User:Erika Acosta/Notebook/Biology 210 at AU
3/17/14 Embryology and Zebrafish Lab
Objective: For this lab, we had to observe certain factors that can effect zebrafish development. Zebrafish are part of the minnow fish family and are used for much experimentation due to their transparency during development that can be easily observed. My partner and I chose to expose 40 zebrafish (20 test and 20 control) to caffeine, by which we observed on days 1, 3, 6, 7, 9, and 13. According to a Taiwanese study titled, “Movement disorder and neuromuscular change in zebrafish embryos after exposure to caffeine” (Chen, et al. 2008), they have found that when zebrafish were exposed to caffeine, it resulted in disorganized muscle fibers, tactile sensitivity, a shorter tail length, shorter life span, and failure of neural tube closure. Studying this topic would help us gain insight as to what the effects of caffeine may have on humans during early development. The hypothesis we have developed for this experiment was that caffeine would result in stunted growth, increased motility, and shorter life span.
Methods:
Day 1: On this day, we obtained 40 zebrafish larvae and randomly placed 20 in a control group and the other 20 in a test group. The control group was treated with 25 ml distilled water and the test group was treated with 25 ml prepared caffeine solution. We then made initial observations, which were: swimming and movement, yolk absorption, eye pigmentation/movement, melanophore development, heart rate, tail/fin/swim bladder, tail length, total body length, and eye diameter.
Day 3: On this day, we observed how many of the larvae hatched and characterized what larvae sage they were in. We changed the water as well (removing and replacing equal parts of distilled or treated water to the designated group). Then, we made our routine observations (swimming and movement, yolk absorption, eye pigmentation/movement, melanophore development, tail/fin/swim bladder, tail length, total body length, heart rate, and eye diameter).
Day 6: On this day, we counted the number of zebrafish hatched and the number of zebrafish dead in each group and again made the routine observations.
Days 7, 9, and 13: On these days, we again noted the zebrafish who were alive in each group and the zebrafish who were dead in each group and made our routine observations.
Results:
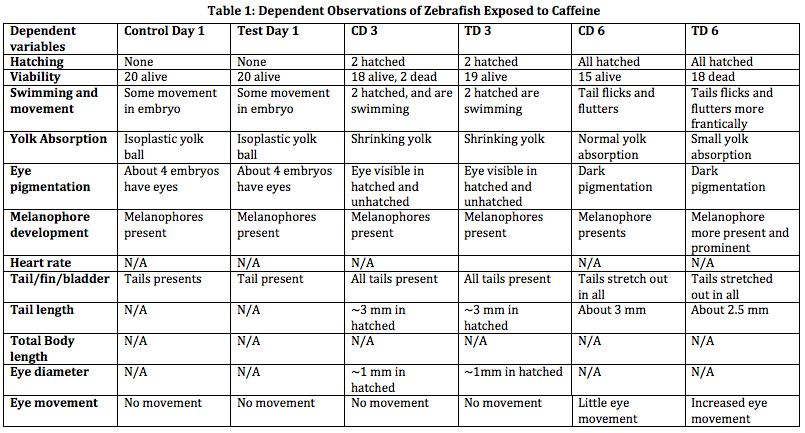
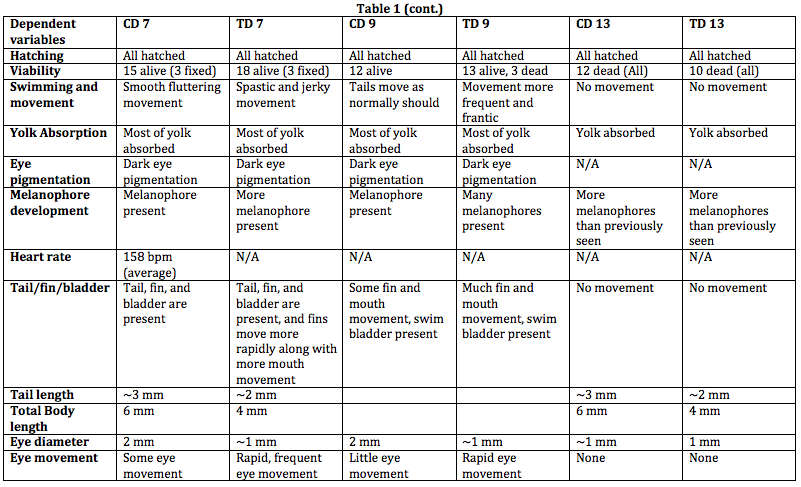
After about 2 weeks of observation, we have found our hypothesis to seem valid. We stated that caffeine would result in stunted growth, increased motility, and shorter life span. As shown in Table 1, the zebrafish was shorter and skinner in the test group than in the control group. The zebrafish exposed to caffeine also seemed to move more frantically and quickly in swimming and has shown to have more rapid eye movement. The caffeinated zebrafish died more quickly than its counter group as well. An increased development is shown in the caffeinated zebrafish as well. This can be seen in the amount of melanophores present in the control group than in the test group.
