells and labeled .Then the Zebrafish eggs were examined under the dissection scope to see if they were healthy or not. One egg was placed in each well and numbered. The wells were covered with its plate cover to protect the fish as they grow. The wells were placed safely on a table until the next time the fish needed to be checked.
Observation:
All of the Zebrafish were either 20-somites or 25-somites that were placed in the wells. By the end of the week all of the Zebrafish were hatched and eating own their own.


Conclusion: This experiment would have been more accurate if the Zebrafish were all at the same stage of development and at the first stage. The data will not be as accurate as it could be because all of the fish are growing at different rate so the drug will affect them differently.
- Keanan K. McDaniel 11:10, 26 February 2016 (EST):
Identifying invertebrates from a transect 2/19/16 Keanan K. Mcdaniel
Purpose:
The purpose of this lab was to identify invertebrates from a transect using a dichotomous key. Also to differentiate the structure and mechanics of the invertebrates and their symmetry. Also how to Analyze products of a Berlese funnel and see how the funnel works.
Method: Take apart the Berlese Funnel carefully Pour 10ml-15ml of 50% ethanol into a petri dish Carefully pour the organism from the Berlese funnel into the petri dish Use writing utensil to move debris around to better see organisms Examine the dish under the dissection scope. Record the observations and use dichotomous to identify the organisms.
Observation:
Found 8 organism and identified all. There was one mite, five ants, one earth worm and a earwig. The earwig was clear and this was interesting because normally earwigs are a dark brown. This earwig however, just shed its skin and when this happens the new shell looks clears and as it hardens the darker it gets.




Conclusion: This lab was fairly easy and very interesting to do. If this experiment could be performed again but with a larger transect and in a different climate to compare the organism from both. This experiment shows how much life is in just a couple hundred grams of soil, and makes the mind think about how many living organisms there are that live in the soil on this planet.
- Keanan K. McDaniel 10:41, 19 February 2016 (EST):
Keanan K McDaniel 2/11/16 assignment 3
Purpose: To identify and distinguish characteristics of different kinds of plants. Also to find specific characterizations to differentiate between angiosperms and bryophytes. Lastly to identify to different species of fungi too. Methods:
Collection of plant samples from transect.
Grab two Ziplock bags and label them with the group names Go outside to transect. Find area with soft soil and dead leaves and place about 500 grams of it into the Ziplock. Took samples from five different plants and placed them into the other bag Pictures of trees were taken and one branch was taken to help identify the genus. Collected berries off a brush
Plant Vascularization:
Leaf was taken and place under dissection scope This was done for all leaf sample that were collected Wild onion was cut in half and placed under Dissection scope Berry was cut in half and placed under Dissection scope Tree branch was cut in half and placed under Dissection scope The onion’s leaf was placed under the Dissection scope Recorded data that was found while observing all the samples from the transect
Berlese funnel:
Pour 250ml ethonal/water solution into 50ml conical tube Fit a piece of screening material into the bottom of the funnel Tape the sides of screen to the funnel Carefully put leaf litter sample in the top of the funnel Set up funnel ring to hold the funnel and the tube Put parafilm at the base of the funnel and where the funnel and tube meet to prevent the ethanol from evaporating Cover the top of the funnel with foil Let funnel set for a week.
Every sample of leaf looked different under the dissection scope. The structure of the leaves’ veins were unique to each plant that was collected. The tree branch had very small fungus growing on it. it was hard to identify what species of fungi it was because of the size. The xylem was found in the middle of the branch was a different color from the wood surrounding it. the berry was hard, soft most eatable fruit that humans eat. There were no evidence of seeds in the berry.
Conclusion: There were not enough different types of plants to collect for the sample due to the weather. This experiment would be easier to do in the spring when many flowers, plants, and fruit are blooming.
