User:Grace Little/Notebook/Biology 210 at AU
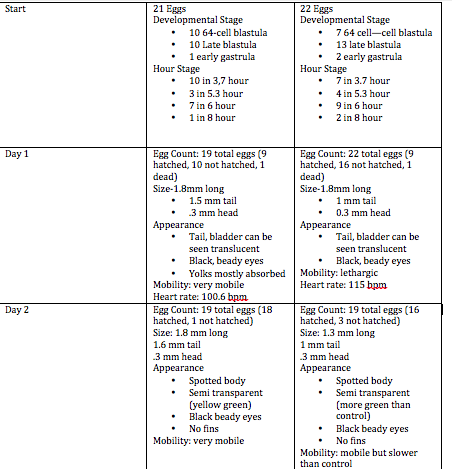
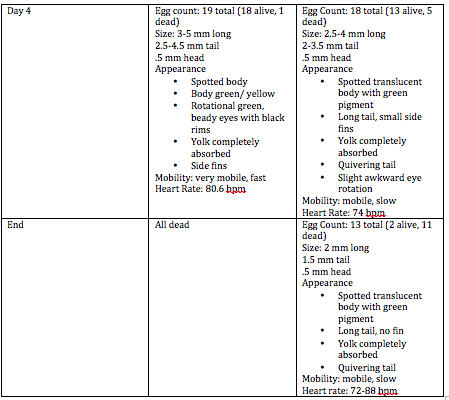
Embryology of Zebrafish March 24 2014 Question: How will fluoride affect the embryological growth of zebrafish? Specific Steps preformed: First we read a lab report that already documented the affects of fluoride in tadpoles. Then we set up 2 petri dishes with 20 zebrafish eggs and 20mL of water, one with fluoride and one as the control with normal water. We then recorded data on their changes as the days went by. We calculated their heart rate, and wrote down their physical development and attributes. We put 3 zebrafish in 4% formaldehyde for future observation underneath the microscope. Raw data:
Conclusion: It was interesting to see the effects of fluoride because it is found in our water as well. We can relate to the effects because it is so widely accepted in our water systems and at the dentist. It is important to note the psychological changes due to fluoride and know that we may need to begin reevaluating the amount of fluoride that we are intaking.
March 16th Mini Lab DNA Questions: What are the specific strands if bacteria/DNA?
Specific Steps Preformed: We first had to isolate the bacteria that we wanted to use in the PCR reaction. We isolated the bacteria that had the best characterization of our transect. We used two primer sequences to amplify the rRNA gene. We then put the colony of bacteria into 100 microliters of water. We incubated the tube at 100 degrees celciuse and centrifuged it. The following week we ran the PCR products in an agarose gel. This separates the DNA into size. Some of the products were sent to Genewiz Inc for sequencing. Our sample did not get sequenced.
Conclusion: Overall this mini lab gave me a better understanding of how to sequence DNA. Even though our specific DNA did not get sequenced to determine the specific names, we analyzed other DNA strands that were sequenced. This was helpful in knowing how to input the DNA bases in the computer and know how to find them.
Feb 25 2014
Lab #5
Questions: What is the importance of invertebrates? How have simple systems evolved into more complex systems?
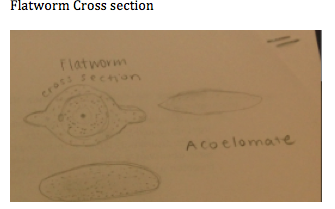
Specific steps preformed: The first procedure that we preformed today was to observe acoelomate, Planaria with the dissecting microscope. We are able to see the digestion of the planaria. We then looked at a nemotode and compared and contrasted the differences of an earthworm and a cross section of the earth worm. We then looked at live worms and saw that worms, in dirt, bended in a wave-like formation, slow moving or not moving at all. They also moved with their entire body. The next activity that we did was to look at our own invertebrates collected by our berlese funnel. We put the invertebrates into two differing petri dishes and looked at them under a dissecting microscope. We used a dichotomous key in order to identify each invertebrate. In order to become more familiar with invertebrates, we looked at many invertebrates in jars to determine which arthropods they belonged to. An example of these were a centipede, a crustceans, an arachnid, and a millipede. This really helped my understanding in knowing how to determine the species of each organism.
