User:Emilie M. Ryan-Castillo/Notebook/Biology 210 at AU
16S Sequence Lab - February 21, 2015
Purpose
Bacteria are diverse and characteristics alone are not sufficient enough to identify species. By looking at the 16S gene sequence you can identify which species it is.
Methods and Materials
Using a sterile spatula scrape off part of the bacteria colony and place it into a a sterile tube of 100 nanometers of water. Then incubate the tube at 100 degrees Celsius for 10 minters. Then we placed the tube in a centrifuge for 5 minutes at 13,400 rpm. This is repeated for another colony of bacteria. While tubes are in the centrifuge we added 20 nano liters of primer water to PCR tube and transferred 5 nanometer of supernatant from the centrifuged samples and placed in PCR machine. Later the samples are analyzed and run through BLAST and the species was identified.
Results
| Colony | Sequence | BLAST Result |
| A | >MB46-For_16S_F06.ab1 NNNNNNNNNNNNNNNNNNNANNNTGCAGTCGNNCGGGNGTAGCNCGNTANNGCTCTCTNATTCGCGGCGGACGGGTGAGT AATGCCTANGAATCTGCCTGGTANTGGGGGACAACGTCTCCAAAGGGACGCTAATACCGCAAACGTCCTACGGGAGAAAG CGGGGGACCTTCGGGCCTTGCGCTATCGNATGANCCTAGGTCGGATTANCTAGTTGGTGAGGTAATGGCTCACCAAGGCC ACGATCCGTAACTGGTCTGAGAGGATGATCATTCACACTGGAACTGAGACACGGTCCNGACTCCTACGGGAGGCAGCAGT GGGGAATATTGGACAATGGGCGAAAGCCTGATCCAGCCATGCCGCGTGTGTGAAGAAGGTCTTCGGATTGTAAAGCACTT TAAGTTGGGAGGAAGGGCAGTAACTTAATACGTTGCTGTTTTGACGTTACCGACAGAATAAGCACCGGCTAACTCTGTGC CAGCAGCCGCGGTAATACAGAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGCGCGTANGTGGTTTGTTAANT TGNATGTGAAANCCCCGGGCTCAACCTGGGAACTGCATTCNNAACTGACAAGCTANAGTATGGTANAGGGTGGTGNAATT TCCTGTGTAGCNGTGAAATGCNNNNNATNNGAAGGAACACCNGTGNCNAAGGCGACCACCTGNACTGATACTNACACTGA NNTGCGAAAGCGTGGGGANCNACANGATTANATNNCCTGNTACTCCACNNCNNTANACNATNTNNNCTANCCNTTTNGAN CNCTTGAGCTGNNNANTGNCGCANCTCACNCATTAANTTTNACCNNCCTNNNNTAGTACNGGCNCNNCGNTANGNNNTCN NNNGGANTNNNNNNNNCNGNCNNAANCANNNGANCTGGTGGTTTNNTTCNNNNNNANANNNANAACNNTACNNNNCCCNN NGNNTNNNNNNNCNNNNNNNNNNNNNNTTNNGGCGNNNNNNNNNNNNNNNNGNNNCNNNANNNNNGNNNNNNNNNNNNTN NNNNNNNNTGNANNNNNNNNNNNNNNNNNNNNANNNNNNN | 93% match for both Pseudomonas mohnii and Pseudomonas putida |
| B | >MB45-For_16S_E06.ab1 NNNNNNNNNNGNNNNCTTNNNNNTGCAGTCGTACGNGTNGNCAGNGCNNNTNCNGATGCTGACGAGTGGCGAACGGGTGA GTAATACATCGGAACGTGCCTAGTAGTGGGGGATAACTACTCGAAAGAGTAGCTAATACCGCATGAGATCTACGGATGAA AGCAGGGGACCTTCGGGCCTTGTGCTACTAGAGCGGCTGATGGCAGATTAGGTAGTTGGTGGGGTAAAGGCTTACCAAGC CTGCGATCTGTAGCTGGTCTGAGAGGACGACCAGCCACACTGGGACTGAGACACGGCCCAGACTCCTACGGGAGGCAGCA GTGGGGAATTTTGGACAATGGGCGAAAGCCTGATCCAGCAATGCCGCGTGCAGGATGAAGGCCCTCGGGTTGTAAACTGC TTTTGTACGGAACGAAAAGCCTGGGGCTAATATCCCCGGGTCATGACGGTACCGTAAGAATAAGCACCGGCTAACTACGT GCCAGCAGCCGCGGTAATACGTAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGTGCGCAGGCGGTTTTGTAA GACAGTGGTGAAATCCCCGGGCTCAACCTGGGAACTGCCATTGTGACTGCANGGCTANAGTGCGGCAGAGAGNGGATGGA ATTCCGCGTGTANCANTGANATGCNTANATATGCGGAGNANCACCGATGGGCGAANNNNGTCCCCTGGGCCTGCACTGNN NCTCATGCACNNAAGCGTGGGGAGCAANANGANTNNNANNCTGGGTAGTCCACNCCCTNNCGATGTCAACTGNTTGTTGG NTNNNNNGTNANTCNNTAANNANNTANNNNTGAAGTTNACNNCNNNGGAGTACGNCCGGGNNGTTAGNANGNANNNNNNG GGNNTTGACCNCNCACCNCACTAGAGGNGGNGGTTGGGTTNNNNNNNNNNANNANNATAANCCCCCTNTCNCTTNNNANN ANNNNNGANNANANNGATAGNTGGTNGGGGGGNANNANNCNAACCGGNCCGCGGGGNCGGNGGGGACATCCCCCCCNNGG GTGNNTGNNACAAGNNNNGAAANCNCCNCANCCGNNGGGGCCNNNNTNGCNGNNGNNNNNNN | 95% match for Comamonas testosteroni |
Conclusion The sequencing of the bacteria in our colonies refuted the findings made in the Gram Stain procedure we did earlier in the semester. Identifying bacteria without the use of DNA sequencing is difficult because most bacteria look the same but are not the same species.
4.7.15 Discuss the sequence results and how they compare with your own gram stain etc. Include the gel image. SK
Effects of 2% Salinity on Embryology & Zebrafish Development - February 19, 2015
Purpose
The purpose of the lab is to understand embryonic development and stages and compare them with other organisms embryonic development. As well as to understand how environmental conditions can impact on embryonic development.
Materials and Methods
First we compared embryonic stages of different organisms like chicks, frogs, starfish, and humans. Then after doing research on the effects of salinity on zebrafish embryos we developed an experiment and hypothesis on how to test the effects of salinity. Two conditions were created for the experiment. One petri dish contained 20 mL of deer park water with about 20 Zebra fish embryos which served as a control group. Another petri dish contained 20 mL of a two percent salinity solution and also contain about 20 embryos. The embryos were observed and their embryonic stages determined. The Zebra fish embryos will be observed over the two week period and stages and life count will be recorded/
Results
In the beginning of the experiment there were 22 live eggs in the control with nine in the 2-somite stage, nine in the 4-somite stage, two in the 8-somite stage, and 2 were budding. There were 20 live eggs in the 2% salinity solution with six in the 2-somite stage, four in the 4-somite, and six in the 8-somite stage.
On day four there there were seven dead eggs, thirteen live eggs, and three live hatchlings in the control petri dish with four the the embryos as hatchlings and twelve 8-somite eggs. In the 2% salt solution there were six dead eggs and fifteen live eggs.
