User:Jennifer Quinn/Notebook/Biology 210 at AU
2/26/16 - Week 7 Lab Notebook
My partner and I retrieved our zebrafish and recorded their length this week, with each fish about 4mm. We removed one mL of old water from each fish's well along with all the old shrimp brine matter we could pick up and added one mL of either fluoride or control water to each well. We also fed all the fish 20 microliters of shrimp brine, in contrast to the 15 microliters normally fed to each fish, in anticipation of not being able to feed them over the weekend when the lab might be closed. This was good as the food had run out on Monday (2/29) when I tried to feed them.
We also learned that the results of the bacteria we collected and submitted for DNA analysis and our transect did not yield any results. The results from bacteria from our transect from spring of 2015 are:
10^-3 Sequence: NNNNNNNNNNGNNGCNNNNNTGCAGTCGAACGGGGTANGCGTAANGCTGCTNCTAGAGGGGGAGAGGGGTGCGTGAGGGC NNAGCGTCTGACTGACCGCGGGAGAGGGGTTGAAANAGGAACGCTAAAACCATGAACGTCACAGAGGAAAAGGCGGGAGA CGGTGGCCTTTTGCTCTATTTAATGATNATGGGTGGGAGCCCGAGANGGTGGTGGTNNTCNCCGTAANGGNAAGATGTAT ATGGGGTCAGAGAANAAGCTNAGACNCAGAGAAACTGAGACAGCGTCCAGACTCATACGGGGGGNGGGTGTGGAATTTGG GGCAATGGGGGAAAGGGCCATATCCAGCCGTGCCGCGTGTGCGAAGAACGCCTGAGTTTTGCCCTGCTTTTTGTCTTGNG ATGAAGGCCAGGAATAAAATATTCTACNCTTGTGATTCTGACTGTACAAAAAGAATAAGGACCGGCTAACTCCGTGCCCG CAGCCGCGGTAATACGGAGGATGCTAGCGTTATTCAGATTTATTGGGTTTAACGCGTGCGTATGCGGTCTTTTAATTCAA TGGTCAAATACTCTACCTCAACTGTCGCATTGCCTTTGATACTGTAGTACTTGAGTCTGTTTGANGTGNGTGCAATTACA CGTGTAACGGTGATATNCATANATATGTCNNGGANCTCCNATTGCGNNNGCCTCTCCCTANNCTNNCACTGACGCTNATG CACGAANGNNNNNGGATCGAACAGGATTACATACNCTCNNNNNCCACNCCCTANACAATTATAACTTGATGTTTGNNAAG ACAAAANNCTNCCCNCCCNCCCCAANCNTTTNNTTNNTNNNCCTNNGGAGGAGGNNCNCCCNGGTGAANACTCNNNNGNN TTTNNNNNCNGNCNNCACNCNCNCANNANNNGNNNNNNNNNNNNNNTNATACNNNAANNAACCNTGCNNCCCCTCCTTNN NNNNNGGGGGNNGNGGANAANNANNNNNNNNNNGNTNNNNNNNNNNANGANGNAANNNNNNNNNGGNGGGGNNNNNNNNN NNNNNNNTNTTNNTANNNNNANNNGGNGNNNNNANCNNCNNNNNGTNNNNN
The blast from this sequence led to the result of "Uncultured bacterium clone SBB39 16S ribosomal RNA gene, partial sequence." This bacteria is unknown and the NIH page about it from teh blast page is linked here: NIH GenBank
10^-3 tet Sequence: NNNNNNNNNCNTANNNTGCAGTCGTANCAANGTAGCCNNNNNNTCCTGGCGGCGAGTGGCGAACGGGTGAGTAATACATC GGAACGTGCCCAATCGTGGGGGATAACGCAGCGAAAGCTGTGCTAATACCGCATACGATCTACGGATGAAAGCAGGGGAT CGCAAGACCTTGCGCGAATGGAGCGGCCGATGGCAGATTAGGTAGTTGGTGAGGTAAAGGCTCACCAAGCCTTCGATCTG TAGCTGGTCTGAGAGGACGACCAGCCACACTGGGACTGAGACACGGCCCAAACTCCTACGGGAGGCAGCAGTGGGGAATT TTGGACAATGGGCGAAAGCCTGATCCAGCCATGCCGCGTGCAGGATGAAGGCCTTCGGGTTGTAAACTGCTTTTGTACGG AACGAAACGGCCTTTTCTAATAAAGAGGGCTAATGACGGTACCGTAAGAATAAGCACCGGCTAACTACGTGCCAGCAGCC GCGGTAATACGTAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGTGCGCAGGCGGTTATGTAAGACAGTTGTG AAATCCCCGGGCTCAACCTGGGAACTGCATCTGTGACTGCATAGCTAGAGTACGGTAGAGGGGGATGGAATTCCGCGTGT AGCAGTGAAATGCGTAGATATGCGGAGGAACACCGATGGCGAAGGCAATCCCCTGGACCTGTACTGACGCTCATGCACGA AAGCGTGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCACGCCCTAAACGATGTCAACTGGTTGTTGGGTCTTCACTG ACTCANTAACNAAGCTAACGCGTGAAGTTGACCGCCTGGGGAGTACGGCCGCAAGGTTGAAACTCAAGGAATTGACGGGG ACCCGCACNAACGGTGGATGATGTGGTTTAATTCNATGCAACNCNAAAAACCTTACCCACCTTTGACATGTACGGAATTC NNCAGANNTNGCTTANGGCTCNAANNANAACCGTTACACNGNTGCTGCATGGNNGTCNTCAGCTCGTGTCNNGAGNNNNT NGGGNTAANTCCCGNAACNAGCNCNNACCCNNTGNNN
The blast from this sequence led to the result of "Variovorax paradoxus strain 57-Y925 16S ribosomal RNA gene, partial sequence." This bacteria is Variovorax paradoxus and, according to a PubMed article, is a gram-negative, aerobic bacteria that helps in breaking down plant matter and complex chemicals.
The sequences are from Liam Purdy's notebook (http://openwetware.org/wiki/User:Liam_Purdy/Notebook/Biology_210_at_AU).
2/19/16 - Week 6 Lab Notebook
My zebrafish lab partner and I presented our paper on the effects of fluoride on frog embryos, talking about how it delayed development.
We got together and created a schedule as to who would take care of the fish when and made a chart for the data we wanted to observe. Then, we each selected 20 zebrafish eggs and placed them in our well plates. The eggs were pretty hard to find because most of the ones in the bowl we selected from were already dead or had fungus growing on them. I placed my 20 eggs in the control solution of water with antifungal and my partner placed hers in the experimental 10mg/L fluoride solution. Throughout the last week, we have come in once a day to feed them shrimp brine solution and change out their water. On my first day, day 4, I removed most of their egg casings as well and have removed any I have seen since.
There are a few procedures in the lab manual, such as observing tadpoles and chick embryos, that we did not have time for in class and were not able to do.
2/12/16 - Week 5 Lab Notebook
First, I observed the live Planaria under the dissecting scope. These looked like writing little brown worms, and the way they moved reminded me of an amoeba.
I next identified the 5 arthropods into their five classes.
1. Insect