- Keanan K. McDaniel 11:25, 12 February 2016 (EST):
 2/5/16 Notebook entry 3: Microbiology and identifying Bacteria
2/5/16 Notebook entry 3: Microbiology and identifying Bacteria

Purpose:the Characterization of different species of bacteria by observing the motility, gram stain, colony morphology and sequencing of the 16s ribosomal subunit gene.
Materials and methods:
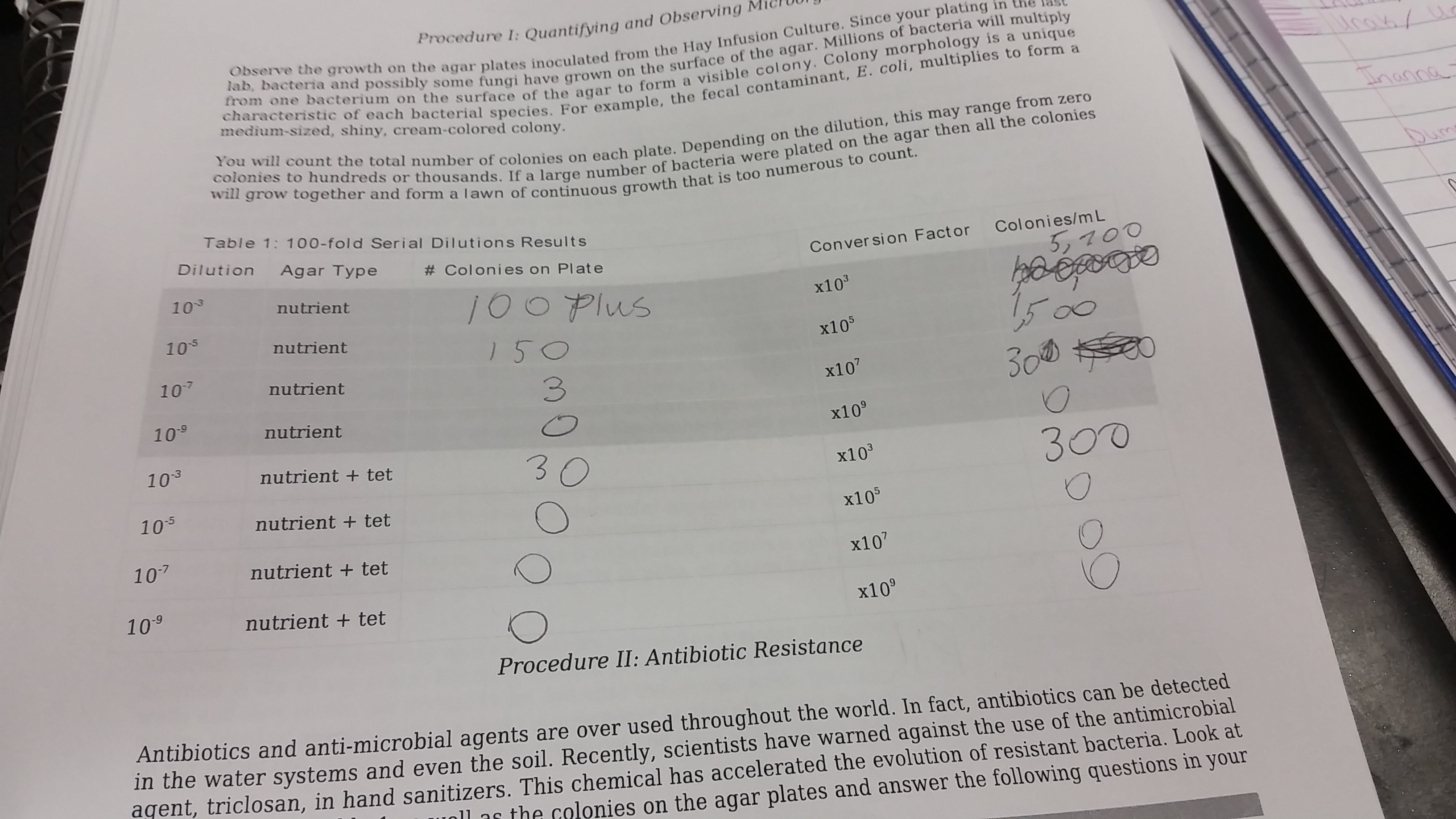
.First, plates were arranged in order of serial dilutions from least diluted to most. . all bacteria colonies in each plate were counted and recorded. . Once accounted for the number of colonies were converted to colonies over mL. .Then Bacteria cultures were observed with microscopes. . Wet mount and Stain Gram were observed with the microscope. Wet: sterilize loop over flame
scrape tiny amount of growth from surface of agar.