Raw Data:



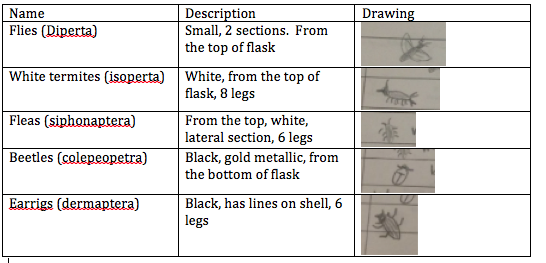 From this data, I saw that the beetles were the largest invertebrates than the others. I also saw that most of the bigger invertebrates were found from the bottom of the test tube when we poured out our invertebrates.
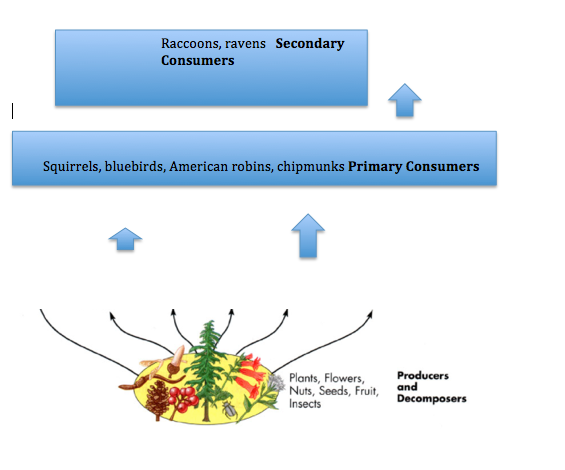
From this information about invertebrates, some vertebrates that may be found in my transect are a sialia, which is a bluebird, and a turdus migratorious, which is an american robin. Other vertabrates that would be found in my transect is a sciuridae (squirrel), a tamias (chipmunk), and a procyon lotor (raccoon). The last three classifications would be eukarya, animalia, chordata, mammal, rodentia
and thena squirrel would be ciuridae, scours, and sciurus carolinensis.
raccoon:
order: carnivora genus: procyon and species:lotor
chipmunk:
suborder: sciuromorpha, sciuridae, marmotini, and its genus is tamias
blue bird classification:
phylum: chordata
class:aves
order: passeriformes
family: muscicapidae
genus: sialia
American robin:
phylum: chordata
class: aves
order: passeriformes
family: turdidae
genus: turdus
These animals could survive in my transect off of the habitat provided. There is green grass for the squirrels, chipmunks, and raccoons to scavenge for fallen nuts on the ground. The birds could live in the trees and build their homes in the branches.
From this data, I saw that the beetles were the largest invertebrates than the others. I also saw that most of the bigger invertebrates were found from the bottom of the test tube when we poured out our invertebrates.
From this information about invertebrates, some vertebrates that may be found in my transect are a sialia, which is a bluebird, and a turdus migratorious, which is an american robin. Other vertabrates that would be found in my transect is a sciuridae (squirrel), a tamias (chipmunk), and a procyon lotor (raccoon). The last three classifications would be eukarya, animalia, chordata, mammal, rodentia
and thena squirrel would be ciuridae, scours, and sciurus carolinensis.
raccoon:
order: carnivora genus: procyon and species:lotor
chipmunk:
suborder: sciuromorpha, sciuridae, marmotini, and its genus is tamias
blue bird classification:
phylum: chordata
class:aves
order: passeriformes
family: muscicapidae
genus: sialia
American robin:
phylum: chordata
class: aves
order: passeriformes
family: turdidae
genus: turdus
These animals could survive in my transect off of the habitat provided. There is green grass for the squirrels, chipmunks, and raccoons to scavenge for fallen nuts on the ground. The birds could live in the trees and build their homes in the branches.
Conlcusions/ Future Plans
This lab bettered my understanding of invertebrates. I also thought that using a dichotomous key to determine types of invertebrates was helpful because it forced us to pay attention to detail of our collected invertebrates. Overall, I thought this lab was a great way to get to know a specific niche and the inhabitants that reside there. It also dictated the importance of keeping our campus clean, because it is an environment which sustains life to others than just humans!
Feb 16 2014
Lab#4
Question: What are the characteristics and diversity of plants? What are the functions and importance of fungi?