On day seven there were there were
| Day One | # of dead eggs | # of live eggs | # of live hatchings | # of dead hatchlings | Stages of Development |
| Control | 0 | 22 | 0 | 0 | 2-somite: 9 4-Somite: 9 8-somite: 2 Budding: 2 |
| 2% | 0 | 20 | 0 | 0 | 2-somite: 6 4-somite: 4 8-somite: 6 |
| Day Four | |||||
| Control | 7 | 13 | 3 | 0 | Hatchlings 48 Hrs: 4 8-somite: 9 |
| 2% | 6 | 15 | 0 | 0 | 8-somite: 8 4-somite: 7 |
| Day Seven | |||||
| Control | 12 | 2 | 4 | 0 | Hatchlings 72 Hrs |
| 2% | 15 | 0 | 0 | 0 |
Conclusion
Organisms have similar development during their embryonic stages as they become more developed their similarities become few. In the salinity experiment the two percent condition seems to be slowing down the development of the eggs since none have hatched but still seem to be alive. Compared to the control which already has three hatchlings.
2.20.15 Good entry. Could include more text description of methods and describing invertebrates found. Good food web. SK
Invertebrates - Feburary 12, 2015
Purpose
The purpose of this lab was to identify different invertebrates in our transects and learn their importance. It was also to understand how simple systems evolved into more complex systems.
Materials and Methods
Procedure I: Acoelomates, Pseudocoelomates, and Coelomates
First we observed three types of invertebrates (Planaria, nematodes, and Annelida) prepared in a petri dish. Their movement and digestive system was observed and noted it and related it to their body structure.
Procedure II: Anthropods
Observed examples of anthropoids and their characteristics and how they different in body pars, segments, and appendages as well as how they are classified.
Procedure III: Analyzing the Invertebrates Collected with the Berlese Funnel
We took down the Berlese Funnel and poured out the ethanol that was attached to the Berlese Funnel into a petri dish. Spliting the organisms and excess ethanol into two different petri dishes. Then examined the organisms under a microscope and try to identify them with a dichotomous key. We Identified at least three invertebrates and measured the length of each one. We then noted the most common type of invertebrate and the smallest and largest.
Procedure IV: Vertebrates and Niches
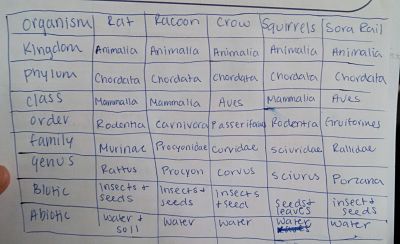
Then we observed our transects and identified vertebrates that inhabit or pass through our transect. We note this by observation or evidence by fecal matter or footprints. Then determine their classification and list biotic and abiotic characteristics of the transect that would benefit each vertebrate species.
Data and Observations
Procedure I: Acoelomates, Pseudocoelomates, and Coelomates
All three of the invertebrates moved in a slithering and back in forth motion. They are capable of doing this with the lack of a spinal cord.
Procedure III: Analyzing the Invertebrates Collected with the Berlese Funnel
Here is a table of observation from analyzing in vertebrates in our transect.
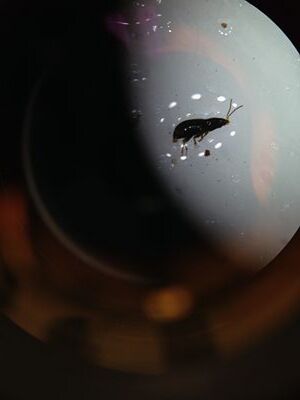
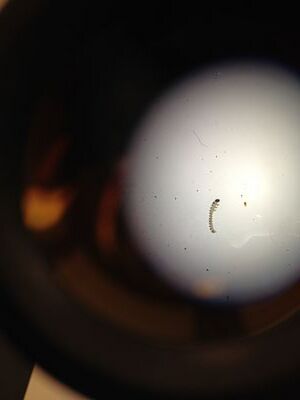
Here is an image of the first sample of invertebrate.
Here is an image of the second sample of invertebrate.
Procedure IV: Vertebrates
The table listed below are some vertebrates that have been seen near or in our transect.
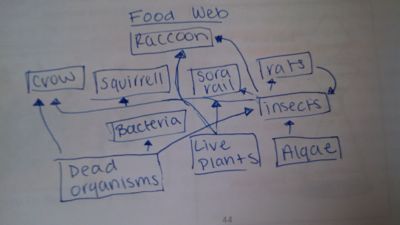
The diagram below depicts the food web for our transect.