Discussion: Something we did not expect in this experiment was that the zebrafish exposed to caffeine were a lot skinner. We have found that for the most part, we have proven our hypothesis as well. During this experiment, however, we did not expect such a mass death upon the 13th day. This is probably due to the fluctuating temperatures the zebrafish were placed in. We also had a hard time find the heart rate of the zebrafish, since we could not locate its heart. An improvement would be that the zebrafish should be placed in constant temperatures and one should also find a way to stabilize the zerafish, such as putting them to sleep in order to locate their heart and record the heart rate.
From this experiment, we can parallel this to human consumption of caffeine during pregnancy. Since there is very little studies that show the effects of caffeine in early development, we can start by experimenting on zebrafish. Similarly in zebrafish, a pregnant mother who consumes large amounts of caffeine may expect to see a fetus who moves frequently, a low birth weight (like how the zebrafish in caffeine were shorter and skinner), and a baby with metabolism issues.
EA
2/26/14 Lab #7
Objective: In this lab, we sent out the DNA sequences we have obtained in Lab #3 from our transects in order to determine what type of bacteria resided in our transects and also to compare it with the results we have found.
Procedure: Refer to Lab #3 for PCR procedure.
Results:
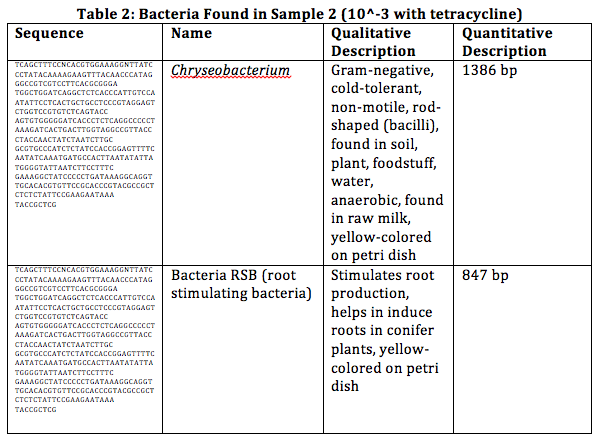
Conclusion: Before this lab, we have found that many of our bacterium were cocci and some were bacilli. From the sequencing, we have found that all of our samples contained bacilli. During the Gram staining, we have found that our samples were Gram-negative, which has been proven to be true through the DNA sequencing.
EA
2/12/14 Lab #5
Lab Objections: Invertebrates are found everywhere and all contribute to the natural cycles the Earth provides for us. There are over 14 different phyla of organisms. In this lab, we explore the importance of invertebrates and also learned more about simple system, as seen in many invertebrates, and how they have evolved into more complex systems (such as our systems). One can identify these systems through the body cavities of the organisms. This would include the ectoderm, the mesoderm, and the endoderm. We will examine these concepts through observing and identifying invertebrates from the Berlese funnel from our transects as well as looking at other invertebrates.