Specific steps preformed:
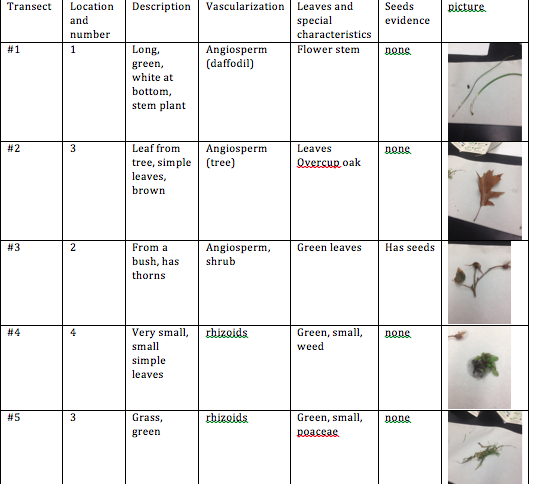
The first activity that we did today was to bring three bags to our transect. This was in order to collect plants and a mix of soft soil and dead leaves. We then took photos of trees and made sure to identify the types of wildlife that we saw. We then looked closer at different types of plants, such as a moss and compared the moss to a stem of an angiosperm, lily. We observed the xylem and phloem. In our transect, most of the plants were angiosperm, meaning that the stem supported the weight of the plant and water/nutrients needed to be transported. On the patio, we collected moss which had rhizoids which anchor the plant and allow it to grow/spread along the ground. The plant that we brought back (the shrub with seeds) was a monocot seed. Next, we saw that the fungi sporangia were important because they are spores for reproductions. The final step of this lab was to set up the berlese funnel to collect invertebrates. Earlier, we had collected a bag filled with soil and leaves. We used this to put into our funnel. We poured 25 mL of the 50:50 ethanol/water solution into the flask beneath the funnel opening. Next, we put a screen to filter out the invertebrates and soil underneath the funnel opening. We put the leaf litter on the stop of the funnel after the soil. We covered our funnel with foil and left it to study for the following week.
Raw data:
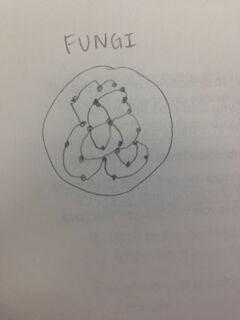 This is a fungi because it has sporangia.
Conclusions/Future plans:
Overall, I think this lab was helpful in understanding plants and their vascularization systems. I am excited to see what will come out of our berlese funnel next week-and what invertebrates we will be able to find from our transect.
This is a fungi because it has sporangia.
Conclusions/Future plans:
Overall, I think this lab was helpful in understanding plants and their vascularization systems. I am excited to see what will come out of our berlese funnel next week-and what invertebrates we will be able to find from our transect.
Lab #3 Question: What are the characteristics of bacteria? What is antibiotic resistance? How are DNA sequences used to identify species?
Specific Steps Preformed: The first step we had to take in this lab was to observe our bacteria and hay infusion. Last lab, we made dilutions of petri dishes. In this lab we observed each petri dish and counted the number of cultures of bacteria that we found on each dish. If there was a lot covering the surface, we called this a lawn. We repeated this step for each plate table and counted the colonies for each plate. The next step we took was to understand the differences of the petri dish with nutrient and antibiotic resistance versus the petri dishes without the antibiotic. We observed that the antibiotic kills bacteria and there was less bacteria on the plates with the antibiotic. Therefore, with the tetracyline, the number of bacteria and fungi was significantly less. Yet, there was still one colony on the plate 10^-7 and 2 colonies on the plate 10^-5. The next step that we took in this lab was we observed bacteria cells on prepared slides. We then made our own wet mount of our bacteria and observed it under the microscope. We then prepared a gram stain on each slide. We observed the slide with the Gram's iodine on it under the microscope. Finally, we started the PCR preparation for next weeks lab. We isolated DNA and transferred a sing colony of bacteria to 100 micro liters of water into a tube. This then was centrifuged and we used 5 micro liters of the supernatant in the PCR reaction.