Mix it into a drop of water.
place cover slip over drop.
observe with 4x and 10x
observed cell shape and size.
Gram Stain: sterilize loop over flame
scrape tiny amount of growth from surface of agar.
Mix it into a drop of water
circle the area underneath the sample with red wax pencil
heat fix the air dried slide by passing it through a flame 3xtimes with bacteria side up
with staining tray cover slide with crystal violet for 1minute.
risne slide off with filtered water
cover slide with gram iodine mordant for a minute
decolorize by flooding the smear with 95% alcohol for 10-20 seconds.
cover the smear with safranin stain for 20-30 seconds.
rinse off with water
dry excess water carefully with kimswipe, then allow air dry
No cover slip needed
observe sample at low magnification with 40x and 100x.
PCR: label 2 PCR tubes with own identification
add 20ul of primer/water mixture into each tube, mix to dissolve PCR bead
using sterile toothpick pick small amount of a bacterial colony on one of the plates
place toothpick into PCR tube and twist for 5 seconds
repeat last two steps for the other tube.
Data and observation: There were only a two bacteria that were sited under the microscope.Before starting the procedures an observation was made of the Hay Infusion cultures plates.the cultures without the antibiotic grew noticeably. the blood agar plat had growth with clear indications that halos were formed, meaning that the bacteria ate all the blood in certain areas leaving wholes or halos in the plate where parts of the blood agar are missing. The smell also changed and was a potent odor similar to spoiled food. Conclusions and Future directions: if the experiment was redone, there would have been more bacteria to look at on the slides instead of just two organisms. KKM.*Keanan K. McDaniel 23:56, 4 February 2016 (EST):
'Keanan K. McDaniel 01:25, 22 January 2016 (EST):Keanan K.McDaniel == Examining Biological Life at AU 1/20/2016'== Purpose/Description: This experiment was about examining biological life on American university's campus. Also examined, Chlamydomonas, Gonium, and Volvox under a microscope. Materials and Methods: Transect: Draw a picture of the Aerial diagram of the transect on campus. Collect samples from the assigned area with an zip-lock bag. observe area and take note of all the biotic and abiotic components in the transect note details about the transect. Hay infusion: Put 10 to 12 grams of the sample from transect into a plastic jar. Pour 500mLs of deep park water into the jar. Put 0.1 grams of dried milk in the jar then close the jar and shake it well for 10 seconds. label the jar and place in a safe place. Volvocine Line: Pipette a one sample of the Volvox, Chlamydomonas, and Gonium. Place each sample on its own slide and examine the the slides separately Record data to fill out Volvocine line chart and sketch what each organism looks like Data: the Transect that was assigned was a small creek on the north side of campus, behind McDowell Hall. The creek is rocks(abiotic) covered with Moss(Biotic) and fallen tree branches(abiotic). There are about 23 trees(Biotic) and small plants(Biotic) that were in the transect. there were also some dead leaves(abiotic) mixed into the soil(abiotic), also there were some plastic bottles(abiotic). Did not see any animals or organisms in the transect at the time. Conclusion: There would have been more biotic components in the transect, if the weather was warmer but because of the cold weather many of the organisms are underground or somewhere else warmer. Keanan K. McDaniel