Procedure 1: In this procedure, we observed an acoelomate, a pseudocoelomate, and a coelomate, which would be a Planarian, a nematodes, and an annelid, respectively under a microscope. We then recorded the characteristics we noticed.
Procedure 2: In this procedure, we dissembled our Berlese funnel from the previous lab and obtained the ethanol (which now should have debris and possibly some invertebrates) and poured it into a petri dish. Then, we observed it under a dissecting microscope in order to get a better look as to what invertebrates may have been lurking in our transects.
Data: From procedure 1, the Planaria has a more primitive digestive system, excretory, and nervous system. It moves like a worm, it stretches across and contracts. In the cross section, it has various gut branches as well. The Planaria twists and turns as it moves.
The adorable planaria and its cross section
The movement of the annelida (the earthworm) can be characterized as stretching and contracting to get from one place to another. Worms are allowed to do this since they have stretchy body segments made for their movements. They also twist and move around as well. As for the cross section, it is a lot more complex, where it consists of the endoderm, the ecoderm, and the mesoderm. This would mean that they are much more evolved.
 The earthworms doing normal worm-like activities, while being unknowingly observed.
The earthworms doing normal worm-like activities, while being unknowingly observed.
The nematodes cross section is complex as well, but not as complex since its organs are not organized as much as an annelid or a planarian. The nematode slides cross and does not seem to exhibit similar movement as planaria or earthworm.
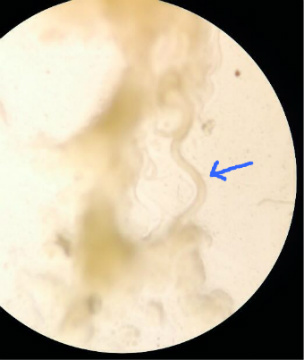 The poorly drawn arrow indicates the nematode (“Ah, nematodes!" – Spongebob Squarepants)
The poorly drawn arrow indicates the nematode (“Ah, nematodes!" – Spongebob Squarepants)
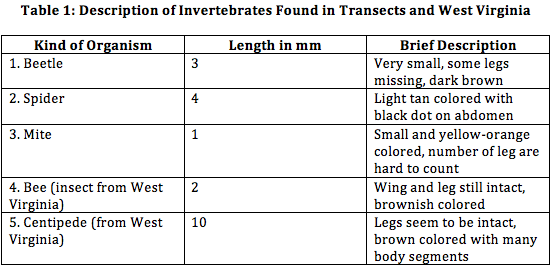
From procedure 2, a chart was organized in order to identify the various invertebrates that were found in our transect:
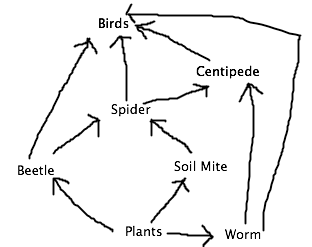
The organisms range from 1 mm to 4 mm, being the beetle and the mite, respectively. The smallest found in West Virginia would be the bee and the largest would be the centipede. Common organisms found in leaf litter would be worms, spiders, and beetles (since the decomposing leaves would provide nutrients and shelter).
Food web of organisms from transect:
Conclusion: There can be many organisms that inhabit a 20 ft by 20 ft piece of land. The reason as to why we had to obtain specimens from West Virginia was due to finding a lack of organisms from our Berlese funnel. To correct this, we can obtain more soil or should have obtained the soil more deeper into the Earth. Although not mentioned, other organisms in our transect would include earthworms, soil mites, ground spiders, sparrows, and pigeons. The sparrow and pigeon would benefit from eating the invertebrates, such as the earthworm and spider, as a mode of nutrition. Some abiotic characteristics that would benefit the birds would be the dead twigs to build nests. As for the earthworm, soil mites, and spiders, biotic factors they would depend on would be other small invertebrates as food or other plant life. Some abiotic factors they would benefit from would be dead leaves for shelter and nutrition, and the soil for shelter as well. From this lab, we have reached our objectives and learned more about invertebrates by identifying the ones we find in our transect.