Raw Data:
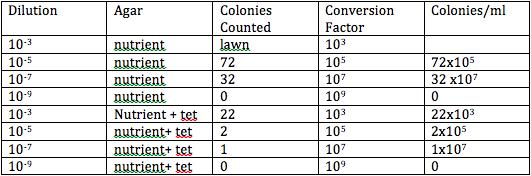
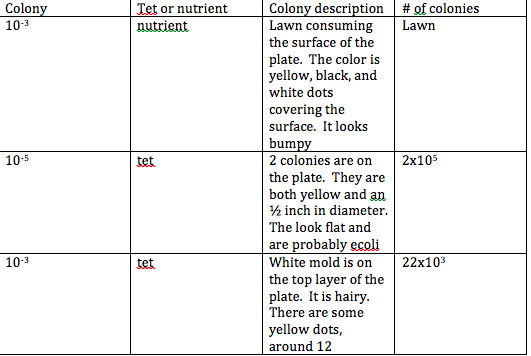 The appearance of the hay infusion would probably grow more moldy and contain more bacteria week to week because we are allowing the bacteria and mold to grow. Tetracyline does not kill bacteria, it curtails their ability to reproduce. There is an enzyme that inhibits the vital processes of bacterial cells.
The appearance of the hay infusion would probably grow more moldy and contain more bacteria week to week because we are allowing the bacteria and mold to grow. Tetracyline does not kill bacteria, it curtails their ability to reproduce. There is an enzyme that inhibits the vital processes of bacterial cells.
Conclusions/Future Plans: In conclusion, this lab allowed me to understand the purpose of an antibiotic. It also exposed me to understanding bacteria and mold, and the differing kinds of each. I am excited to see what PCR reaction will reveal to us about our bacteria next week. It will be interesting to analyze the sequences and see the DNA of our sample. GL Feb 8 2014
Lab #2 Question: How to identify and study unicellular eukarya? How to use a Dichotomous (Ward's Free Living Protozoa) manual to identify cells. What types of of eukarya are in our hay infusion?
Specific Steps Preformed:
In order to get familiar with the dichotomous key, I prepared wet mounts of slides and looked at them under the microscope in order to get a better understanding of how to identify organisms. I did this two times with a KNOWN organism in order to assure that I understood how to work with the dichotomous key. I made observations on size, shape, motility, and characteristics in order to identify the organism . I then drew the organisms in my lab book.
The next step of the lab was to make wet mounts of our hay infusion sample. First we made observations on how our hay infusion had changed since last week--for example, smell, color, and any seen organisms. We then made wet mounts of our sample to study underneath the microscope. To do this we extracted some of the liquid from two different areas of the jar and made a wet mount. I recorded and drew the organisms observed and used the dichotomous key to characterize the organisms.
The final step of lab was to prepare dilutions and put them on agar and nutrient plates in order to observe bacterial growth for next class. We had to mix the infusion up and then use a micropipete in order to create the ratio of sample to water. We had to do math in order to get the ratio of water to sample exact. We then smeared the dilution on the agar plates for observation for next weeks lab.
Raw Data:
Hay infusion: smells like a pond, the water is a brownish color, algae growing on the side, brownish film on the top of the water, layer of dirt on the bottom of the jar.
We obtained our two samples from the infusion from the bottom layer near the dirt, and another sample from right next to where the blade of grass is. I feel like there will be more diversity near the blade of grass because it will provide more nutrients for organisms, whereas the dirt is a little bit darker and not as much photosynthesis is happening. There is also nutrients in the dirt that would provide life for organisms.
If we had let the hay infusion sit for another two months, i feel like the humidity within the jar would have increased and there would be more of an ecosystem growing, such as more algae on the sides.
Selective pressures affected sample: The yeast added to the jar is an added pressure and probably increases the amount of production. It was also indoors, and thus a warmer temperature than the outdoors. Lastly, there was no human impact that could have altered the infusion, whereas outside where we collected our sample human impact would have altered the ecosystem.
Pictures of organisms and dilution procedure attached.
