User:Juliana Delgado/Notebook/Biology 210 at AU
Observing a Transect at AU January 13th, 2015
For this laboratory procedure, a group of lab students were given a transect at American University to observe a 20 foot dimension and study characteristics of the protists, bacteria, plants, invertebrate, and animals in a given transect. Students went to their transect with a plastic bag to collect samples within their transect. With gloves, students were instructed to carefully collect dirt, weeds, plants, flowers, or any water if present to prepare a Hay Infusion Culture. Water, nets, rocks, statues, and pollution were some of the abiotic factors that were found at the given transect, Transect #4. Biotic factors that were observed included plants, flowers, microorganisms in the soil, squirrels microorganisms in the water. Pictures were also taken to have evidence of what the transect that can be seen below.
'Preparing and Plating a Serial Dilution
January 20th, 2015
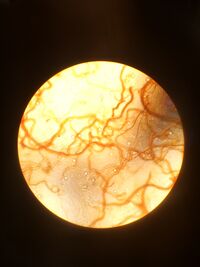
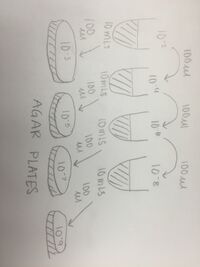
For this lab, students used used their hay infusion culture to make serial dilutions for further the investigation of organisms in the small micro biome that was collected from a given transect. Students were instructed to leave their hay infusion culture (without the lid on) at room temperature in the lab for 7 days. After seven days, the culture began to smell like rotten vegetables and dirt. There was a layer of orange- colored algae that formed at the top of the infusion, while at the bottom of the jar there was dirt, mud, and small pebbles, this can be seen in Figure 2.1. Two samples were taken from the top layer of the jar and from the bottom of the jar. At the bottom of the jar, dirt was found and no microorganisms were recorded. However, after placing the orange algae from the top of the sample under the light microscope, students were able to identify the species using a Dichotomous Key. In Figure 2.1, you can see the species recognized was recorded as _______. The microorganisms observed in the culture seemed to be non-motile, photosynthetic organisms. If the culture was allowed to grow for another 2 months without interruption, the sample would produce a foul odor and continue to grow algae which would be able to support more organisms in the sample. Students then were instructed to make a serial dilution from the sample. A drawing of the dilution that was completed can be seen in Figure 2.3.
"Microbiology and Identifying Bacteria January 27th"
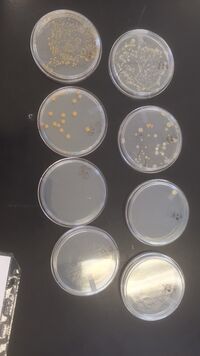
In this lab, students were instructed to characterize and identify different species of bacteria based on motility, gram stain, colony morphology and sequencing of the 16s ribosomal subunit gene. First, one last observation of the Hay Infusion Sample was taken to note any changes in smell or appearance. The appearance of the Culture seemed to have no real change, see figure 3.1. The smell of the culture became more pungent, most likely do to more bacteria growing and reproducing. The agar plates that were made in the previous lab were then taken and analyzed to count the colonies and then calculate the colonies/ mL. A picture of the Agar plates can be seen in Figure 3.2. The data recorded can be seen below in figure 3.3. Although there was not a huge difference in the number of colonies on the plate, the agar type with the nutrient and the tetracycline produced slightly more colonies per mL than the agar type with just the nutrient and no tetracycline. Next, wet mounts and gram stains were made to differentiated different microorganisms in the Hay infusion Culture. First, a wet mount was made by sterilizing a loop over a flame, scarping a sample of the growth from the agar plates, mixing it with a drop of water and placing it on a slide topped with a cover slip. Students were instructed to try to determine the cell shapes and organisms, which is difficult without using a gram stain. So; next, student made a gram stain by sterilizing a loop over a flame, scarping a sample of the growth from the agar plates, mixing it with a drop of water and placing it onto a slide. Then, a circle was drawn underneath the sample with a red wax pencil. The dry side was then passed through a flame three times, and then rinsed with Crystal Violet for 30 seconds, and then rinsing again with water. Next, the bacteria needed to be decolorized by flooding the smear with 95% alcohol for 10-20 seconds. The sample was then again rinsed with water. The excess water and dye was then blotted off the slide and was placed at low magnification to observe the samples (no coverslips are needed for gram staining. This was done to all 4 samples of bacteria. In the samples from Transect 4, Cocci bacteria and Palisades were found along with many rod shaped bacterium. It was also found that both plates that were 10x10^-3 both with the tetracycline and without, were both found to be gram positive, meaning they have a thick layer of peptidoglycan in their cell walls, whereas both agar plates that were 10x10^-5 were gram negative. The colony descriptions and cell descriptions can be seen in figure 3.4. below. Next students prepared samples for a PCR 16S Amplification. Students selected the most complete bacteria that they were able to characterize from the gram staining and run a gel. 2 PCR tubes were labeled and 20 micro liters of primer/water mixture to dissolve the PCR bead at the bottom of the PCR tube. A toothpick was used to pick up a small sample of the bacteria and submerged in the primer/water mixture, and then the PCR tube was placed into the PCR machine.