EA
2/5/14 Lab #4
Lab Objections: Plants are very diverse and just as complex as animals. Plants also contribute to Earth’s natural process. In this lab, we explore the diversity of plants and also examine the function and processes of fungi. To do this, we collected plant samples from our transects in order to further appreciate plants.
Procedure 1: The first part of this experiment requires us to obtain plant samples from the transect. To do this, we first got rubber gloves and gallon-sized. We then headed over to our transects and proceeded to collect various samples of our transect from different areas. We then recorded the plants we found on a chart.
As for the rest of the procedures, it required us to classify and identify other plant species and to observe some fungi.
Procedure to Prepare Berlese Funnel to Collect Invertebrates: To prepare the Berelese funnel, we first have to pour equal parts of ethanol and water into a flask. Then, we fit a mesh screen at the bottom of the funnel and secured it with tape. To assemble, we suspended the funnel over the flask and placed some of the soil, dirt, etc. from the transect and placed a lamp over the entire structure. We then left it for a week for the invertebrates to collect.
Data:
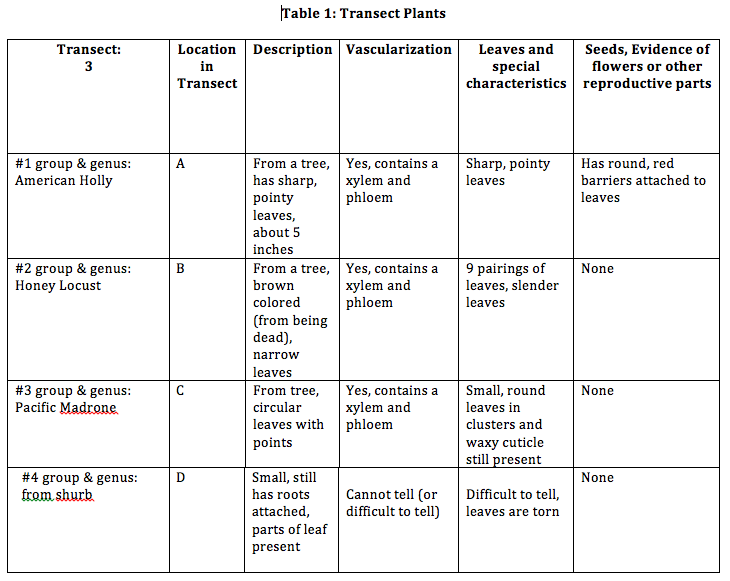
 Image of the plants we have collected from our transect (from right to left: plant from shrub, Pacific Madron, Honey Locust, American Holly)
Image of the plants we have collected from our transect (from right to left: plant from shrub, Pacific Madron, Honey Locust, American Holly)
The letters for “Location in Transect” correspond to the letters as to where the sample was taken.
Fungi Sample:
 This picture was taken while observing the fungi under the microscope.
This picture was taken while observing the fungi under the microscope.
Conclusions: Many of the plants we have found contained a xylem and phloem. These structures help with the transportation of water and food materials (sugar). Under the microscope, we can identify the xylem and phloem more closely. From the seeds we have collected back from the transect, we have found some to be dicots and some to be monocots. From observing the fungi, we have found that the reason as to why fungi sporangia is so important because it is the fruiting body involved in the asexual reproduction of spores. They are important because it is the spores that will spread around so that more fungi will grow. From the fungi sample we observed, we believe that it is a Ascomycota, since it contains sporangia and mycelium, which are structures that are common in Ascomycota. Through the observation of plant structures, we are able to classify plants and better understand them.
