User:Emily L. Brackett
Definition
Tissue engineered vascular grafts work as replacement veins and arteries. They can either replace a damaged vein or artery or connect veins and arteries. These grafts can be made from cultured cells or from biomaterials.
Motivation
In the United States, there are 1.4 million patients in need of artificial vasculature each year [1]. These cases pertain to hemodialysis, coronary artery bypass, replacement of damaged vasculature, and more. As the need for artificial vasculature increases, research into synthetic and biosynthetic grafts becomes more and more common and desired.
The main use of vascular grafts is for vascular access during hemodialysis. Hemodialysis (or blood dialysis) is the preferred method used to treat kidney failure and is performed 350,000 times annually [2]. This method uses concentration gradients and a semipermeable membrane to remove waste from the blood. There are three methods to hook up to blood flow in the body to perform hemodialysis. The first method is an arteriovenous fistula, which involves attaching an artery to a vein to increase blood flow to an area, most commonly the arm, then attaching a needle to this vein to extract contaminated blood and put back clean blood. Fistulas usually last the longest out of the three choices and are associated with the least negative side effects. A fistula is not always possible to make depending on size and health of the vasculature. The second method is a venous catheter. This is a temporary fix where a tube is inserted into one of the body’s larger veins, either the neck or leg near the groin. These devices often clog, become infected, or cause the veins in which they are placed to become narrow. The final method is an arteriovenous graft, which is similar to a fistula except that it uses some sort of tubing to indirectly connect an artery to a vein. This artificial vein can be punctured with a needle repeatedly for hemodialysis treatment. These grafts require less preparation time before use than the fistulas, but are more prone to infection and clogging and do not last quite as long [3].
Vascular grafts are also commonly used for coronary artery bypass in patients with cardiovascular disease when the patient has no suitable counterparts to take elsewhere from their body due to either disease or having previously used other vasculature. The most widely harvested vasculature for a coronary artery bypass graft (CABG) is taken from the greater saphenous vein in the leg [4]. CABG is usually used to increase blood flow due to coronary artery disease (CAD) and is competitive with percutaneous coronary intervention (PCI), a form of angioplasty where a balloon is inflated to correct for vasculature that has decreased in size. Studies have shown that CABG can provide a lower rate of repeat treatments, improved quality of life, and survival benefits in comparison to PCI [5]. Like any vascular graft, CABG can lead to complications such as thrombosis, compliance mismatch, and ruptures.
History
-1906 - Popliteal vein used to correct popliteal artery aneurysm, considered first graft [7]
-1940 – Dr. Willem Kolff filled sausage casings with blood and incubated in a saltwater bath as the first form of dialysis for kidney failure [8]
-1943 – Dr. Willem Kolff invents first dialysis machine [8]
-1949 - First femoropopliteal bypass performed through saphenous vein [7]
-1952 - Vinyon-N cloth used as first documented synthetic material graft [7]
-1957 - Dacron, material first used as commercially available vascular graft [7]
-1960 – Scribner Shunt used to connect arteries to veins to allow for ease of connection to dialysis machines [6]
-1970 – Artegraft’s vascular graft approved by the FDA [14]. PTFE grafts become available [7]
-1986 –Weinberg and Bell produced cell seeded collagen tubes trying to create novel blood vessels in vivo [9]
-2001 - First autologous cell-seeded construct produced in vitro was successfully used in a clinical trial [10]
Current Procedures
Current tissue engineered vascular grafts are made patient specific and can take 6-9 months to make. These grafts are formed by seeding autologous bone marrow cells onto a polymer or by expanding fibroblasts and endothelial cells in culture. These cell sheets are then rolled around stainless steel into a tube structure of varying diameters. The inner cell layer of the tube structure is dehydrated and seeded with more endothelial cells from the patient. This procedure can cost upwards of $15,000 so alternative methods are being explored [4].
Currently, when autologous grafts cannot be utilized, other grafts can come from animals (xenografts), cadavers (allografts), or made from synthetic materials, such as tubes of polytetrafluoroethylene (PTFE). The xenografts and allografts are susceptible to rupturing (aneurism), hardening (calcification), and blockage (thrombosis), which can result in harm to the patient. The biomaterials are also prone to thrombosis and changing size of the opening (both increasing and decreasing) due to blood flow pressure and growth on the inside of the tube, respectively [4].