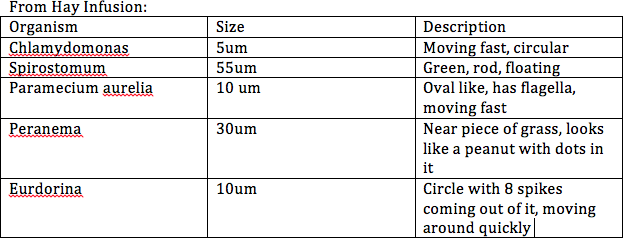

 Conclusions/Future Plans:
For the next lab we will observe the agar plates and how each dilution differs, as well as the impact of of tetracycline on the samples. In conclusion, this lab really bettered my understanding of how to use a dichotomous key and characteristics of organisms. It was very interesting to see the diversity within our own hay infusion and I am excited to see what will happen to the agar plates next week.
Conclusions/Future Plans:
For the next lab we will observe the agar plates and how each dilution differs, as well as the impact of of tetracycline on the samples. In conclusion, this lab really bettered my understanding of how to use a dichotomous key and characteristics of organisms. It was very interesting to see the diversity within our own hay infusion and I am excited to see what will happen to the agar plates next week.
GL
2/6/14, lab 1 notes
Great job, Grace! Make sure to include pictures from lab 1 and lab 2 by Sunday. Second, Make sure that the newest lab entry is the first to appear. So, move 1-22 down to the bottom. For lab 2, that will be the top entry by Sunday. Third, start working on building a map of your transect to detail your land and where your samples are taken from. We will talk about this more Wednesday. Good job!
AP
January 22 2014 Successfully entered username and text into my lab notebook GL
January 31 2014 Lab #1
Question: Our objective for lab today was to understand the ecosystem of a niche and how abiotic and biotic materials contribute to an overall setting. Questions that went a long with this lab were what is natural selection? what is a niche? What are biotic and abiotic factors? How do specific organisms evolve? Specific Steps Preformed: For the first part of this lab, we wanted to observe evolution. I made a wet mount of differing green algaes, such as Chlamydomonas, Gonium, and Volvox. By looking at these varying cells, I can see the differing traits that they obtain. I then calculated the size of each cell, and drew them in my lab notebook. I described specific traits that I could see for each cell--such as isogamy or oogamy.
The next part of the lab was being assigned a transect outside and describing the abiotic and biotic features of the landscape. In my lab notebook, we wrote down the location, topography, biotic and abiotic factors. We then used a sterile conical tube and collected 50 mL of soil in order to make a hay infusion. To make the hay infusion, we collected 10grams of our sample, mixed it with 500 mls of water, and .1 gm of yeast in a jar. We shook the jar and placed it on our sections tray for next weeks lab.
Raw Data: Chlamydomonas: number of cells: 60-65 unicellular colony size: 10 micrometers functional specialization: stigma, flagella reproductive specialization: isogametes, isogamy Gonium: number of cells: 1 colony size: 550 micrometers functional specialization: motile colony reproductive specialization: isogamy Volvox: number of cells: 5 colony size: 280 micrometers-150 micrometers functional specialization: spiked cells anterior and posterior poles reproductive specialization: oogamy
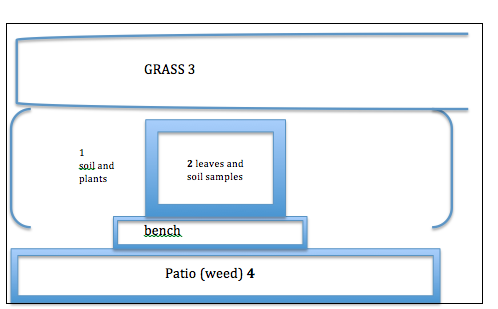
For my specific lab group, our transect was on the quad. The specific dimensions had varying landscape. Some of it was covered by a stone patio, some was green grass, and the other part were shrubs growing. Location: American University, quad, Washington, DC. Eric A Friedheim Quadrangle Topography: flat, grass, planted bushes, elevate soil, stone, stone bench. Human impact/walking Abiotic: soil, stone, patio, bench, fallen leaves, litter/ gum wrapper, rocks, fallen sticks Biotic: grass, bushes, humans/students, weeds, daffodils
Conclusions/Future Plans:
In conclusion, I gained a better understanding of evolution and how cells change through time. Cells adapt to their surroundings--natural selection.
I also got a better understanding of a niche and how it plays into the biotic and abiotic factors.
Next week, we are going to do more tests to our hay infusion for our specific transect. I am excited to see what organisms are inhibiting in our transect, and if there are a lot of growing organisms or if our niche is non-habitiable
GL