EA
1/29/14 Lab #3
Lab Objections: The purpose of this lab was for us to become familiar with various types of bacteria and structures. These structures consist of cocci (round shaped), bacilli (rod shaped) and spirochetes (spiral shaped). In the previous lab, we prepared agar plates to grow bacteria from out transect. Some of these plates had an antibiotic, tetracycline, in which we will also observe and compare it to the plates without antibiotic. Finally, we will try to identify the DNA sequence of some of the bacteria we collected.
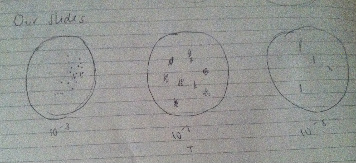
Procedure 1: In this procedure, we obtained our cultures and tried to estimate the amount of colonies where in each dish:
Procedure 2: In this procedure, we had to look at the agar plates with and without and compare the growths. The only materials needed were our agar plates and our eyes.
Procedure 3: In this procedure, we had to take samples of the bacteria on our agar plates and observe any structures that we see. To perform this, we obtained agar plates 10^-3, 10-^-3 (tetracycline), and 10^-5. In order to obtain a sample, we used a stile loops, scraped it gently from the colonies and placed it on a slide. We then observed the bacterial structures under the microscope. Next we had to prepare a gram stain. To do this, we first had to label the slides from what agar plate it was obtained from. Then, we passed the slide through a Bunsen burner three times. Next we covered the smear with crystal violet for one minutes, rinsed with water, covered with iodine for one minutes, decolorized and rinsed by covering it with 95% alcohol, covered the smear with safranin for 20-30 seconds and rinsed. Lastly, we blotted the excess water with a paper towel to dry.
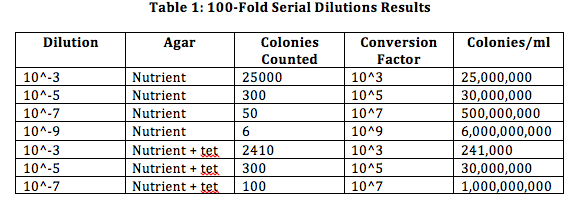
Procedure 4: The last part was to prepare the bacteria for PCR amplification. For this we first had to transfer a single colony of bacteria to 100 microliters in a sterile tube, incubated it at 100 degrees Celsius for 10 minutes and centrifuge, and use 5 microliters of the supernatant in the PCR reaction. Data: From procedure 2, we found that the colonies with antibiotic have shown to be having less colonies, while the ones without the antibiotic had more colonies (especially the 10^-3 ones). This indicates that the antibiotic is working effectively. The effect of tetracycline reduces the amount of bacteria and we did not find any fungi on our cultures. This orange-colored species is not affected by tetracycline. The way tetracycline works is that it inhibits protein synthesis to occur. When protein synthesis does not occur, the bacteria cannot perform its regular functions and will die. Tetracycline works on various types of bacteria such as E. coli, Haemophilus influenza, Mycobacterium tuberculosis, and Pseudomonas aeruginosa.
From observing our bacteria from 10^-3, 10^-3 (with tetracycline) and 10^-5 (in the order from which it appears), we have found various types of bacterial growth in different structures. Most of the bacteria we have observed were cocci, although we did find some that were bacilli.
Conclusion: From this lab, we explored various bacterial shapes and sizes, which helped to identify the bacteria we had in our transects. We also explored the how the antibiotic tetracycline worked. Something interesting from this lab was that we have found that millions of colonies can derive from a square inch of dirt/soil. In this lab, our objections were met and we also prepared the PCR in preparation for this next lab.
EA
2/6/14, lab 1 notes
Great job, Erika! -Make sure each lab has the date and lab number at the top bolded, even if entries were written on the same date.