Artificial grafts require the ability to sustain and withstand blood flow without leakage or clogging. This requires certain mechanical properties, which are different depending on the procedure as can be seen in Table 1. Discrepancies between values are due to differences in analysis of data. Materials can be synthesized to mimic these mechanical properties. Varying concentrations of polymers can allow for the same material to be more or less elastic, stiff, brittle, hard, porous, etc. These properties are also important when considering cell penetration, growth, and degredation depending on the purpose of the graft [1].
Market
The most common groups receiving treatment in the form of vacular grafts are the elderly and diabetics. Obesity, commonly occuring in diabetic patients, leads to damaged or restricted vasculature and can require grafting. Elderly patients are those most likely to have kidney malfunctions, and thus need grafts for hemodialysis, coronary diseases, and damaged vasculature from age. Though these groups are the most common, studies have shown that age, sex, diabetic status, and body mass index are not factors contributing to the success of the graft [11].
In the United States, a CABG surgery costs approximately $20,700 [12]. In the United States, the cost of a vascular graft for hemodialysis during year 1 of treatment is approximately $11,700 [13]. For hemodialysis, while as few at 29% of graphs failed in the first few months, as many as 96% of grafts fail by the end of 2 years [11].
There are many companies that utilize different styles of vascular grafts. Some notable companies are: Artegraft – first company to have a vascular graft approved by the FDA (1970) [14]
Cytograft – tissue engineered blood vessels from autologous fibroblast sheets rolled into tubes (LifeLine) [15]
Bionova - biosynthetic vascular patch goes around the vascular wound (Omniflow II) [16]
Latest Research
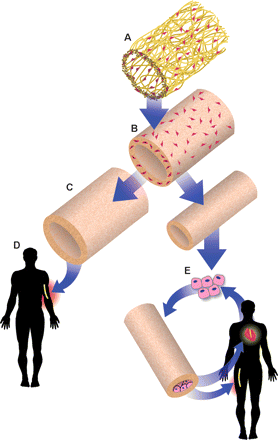
Since research began in artificial vasculature, there has seemingly been a common desirable outcome: a cost effective, efficient, and successful partially synthetic material based and partially cell or tissue based construct with the necessary mechanical properties, but also the necessary biocompatability properties. There are many ways to approach and remodel this product, everything from obtaining a structure from a living being (whether it be an auto-, allo-, or xeno-graft) to an electrospun carpet of polymers and cells. These grafts have been designed to degrade or maintain the structure in vivo and are generally applicable or tailored to specific treatments. Descriptions of cutting edge research can be found below:
Biodegradable scaffolds seeded with bone marrow mononuclear cells form into new blood vessels in about 6 months in mice via an inflammatory response. This starts with the seeded bone marrow cells releasing a chemoattractant to initiate the migration of monocytes to the scaffold. These monocytes release cytokines that attract endothelial and smooth muscle cells that completely replace the degraded scaffold [17].
Polyglycolic acid (PGA) tubular scaffolds seeded with smooth muscle cells from a cadaver. The PGA scaffold degrades and is replaced by native extracellular matrix (ECM) proteins, such as collagen I and III, fibronectin, and vitronectin. The cells are exposed to continual, cyclic radial strain in a bioreactor. When the scaffold has degraded significantly, cells are removed from the secreted ECM structure so that it is non-immunogenic. These vascular grafts can be made from any bank of cells and can be used in any person. These engineered grafts can also be stored in phosphate buffers saline (PBS) at 4 degrees celsuis for extended periods of time. These grafts can be made at the same diameter 6-7 mm and smaller diameters 3-4 mm than can be made from rolling up cell sheets. In this study, the smaller diameter vessels were seeded with additional endothelial cells to help prevent thrombosis, though further research is needed to prove if this additional seeding is actually necessary. These novel vasculature can also withstand the necessary forces generated in the human body [4].
Effects of surface proteins were assessed to determine optimal binding of endothelial cells to synthetic structures. This was meant to increase bioactivity of the grafts as well as biocompatability. Electrospinning was utilized for increased diffusive properties of materials and showed significant modulation of endothelial cell behavior when functionalized with fibrinogen [18].
References
[1] Hasan, Anwarul, Adnan Memic, Nasim Annabi, Monowar Hossain, Arghya Paul, Mehmet R. Dokmeci, Fariba Dehghani, and Ali Khademhosseini. "Electrospun Scaffolds for Tissue Engineering of Vascular Grafts." Acta Biomaterialia 10.1 (2014): 11-25. Print0.