2. Chilopoda

3. Crustacea
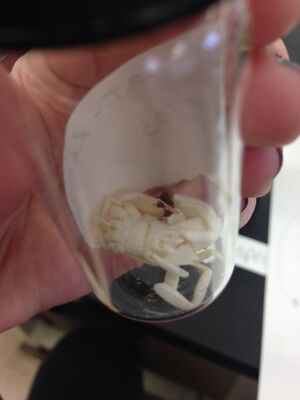
4. Arachnida

5. Diplopoda

Then, my group members and I looked at the contents of our Berlese funnel under a dissecting microscope. From this point, we could see two creatures in the bottom half of our Berlese funnel. We could not see any more than those, though, so I took a sample of the liquid in the petri dish and put it in a depression slide to look at it under a more powerful microscope. Now, I could see a third, smaller creature. The following were what we saw in our Berlese funnel.
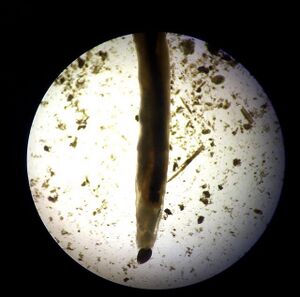
1. Annelida Amelida: 3 in sample at length 400 micrometers. Long body and visible digestive tract.
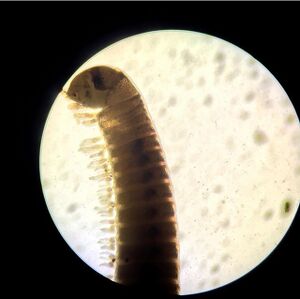
2. Diploda (round millipede): 4 in sample at length 1200 micrometers. Long body with many legs and a head.
3. Mite: 1 in sample at length 100 micrometers. Clear body with legs and small hairs on back.
Food Web for my transect
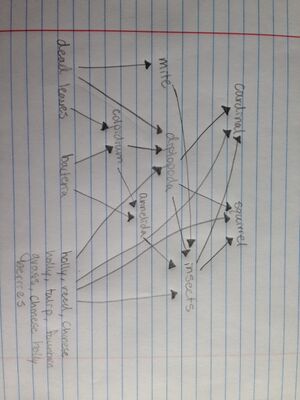
2/5/16 - Week 4 Lab Notebook
My group members and I went out to our transect again to collect more samples. This time, I collected leaf litter and soil in a gallon-sized plastic bag while the other group members selected several different types of plants to take samples from. Once everything was collected, we brought it back to the lab and began identifying the plants. We searched though guides and databases online to figure out what types of plants were present in our transect. The five plants we collected and identified were:
1. American Holly