Conclusion
There are a wide variety of invertebrates in our transect each varying in characteristics and the biodiversity within our transects. Many of the invertebrates that we discovered in our transects were very small insects but were were still able to identify them from their distinct characteristics from a online key. While identifying vertebrates and designing a food web would have helped us understand the different level of systems that are going on in the transect. Although no vertebrates were actually observed in our transect.
2.20.15 Very good entry. Well organized. SK
Plantae and Fungi - February 4, 2015
Purpose
The purpose of this lab was to obtain an understanding of the variety of characteristics and diversity of plants especial with the means and function of reproduction. As well as to acknowledge the function and importance of fungi.
Materials and Methods
Procedure I: Collecting five plant samples from the transect
We obtained three ziplock bags and collected five different samples of plants from our transect and note location and height. In one of the ziplock bags collect a combination of soil and leaves from the ground of transect.
Procedure II: Plant Vascularization
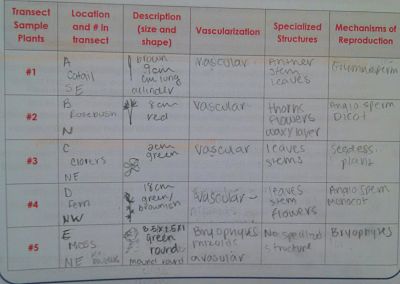
We compared and contrasedt plant samples found in transect vascularization and height and note in table. We looked for xylem and phloem layers for angiosperms and as well as rhizoids for bryophytes.
Procedure III: Presence of Specialized Structures
Observe each sample under low magnification and note presence of specialized structures such as cuticles, stomata's, and guard cells. Describe the shape, size, and cluster arrangement of the leaves from the transect plants.
Procedure IV: Mechanisms of Plant Reproduction
Compare and contrast plant samples reproductive cycles. Note if they are gametophyte or sporophyte and identify the seeds as either monocot or dicot. Note if there id any evidence of flowers or spores.
Procedure V: Observing Fungi
Observe black bread mold. Note importance of fungi sporangia and decide what group the samples belong in and draw a picture in notebook.
Procedure VI: Setting up the Berlese Funnel to Collect Invertebrates
Obtain 50 mL conical tube, funnel, tape, screen, and ethanol water mix. Put 25 mL of ethanol water solution into the 50 mL conical tube. Then tape screen to bottom of the funnel. Then place funnel into the conical tube and tape into place and attached to stand. Once contraption is set up add soil and leaves collected from transect into funnel and place a 40 watt lamp above funnel and leave for a week.
Data and Observation
Data for all procedures in table below.
Here is an image for sample number one. Media:IMG Plant1.JPG
Here is an image for sample number two Media:IMG Plant2.JPG
Here is an Image for sample number three Media:IMG Plant3.JPG
Here is an image for sample number four Media:IMG Plant4.JPG
Here is an image for sample number five Media:IMG Plant5.JPG
Here is an image of the fungi examined in Procedure V Media:IMG Mushroom.JPG
Conclusion
Majority of the plants that we took from our transects were shorter in height but varied in species. Short statued plants are common in marshes. After this lab we were better able to understand the relationship of different environments and characteristics that plants have and how to identify them from those characteristics. There were different ways that plants reproduce and we were able to identify those among the samples we obtain from the transect. The fungi that we observed was a mushroom and was a basidiomycota which have sexual basidiospore which produce under the cap. It was difficult to characterized some of the plants means of reproduction because most of them were dead due to the fact that it is winter.
2.10.15 Good start. The purpose section is poorly written and doesn't really make sense. All relevant data is present but conclusions were a little brief. SK
Microbiology and Identifying Bacteria with DNA Sequences - January 29, 2015
Purpose
The purpose of this lab is to understand the variety of characteristics that bacteria have and observe how bacteria antibiotic resistance. The purpose is also to understand how and why DNA sequences are used to identify species.