[2] McAllister, Todd. "First Human Use of an Allogeneic Tissue Engineered Vascular Graft." American Heart Association, 27 June 2011. Web. 13 Mar. 2012. <http://my.americanheart.org/idc/groups/ahamah-public/@wcm/@sop/@scon/documents/downloadable/ucm_428738.pdf>.
[3] "Vascular Access for Hemodialysis." National Kidney & Urologic DiseasesInformation Clearinghouse (NKUDIC). US Department of Health and Human Services. Web. 13 Mar. 2012. <http://kidney.niddk.nih.gov/kudiseases/pubs/vascularaccess/>.
[4] Dahl, Shannon L. "Readily Available Tissue-Engineered Vascular Grafts." Science Translational Medicine 3.68 (2011). PubMed. Web. 13 Mar. 2012. <http://stm.sciencemag.org.silk.library.umass.edu/content/3/68/68ra9.full>.
[5] Hemmelgarn, B.r., D. Southern, and B.f. Culleton. "Survival after Coronary Revascularization among Patients with Kidney Disease." ACC Current Journal Review 14.2 (2005): 48. Print.
[6] "Pioneers in Kidney Dialysis: From the Scribner Shunt and the Mini-II to the "One-Button Machine"" Pathbreakers. University of Washington, 1996. Web. 13 Mar. 2012. <http://www.washington.edu/research/pathbreakers/1960c.html>.
[7] "Tissue Engineered Vascular Grafts." Nautica2.2 Liquid. Drexel University, n.d. Web. 11 Mar. 2014.
[8] Blakeslee, Sandra. "Willem Kolff, Doctor Who Invented Kidney and Heart Machines, Dies at 97." The New York Times, 12 Feb. 2009. Web. 13 Mar. 2012. <http://www.nytimes.com/2009/02/13/health/13kolff.html?pagewanted=all>.
[9] L'Heureux, Nicholas. "Technology Insight: The Evolution of Tissue-Engineered Vascular Grafts: History of Tissue-engineered Vessels." Nat Clin Pract Cardiovasc Med 4.7 (2007): 389-95. Medscape. Web. 13 Mar. 2012. <http://www.medscape.com/viewarticle/559495_2>.
[10] L'heureux, Nicolas, Nathalie Dusserre, Alicia Marini, Sergio Garrido, Luis De La Fuente, and Todd Mcallister. "Technology Insight: The Evolution of Tissue-engineered Vascular Grafts—from Research to Clinical Practice." Nature Clinical Practice Cardiovascular Medicine 4.7 (2007): 389-95. Print.
[11] Miller, Paul E., Donna Carlton, Mark H. Deierhoi, David T. Redden, and Michael Allon. "Natural History of Arteriovenous Grafts in Hemodialysis Patients." American Journal of Kidney Diseases 36.1 (2000): 68-74. Print.
[12] Eisenberg, M. J. "Outcomes and Cost of Coronary Artery Bypass Graft Surgery in the United States and Canada." Archives of Internal Medicine 165.13 (2005): 1506-513. Print.
[13] Manns, B. "Establishment and Maintenance of Vascular Access in Incident Hemodialysis Patients: A Prospective Cost Analysis." Journal of the American Society of Nephrology 16.1 (2004): 201-09. Print.
[14] "History." Bovine Carotid Artery Graft. Artegraft, 2012. Web. 13 Mar. 2012. <http://www.artegraft.com/About-Us/History>.
[15] "Tissue Engineered Blood Vessel (TEBV)." Cytograft. 2009. Web. 13 Mar. 2012. <http://www.cytograft.com/tebv.html>.
[16] "Omniflow II for Vascular Repair." Bionova. Storm Worldwide, 2011. Web. 13 Mar. 2012. <http://www.bionova.com.au/bionova-products/714-2/>.
[17] Roh, Jason D. “Tissue-Engineered Vascular Grafts Transform into mature Blood Vessels via an Inflammation-mediated Process of Vascular Remodeling.” PubMed Central. 9 Mar. 2010. <http://www-ncbi-nlm-nih-gov.silk.library.umass.edu/pmc/articles/PMC2842056/?tool=pubmed>.
[18] Ahmed, F., NK Dutta, A. Zannettino, K. Vandyke, and NR Choudhury. "Engineering Interaction between Bone Marrow Derived Endothelial Cells and Electrospun Surfaces for Artificial Vascular Graft Applications." Biomacromolecules (2014): n. pag. ACS Publications. Web. 11 Mar. 2014.