2. Common Reed

3. Chinese Holly (and berries)

4. Tulip Leaves

5. Fountain Grass

After we identified and found the characteristics of our plants, we set up the Berlese Funnel. The funnel was set up as an ethanol/water solution poured into a conical tube with an enormous funnel placed inside of it. We put a piece of screen in the bottom of the funnel and taped the funnel to the conical tube with parafilm and tape. Then, we poured some of the leaf litter I had collected into the funnel and attached the whole thing to a ring stand close to (although not quite underneath) some bright lights. The lights were there to dry out any plant matter so that any invertebrates would fall through the funnel and be preserved in the ethanol. I know that I saw at least two small worms and one ant in the bag that I collected, so I will be interested to see if they show up in the solution.
1/22/16 - Week 2 Lab Notebook
The first activity that we did was to examine our hay infusion from our transect that had been sitting for a week. The appearance of the hay infusion was rather disgusting, looking like a tiny swamp. It contained assorted green, brown, and gray colors. There was no apparent growth visible with just the eye other than a thin, clear film on the top of the water. I took two samples of the hay infusion, one from the top of the liquid and one from the bottom, to prepare wet mounts.
I was then to find three organisms from each niche, however I could only find two different organisms on the bottom niche, which I examined first. These two creatures were (1) colpidium, measured at 70 micrometers, and (2) what I can only imagine is some sort of diatom, measured at 2.5 micrometers. This second organism was very small, and looked like a tiny brown worm. (picture help needed, cannot get picture to have both normal size and frame with description)
The top niche had three organisms that I identified. These were (1) bacillus, measured at 25 micrometers, (2) phacus, measured at 30 micrometers, and (3) what I think is some sort of infusoria, measured at 20 micrometers. I believe it to be some sort of infusoria because this video about an infusoria is the closest I could find to what I saw. https://www.shutterstock.com/video/clip-5001734-stock-footage-real-video-of-cilia-motion-of-live-cell-infusoria-with-simbiontic-green-bacteria-microcystis.html?src=search/2XhBWyeMP0Lgjd2aCABcLQ:1:14/gg
After looking at the slides from the hay infusion, my partner and I did a serial dilution with the liquid from the hay infusion to put onto agar petri dishes. We added 100 microliters of the hay infusion to 10mL of broth in a test tube, making a 1:100 dilution. 100 microliters were then taken from this test tube and placed into the next test tube with 10mL of broth. This continued until the four test tubes, labeled 10^-2, 10^-4, 10^-6, and 10^-8, were each infused. Then 100 microliters of each solution were pipetted into the agar petri dishes, labeled 10^-3, 10^-5, 10^-7, and 10^-9. Each solution was pipetted into two petri dishes, one with tetracyclane (marked with three black stripes) and one without. The petri dishes were then left to incubate.
1/15/16 - Week 1 Lab Notebook
The transect that my lab group and I got is Transect 3. The first thing we did was go out and inspect this plot. We located all four of the popsicle sticks marking the corners of our transect and assigned them each a number. Then, I walked around and took pictures of the area, some of which are displayed below.
After taking pictures, I drew an aerial sketch of the transect, which is shown here.
Finally, my lab group and I took samples of dirt and plants from different parts of the transect. These samples included soil from different locations, leaves, flowers, and twigs.
When we returned to the lab after inspecting the transect, we prepared the hay infusion. The hay infusion consisted of 13.33g of our sample from the transect (soil, leaves, flowers, and twigs), 500mL of distilled water, and 0.12g of dried milk powder. These were put into a large jar and shaken together. Then, the jar was left out with its lid off.
After the hay infusion, we looked at three types of Volvocine, Chlamydomonas, Gonium, and Volvox, under a microscope. The following data was collected about the Volvocine in a table.