Materials and Methods
Procedure I: Quantifying and Observing Microorganisms
I this procedure we observed the agar plates that we set up in last weeks lab from the Hay Infusion Culture. Then we recorded the number of different colonies.
Procedure II: Antibiotic Resistance
In this procedure we observed the differences in the colonies in the agar plates with tetracycline and the agar plates without tetracycline. We noted the differences in relations to how many colonies, species, and presence of fungi.
Procedure II: Bacteria Cell Morphology Observation
In this procedure we prepared wet mounts with different bacteria from different colonies from two of the agar plates with tetracycline and two of the agar plates without tetracycline. One of the set of wet mounts is used to be observed the other set is used for the gram stain.
A) Met Mount Procedure: First sterilized a loop with a flame and scooped up a little of the growth from the surface of the agar plate. Mix the loop into a little water on the slide and place a cover slip. Observed first and 10x and 40x. Then observed at 100x oil immersion.
B) Gram Stain Procedure: First sterilize loop with a flame and scoop up a small amount of the growth from the surface of the agar plate. Mix the loop into a little water on the slide. Then heat fix the slide by placing it through the flame three times, drying the water up. Then pour over the bacterial smear Gram's Iodine mordant for one minute and rinse it off with water. Then pour 95% alcohol for 10-20 seconds over the bacterial smear. Then soak bacterial smear with safrain stain for 20-30 seconds and then rinse with water. Then carefully dry the slide and observe slides first under 40x then at 100x oil immersion.
Procedure IV: Set up PCR for 16S sequencing
Select one from each of the two plates of agar and two plates of tetracycline to sample for the PCR and make sure they are labeled. First we transfered a colony of bacteria to 100 uL of water in a sterile tube. Then we incubated the tube at 100 degrees Celsius for 10 minutes. Then we placed the tubes in the centrifuge for 5 minutes at 13,400 rpm. Then we placed 20 uL of primer water to PCR tube. Then transfer 5 uL of supernatant from the centrifuged samples to the 16S PCR reaction and then placed it in the PCR machine.
Data and Observations
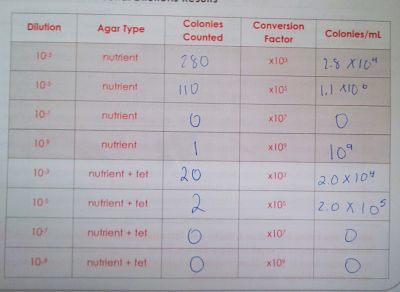
Procedure I: Qualifying and Observing Microorganisms
Procedure II: Antibiotic Resistance
The agar plate with tetracycline had fewer number of colonies and they tended to be larger in size. There seemed to be more fungi in the the agar plates with tetracycline.
The agar plates without tetracycline had more colonies and they tended to be smaller in size. There were fewer fungi colonies in the agar plates without tetracycline than those with tetracycline.
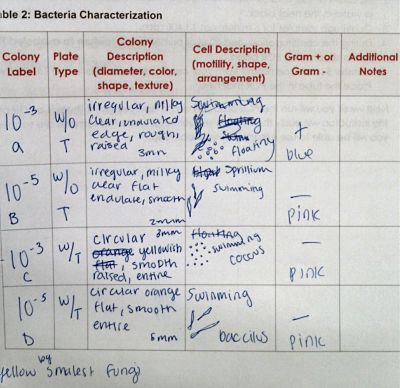
Procedure III: Bacteria Cell Morphology Obsercations
Gram Stain Bacteria sample A from agar plate without tetracycline
Gram Stain Bacteria sample B from agar plate without tetracycline
Gram Stain Bacteria sample C from agar plate with tetracycline
Gram Stain Bacteria sample D from agar plate with tetracycline
Conclusion
There are varying amounts of bacteria living in our transects some of them even exhibit antibiotic resistance based on the fact that some colonies of bacteria still grew on the agar plates that contained the the tetracycline antibiotic.