Results of the PCR, January 27th-March 1st Due to errors of the PCR test this year, the data below was taken from Spring of 2016, at Transect number 4. The data collected can be seen in the table below:
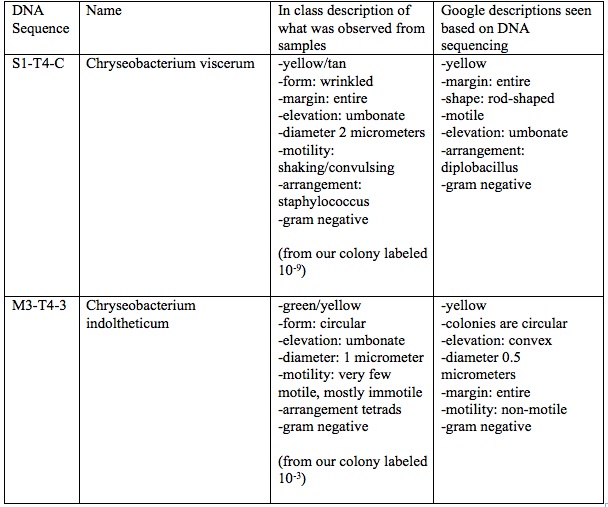
It seems to be that most of the bacteria from Transect #4 is from the chryseobacterium genus. Pont noted, "Their sample labeled S1-T4-C was also umbonate, yellow, motile, had entire margins, and was gram negative; however, this bacterium was diplobacillus, being rod shaped, rather than circular shaped. The other sample observed from our transect was also yellow, umbonate, and gram negative like our previous sample, but this bacterium was also a little green, circular, mostly immotile, and was arranged in tetrads.". By this description, it could be hypothesized that the bacteria that was identified 2 years ago is the same type that was observed this year, 2016, based on the sample that was taken which is imaged in Figure 2.2. The bacterium identified by Pont was Chryseobacterium indoltheticum, which is yellow, circular colonies, non-motile, and gram negative. Based on this evidence, one can assume the sample taken from 2016 was also Chryseobacterium indoltheticum. However, this data is still 2 years old and is not ideal to use presently. The experiment should be replicated again and there PCR sample taken again to ensure these are the same bacteria.
- Data taken from Cassidy Pont, Biology 210, Spring 2014*
"Identifying and Studying Plants February 3rd"
For this laboratory investigation, students were instructed to return back to their transects to collect 500g of leaf litter into one bag to set up a Berlese funnel for the study of invertebrates. Next, 5 different plants were collected and were viewed under a light microscope to identify the species and study their structures. Transect sample plant #1 was bright green, with points on either side of the leaf. Transect sample plant #2 was a much smaller leaf than transect plant leaf #1, coming to only about 0.05 inches with a very thick leaf. transect plant #3 was a long thin leaf, hypothesized to be a blade of grass, that was extremely long coming out to be around 6 inches. Transect plant #4 was red in color and also had points on either side, similar to transect plant #1. Finally, transect plant #5 was a very dark green and had an ovular shape.The first two types of leaves were both taken from separate bushes far away from the pond, closer to Clark and Roper dormitory halls. The blades of grass that were collected were found closer to the pond and were carefully extracted from the ground. The last two leaves were taken from two separate bushes that were located on the other side of the pond, prior to the previous two bushes. There were limited resources to fins seeds or flowers at Transect #4. The vascularization of the plants found the reproductive strategies are still being determined. There was difficulty viewing the vascularization patterns under the microscope, therefore a Dichotomous Key must be used to identify the species and genus. The new data will be uploaded when those species are identified. The data and observations of the 5 diverse plant species can be found in the table below, Figure 4.1.