-Start working on building a map of your transect to detail your land and where your samples are taken from. We will talk about this more Wednesday
Awesome job! AP
1/22/14 Lab #2
1/22/14 Lab #2
Lab objections: There are many types of living creature on our planet. These diverse beings can range from something unicellular and microscopic to something multicellular and as large as an elephant. In this lab, we had to explore the usage of a dichotomous key, which helps us to identify many organisms and in this lab, we also explored two groups of eukaryotes, algae and protists. The purpose of this lab was to expand our knowledge of eukaryotic cell types and to further explore our transects. This lab also included growing cultures on agar plates from our Hay infusion in order to grow bacteria and understand what type of bacteria would thrive. For the first part of the lab, we had to look at various eukaryotes and identify them using a dichotomous key. We also observed this organisms under a microscope to help with the identification process. Then, we made observations of our Hay infusion after having it culture for 2 weeks.
Materials for Culture samples: Hay infusion, micropipettes, slides, slide covers, and microscopes.
Procedure to Observe Culture samples: First use the micropipettes in order to obtain liquid cultures and place on slides. Take cultures from away plant-life and cultures near plant life. Next, use a dichotomous key in order to identify the organisms.
Materials for Plating Dilutions: 4 tubes of 10 ml sterile broth (labeled 2, 4, 6, 8), micropipeter set at 100 microliters, and tips, 4 nutrient agar plates (labled 10-2, 10-4, 10-6, 10-8), 4 nutrient agar plates with tetracycline (labled 10-3, 10-5, 10-7, 10-9).
Procedure: First, swirl the Hay infusion culture to mix all the organisms, then take 100 microliters for this mix and add it to Tube 2 for a 10-2 dilution. Take the 100 microliters from Tube 2 and add it to Tube 4 for a 10-4 dilution and repeat for Tube 6 and 8. Then take 100 microliters from the Tubes and place them on the corresponding labeled agar plates (i.e. Tube 2 goes to 10-2 and so on) and spread. Next do the same for the agar plates with tetracycline.
Data: From observing my culture, I observed that there was a putrid smell, the liquid was green-brown, there was foam on the top of the surface, the water looked murky, and some of the vegetation floated and some sank to the bottom.
I have found that all the organisms found were motile, in which I have identified these organisms to be two arcella, paramecium, two gonium, and a euglena.
The reason as to why the organisms might differ near vs. away from the plant matter is because some of the organisms may use the plant matter for food or energy.
Conclusions: All of the organisms I have collected are found to be motile. The paramecium I have found is a protozoa, the euglena I found is a protist, and the gonium is a type of algae. The euglena and the gonium both undergo photosynthesis, and one can detect this through their green color.
The paramecium meets all the needs of life because it is able to move, it is able to obtain nutrition, it is able to excrete, and it is able to reproduce.
If the hay infusion culture had been observed for another two months, some changes I would observe would be even more organisms and new species. I would probably also observe an even more putrid smell.
Some of the selective pressures that could have affected the compositions of my sample would be the temperature and the limited food supply.
This lab did address the objections, where we first learned how to use a dichotomous key and looked at samples from our cultures. We used the dichotomous key in order to identify these species and also prepared dilutions for our next lab.
EA
Lab #1
Lab objections: In this lab, we were assigned an area on the AU campus and had to observe this transect. Then, we took a sample of the transect (which includes taking a sample of the soil, vegetation, etc.) and prepared it for a hay infusion for the next lab.
Materials: Shovel, tube for soil collection. For Hay infusion: 500 ml of water, 0.1 g of dried milk, 10 - 12 g of soil from transect, and a jar.
Procedure: First we observed our transect and took detailed notes. To prepare for the Hay infusion, we collected 12 grams of soil using the shovel from this transect. For the Hay infusion, we placed the dried milk, the water and the soil that we collected into the jar and stirred. We then left the jar, without the lid, alone for a week.
Conclusion Transect Description: Many shrubs, lots of bare, 6 small trees, nest in the trees, nest in tree, concrete (abiotic), two light poles (abiotic), dead leaves and other dead vegetation (abiotic), a lone bird, spider web on tree (abiotic), rocks (abiotic), cigarettes butts (abiotic), paper cups (abiotic), water.
Due to the season, there is a lot of dead plants on the ground and not a lot of animals.

EA
1/22/14
Successfully entered text into my notebook
EA