2.4.15 Good notebook entry. Could include some more detailed descriptions of identified protists. Photo is a bit large, try saving image as a smaller file size and then uploading. SK
Indentifying Algae and Protists - January 22, 2015
Purpose
The purpose of this lab was to practice identifying algae and protists using a dichotomous key. Understand characters of algae and protist and therefore help us identify them in our transects.
Material and Methods
Procedure I: Using a Dichotomous Key First we made wet mounts of a known organisms and used the Dichotomous key to identify the organism. We observed it with the microscope at 4x and 10x and recorded its size.
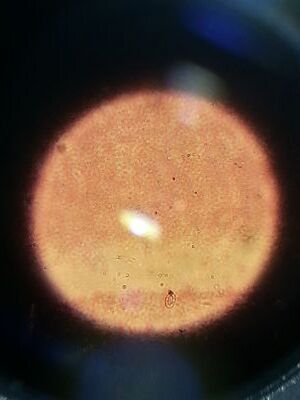
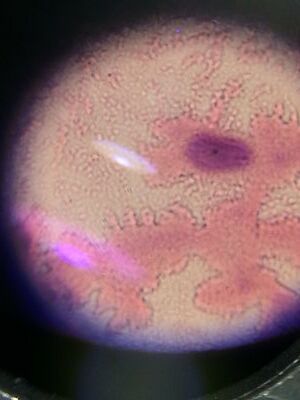
Procedure II: Hay Infusion Culture Observations We observed the Hay Infusion Cultures that we had in a past lab. We noted the smell and it's appearance. Then we took samples from different areas of the Hay Infusion Culture and made a wet mount and observed it under a microscope. We then used the Dichotomous Key to try to identify the organisms in the sample of Hay Infusion Culture.
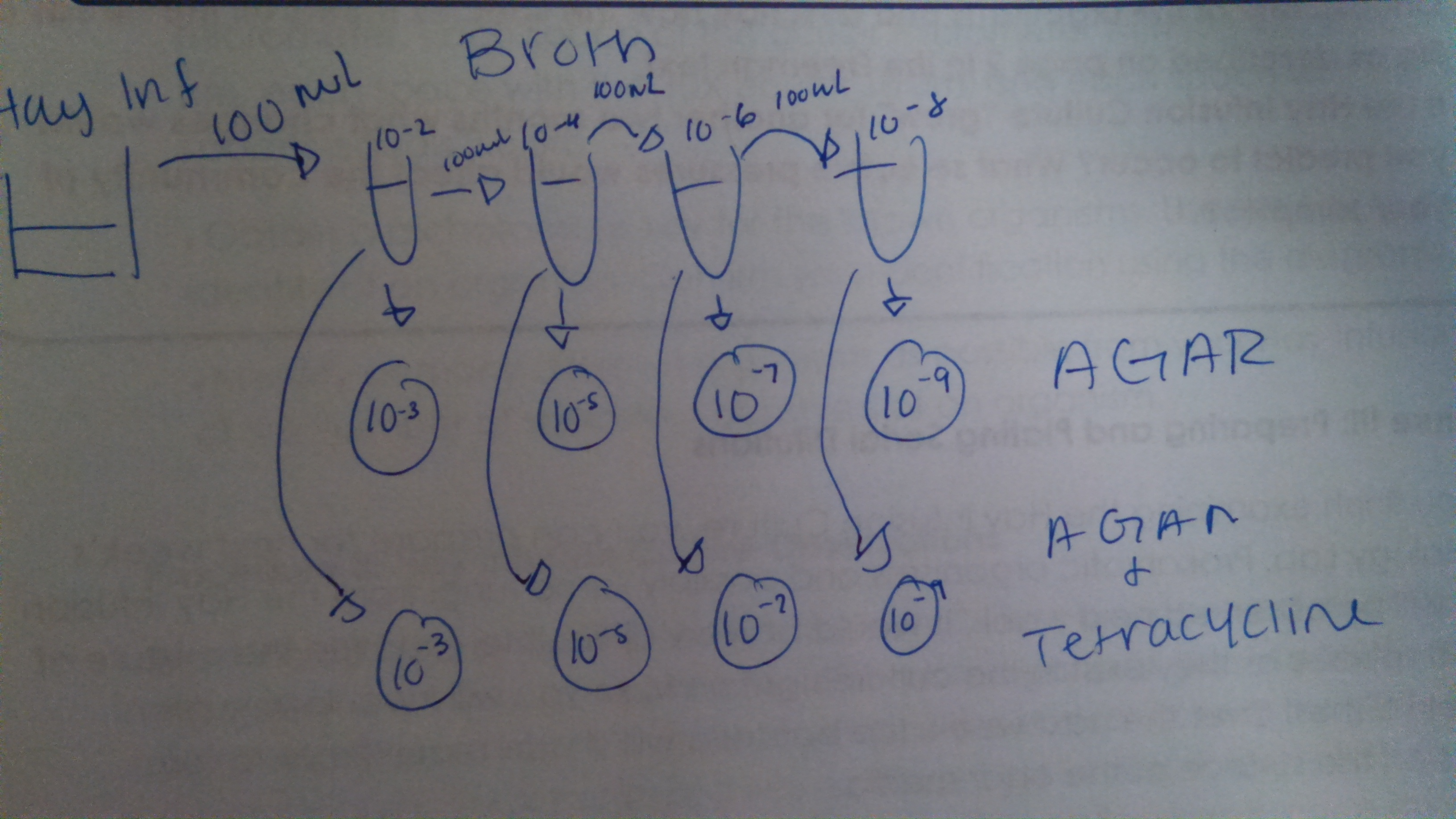
Procedure II: Preparing and Plating Serial Dilutions
We obtained four tubes of 10 mL sterile broth with concentrations of 10^-2, 10^-4, 10^-6, 10^-8 and a micropippetor at 100 mL and tops. Then we obtain four nutrient agar and four nutrient agar plus tetracycline plates and labeled them. Then we swirled the Hay Infusion Culture to mix up all organisms. Then took 100 uL from the culture