Invertebrates February 10th
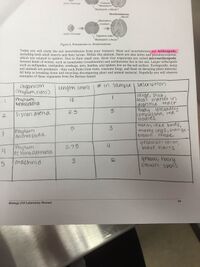
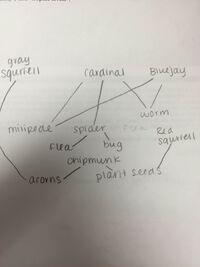
In this lab, students were to use a dichotomous key to identify invertebrates found in a given transect from the Berlese funnel that was prepared last lab. Transect #4 was one of the most populous and diverse samples of microorganisms that was found on the American University campus. Students were instructed to identify at least five microorganisms from the collections using the dichotomous key. Pictures of the samples can be seen in figures 5.1. and 5.2. Transect 5 yielded over 8 different species, here are some of the species that were looked at closer. A large worm was found, believed to be Phylum Nematoda that was pinkish in color and students observed fecal matter in the digestive tract. 3 fleas were also recorded that are believed to be Siphonatetra, their bodies were laterally compressed with legs for jumping and red bodies. 3 millipedes were also observed believed to be Phylum Arthropoda. Next, 4 Phylum Echinodermata were found, they were yellow in color with black hairs, with 6 legs, 2 antennas and 2 tails. Lastly, a spider was discovered that was yellow with brown spots and thick hairs. The table of characterizations can be seen in figure 5.3. Based on these finding of the invertebrates one can assume what vertebrates are present in this community based on climate, biome, and food sources. Examples of some vertebrates may include, the European Red Squirrel, the Eastern Chipmunk, the Gray Squirrel, Cardinals, and the Eastern Bluebird. A hypothetical food chain of all of the vertebrates and invertebrates can be seen below in figure 5.4. Carrying capacity is essential the number of organisms that a region can support without environmental degradation. Therefore, to remain at carrying capacity, the community must be essentially balanced at all times. Balanced, meaning there are ample autotrophs and heterotrophs to support the entire community.
Identifying Starfish, Chick, and Frog Embryos &Zebrafish Set-up February 17th The first part of this lab was to help students identify the embryos of frogs, chicks, and starfish to prepare for the zebra fish experiment. Students looked at prepared slides of the stages of embryology for all three organisms. Students then made a table of the comparison stages, that can be seen below in Figure 6.1.
For this lab, students were instructed to pick a variable, and test its affects on the embryology of zebrafish. Embryology describes the sequence of events after egg fertilization that involve growth, differentiation, and morphogenesis of an animal, in this case, zebrafish. Zebrafish are extremely sensitive to their environments, so picking a variable and the amount of the variable to test was an extremely important task. For the purpose of this lab, twenty zebrafish were tested on the basis of different salinity levels. It has been proven that at a salinity level too high, zebrafish will wither die or be unable to undergo gastrulation during embryology. To study the effects of salt on growing zebrafish, a random sample of 40 zebrafish was taken and placed into two separate petri dishes with individual compartments (1 zebrafish per compartment). Twenty of the zebrafish were labeled control, and were placed in 2 mLs of Deerpark water. The other twenty zebrafish were placed in 2 mLs of Deerpark water with 0.5 M NaCl. The embryos were checked on after 3 days, and then every day after since all of the embryos had been hatched and needed to be fed.
Zebrafish Data 02/19/16 Control:
| ' | 1 | 2 | 3 | 4 | 5 | ' |
| A | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 2 hatched | 1 Live, Embryo Egg | |
| B | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 1 Hatched | 1 hatched | |
| C | 1 Live, Embryo Egg, 1 hatched | Embryo Dead | 1 Live, Embryo Egg | 1 hatched | 3 Live, Embryo Eggs | |
| D | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 2 hatched, 1 Live, Embryo Egg | 1 hatched | |
Treatment:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | 1 hatched | 1 Live, Embryo Egg | 1 Live, Embryo Egg | 2 hatched | Dead Embryo |
| B | Dead Embryo | 1 Live, Embryo Egg, 1 hatched | 1 hatched | 1 hatched | 1 Live, Embryo Egg |
| C | 1 Live, Embryo Egg | 1 hatched | 1 Live, Embryo Egg | 1 Live, Embryo Egg | Dead embryo |
| D | 1 large hatched | 1 Live, Embryo Egg | Dead Embryo | Dead Embryo | Dead Embryo |
Final Count: Control: 7 hatched, 16 still in egg, 1 dead Treatment : 8 hatched, 8 still in egg, 6 dead.
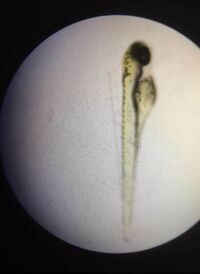
Figure 7.1. Picture of a newly hatched Zebrafish
Zebrafish Data 2/22/16 Control:
| CONTROL: | ' | ' | ' | ' | ' |
| 1 | 2 | 3 | 4 | 5 | |
| A | Alive | Dead | Alive | 2 Alive | Nothing |
| B | Alive | Alive | Alive | Alive | Alive |
| C | 2 Alive | Nothing | Alive | Alive | 3 Alive |
Treatment:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | Alive | Alive | Alive | 2 Alive | Dead |
| B | Dead | 2 Alive | Alive | Alive (deformed?) | Alive |
| C | Unhatched egg | Alive | Alive | Alive | Dead |
| D | Alive | Alive | Dead | Dead | Dead |
From the control, A3 and B2 were removed and looked at under the compound microscope. From the treatment, D1 and A3 were removed and studied under the compound microscope. Control: A3 : Still in egg, wrapped around itself, greenish yellow in color, potentially about to hatch, covering eyes with tail, frequently moving in shell.