and added to the 10mL of broth that was labeled 10^-2 and swirled it thoroughly. Then we took of 100 uL of broth from the 10^-2 dilution and put in it the 10^-4 tube. We repeated these steps to make the 10^-6 and 10^-8. Then we took at 2 sample of 100 uL of each tube and place and placed one in a nutrient agar and a nutrient agar plus tetracycline plates and carefully spread the sample on them. Then we placed the plates in a rack and incubated them at room temperature.
Data and Observation
Procedure I: Using the Dichotomous Key
The known organism we identified was Paramecium Blepharisma. It was clear and ovalish shaped and around 100 uL. It had cilia.
Procedure II: Hay Infusiont Culture Observations
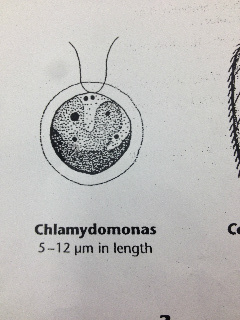
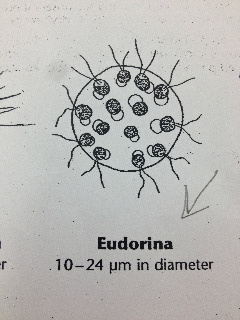
Our Hay Infusion culture was a deep brown liquid with lots of soil sitting at the bottom. It smelled very bad and had a film of some sort sitting at the top of the water. There were multiple leaves floating and sitting at the bottom of our Hay Infusion Culture. Some organisms that we identified from the sample we took from our Hay Infusion Culture Bursaria truncotella at 700 um, Chlamydomonas at 7.5 um, colpidium at 200 um, and eudorina at 25 um.
Conclusion
Although the Dichotomous Key was very helpful identifying organisms based on their appearance is difficult especially with organisms with this size. The variety of organisms that can live in our transect we endless and their innate ability to survive in those conditions based on differing characteristics.