B2 : Stage 1 of embryogenesis, in the long pec stage. Still on its side no eye movement.
Treatment: D1: Stage 1 of embryogenesis, in the long pec stage. Over 100 um, no eye movement, strong movement, flipped over on belly while under the microscope, fins flapping strong heartbeat.
A3: Still in egg, coiled in egg, yellowish color, eyes forming, two large black spots on either end of the body
Figure 7.2. Figure 7.1. Picture of a Zebrafish still in egg case. Zebrafish Data 2/23/16
Control:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | 1 Alive | 1 Alive | 1 Alive | 2 Alive | N/A |
| B | 1 Alive, 1 egg | 1 Alive | 1 Alive | 1 Alive | 1 Alive |
| C | 2 Alive | N/A | 1 Alive | 1 Alive | 3 Alive |
| D | 1 Alive | 1 Alive | 1 Alive | 3 Alive (Light, medium, dark) | 1 Alive |
Treatment:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | Alive | 1 Alive | 1 Alive | 1 Alive | 2 Alive |
| B | Dead | Dead | 2 alive (1 bigun) | Dead | 1 Alive |
| C | 1 Alive | 1 Alive | 1 Alive | 1 Alive | Dead |
| D | 1 Alive | 1 Alive | Dead | Dead | Dead |
Zebrafish Data 02-24-16
Control:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | Alive | Alive (a little slow?) | Alive | 2 Alive | N/A |
| B | 1 Alive 1 egg | Alive | Alive | Alive | Alive |
| C | 2 Alive | N/A | Alive | Alive | 3 Alive |
| D | Alive | Alive | Alive | 3 Alive (a lot of food left over) | Alive |
Treatment:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | 1 Alive | 1 Alive | 1 Alive | 2 Alive | N/A |
| B | Dead | 2 Alive | 1 Alive | Dead, extremely deformed, no heartbeat, no movement | 1 Alive |
| C | 1 Alive | 1 Alive | 1 Alive (slow movement) | 1 Alive | Dead |
| D | 1 Alive | 1, Very large alive | N/A | N/A | Embryo that never hatched |
From the Treatment group, D5, and B4 were removed and put in formaldehyde.
Zebrafish data 02/26/16 Control:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | 1 Alive, mold growing mass in center | ALive | 1 Alive | 2 Alive, fungi present | N/A |
| B | 1 Alive, 1 dead, may have never hatched | 1 Alive | 1 Alive | 1 Alive | 1 Alive |
| C | 2 Alive fungi present | N/A | Dead | 1 Alive | 3 Alive |
| D | 1 Alive | 1 Alive | 1 Alive | 2 Alive | 1 Alive |
Treatment:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | 1 Alive | 1 Alive | 1 Alive- very large eyes | 1 Alive, 1 (very large) dead | N/A |
| B | N/A | 2 Alive | 1 alive | N/A | 1 Alive |
| C | 1 alive | 1 Alive | 1 Alive | 1 Dead | N/A |
| D | 1 large alive | 1 Alive | N/A | N/A | N/A |
For the control group, the unhatched embryo was removed from B1. Fish C3 was also removed. The treatment group seems slow moving, they seem to just be floating and then flick tail, and then continue to float.
Zebrafish Data 02/29/16
| ' | 1 | 2 | 3 | 4 | 5 |
| A | Alive | Dead | Alive | Alive | N/A |
| B | Dead | Dead | Dead | Dead | Dead |
| C | 1 Alive, 1 Dead | Dead | Dead | Alive | 3 Alive |
| D | Alive | Alive | Dead | Dead | Dead |
Treatment:
| ' | 1 | 2 | 3 | 4 | 5 |
| A | Dead | Dead | Dead | Dead | N/A |
| B | N/A | 2 Dead | 1 Dead | N/A | Alive- very slow moving |
| C | Dead | Dead | Dead | N/A | N/A |
| D | Dead | Alive | N/A | N/A | N/A |
Only 2 fish remain in the treatment, both are extremely slow moving, the salt may have affected their speed. Under the compound:
D1, B2,
D1: Curled in formation, body in the middle of disintegrating B2: Newly dead, extended abdomen, something inside of abdomen.