1.27.15 Good first entry. Some missing detail eg. Hay Infusion set-up. SK
Biological Life at AU - January 15, 2015
Purpose:
Observe characteristics of niches within a transect at American University. Understand the diversity and complexity of ecosystems and biotic and abiotic features within them.
Materials and Methods:
A transect of land of 20 by 20 meters within American University grounds was given to each group of students. The transect observed was the marsh land or transect one. Transect will be observed over a series of weeks. Many different factors would be observed such as weather, amount of water, pollution, wildlife, and abiotic features. A collection of 50 mL of soil and ground vegetation was taken in order to make a Hay Infusion Culture.
Data and Observations:
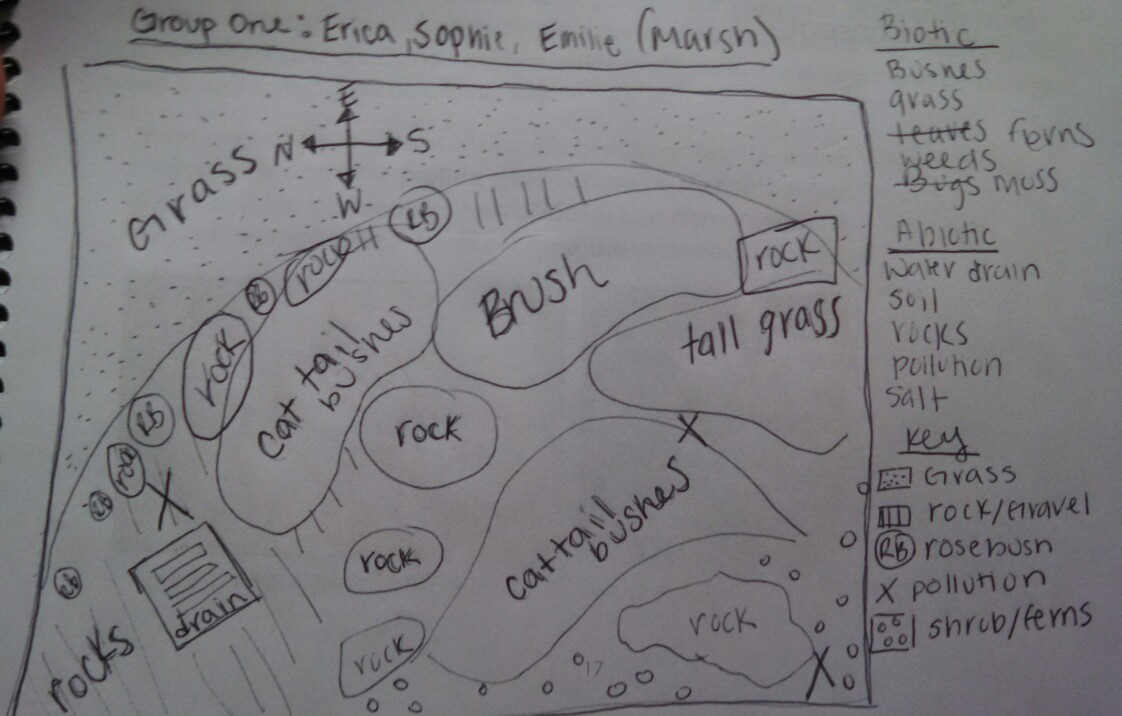
Transect one assigned to my group was the marsh lands. The marsh lands were located near the Massachusetts entrance to American University across from Kogod School of Business.
Our transect was very diverse and many different types of abiotic and biotic features. Some biotic features it contained were cattail bushes, grass, ferns, moss, and weeds. Some abiotic features transect one contain were soil, boulders, pollution, salt, and a water drain.
Conclusion:
After a few weeks of observation you an tell that although the marsh transect seemed like a simple man made landscape it contain niches. In just 20 by 20 meters there was a lot diversity within in it.