User:Caroline Vill/Notebook/Biology 210 at AU
Bacteria from Transect
5 March 2015
Purpose: The purpose of this lab was to identify different bacterial species in transect 5 (manicured grassland) based on the amplification of the 16S ribosomal RNA gene.
Materials and Methods: Procedure I: A single colony of bacteria was transferred to a sterile tube filled with 100μL of water. The tube was incubated at 100°C for 10 minutes in a heat block. The sample was then centrifuged for 5 minutes at 13,400 rpm. While the sample was in the centrifuge, 20μL of primer/water mixture were added to a labeled PCR tube. The tube was mixed gently to dissolve the PCR bead. Finally, 5μL of supernatant from the centrifuged sample was added to the 16S PCR reaction and the tube was placed in the PCR machine. This was repeated for 3 other samples.
Procedure II: The following week, the PCR products were ran out on an agarose gel. Ladder was placed in lane 1, sample 1 (nutrient + tet (10^-3)) was placed in lane 2, sample 2 (nutrient + tet (10^-5)) was placed in lane 3, sample 3 (nutrient (10^-5)) was placed in lane 4, and sample 4 (nutrient (10^-5)) was placed in lane 5. The two best samples were then selected, sent off to a lab, and sequenced to identify the bacteria species.
Procedure III: The next week, the results came in through GeneWiz and Genomic Blast (a sequencing software by the National Library of Medicine) was used to identify the species.
Data and Observations:
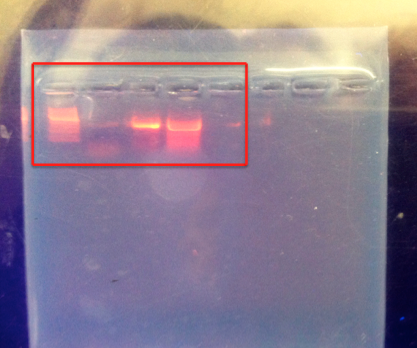
Figure 1 shows the agarose gel the PCR products were run out on. The red box represents the 5 lanes that product was put into. Lane 1 (the furthest to the left) contains ladder. It shows a bright band at 1300 base pairs (bp). Lane 2 is very faint. Lane 3 and 4 are very bright and display a bright band at about 1300 bp. Lane 4 did not amplify.
Raw Sequence of Species 1*: TGNANGCCNANCGNGTNAGANGANCGNNNTNCTGNGGNANNCTNTGNGNNAGCGNGNTG ATACGGGTGCGGAACACGTGTGCAACCTGCCTTTATCAGGGGGATAGCCTTTCGAAAGGAAG ATTAATACCCCATAATATATTGAATGGCATCATTTGATATTGAAAACTCCGGTGGATAGAGATGGG CACGCGCAAGATTAGATAGTTGGTAGGGTAACGGCCTACCAAGTCAGTGATCTTTAGGGGGCC TGAGAGGGTGATCCCCCACACTGGTACTGAGACACGGACCAGACTCCTACGGGAGGCAGCA GTGAGGAATATTGGACAATGGGTGAGAGCCTGATCCAGCCATCCCGCGTGAAGGACGACGGC CCTATGGGTTGTAAACTTCTTTTGTATAGGGATAAACCTTTCCACGTGTGGAAAGCTGAAGGTAC TATACGAATAAGCACCGGCTAACTCCGTGCCAGCAGCCGCGGTAATACGGAGGGTGCAAGCGT TATCCGGATTTATTGGGTTTAAAGGGTCCGTAGGCGGATCTGTAAGTCAGTGGTGAAATCTCATA GCTTAACTATGAAACTGCCATTGATACTGCAGGTCTTGAGTAAAGTANAAGTGGCTGGAATAANT AGTGTANCGGTGAAATGCATAGATATTACTTANNAACACCAATTGCGAAGGGCAGGTCNCTATGT TTTAACTGACGCTGATGGACGAAAGCGTGGGGAGCGAACAGGATTANATACCCTGGTNGTNNN NGCCGTANACGATGCTNACTCNTTTTTNGNNCTTCNGATTCAGAGACTAAGCNAAANTGATAGT TAGNCNNCCTGGNGAGTNCNTTCNCAANAATGAAACTCANAAGAANTGACGGGGGNCCCNCN CANCCGTGNATTATGTNGTTTAATTCANNNNNCNNNNGNANCCTTNNCNACGCTTAANNGGGAT TGNGGGGGNTTAGANNNNANNNGTCTCNNCATTTCNANNTTCTNCNNGGGNNGNCGGNGGNT GGTCCCCCNNTGTANGNNNNGGTCAAGNACNNGNNGNNCCCNNT
Raw Sequence of Species 2* (poor quality): GCAGTCNAGCGGATGANANNAGCTTGCTCTTCGATTCAGCGGCGGANGGGTGAGTAATGCCTA GGAATCTGCCTATTAGTGGGGGACAACGTTTCGAAAGGAACGCTAATACCGCATACGTCCTACGG GAGAAAGCAGGGGACCTTCGGGCCTTGCGCTAATAGATGAGCCTAGGTCGGATTAGCTAGTTGG TGAGGTAATGGCTCACCAAGGCGACGATCCGTAACTGGTCTGAGAGGATGATCAGTCACACTGG AACTGAGACACGGTCCAGACTCCTACGGGAGGCAGCAGTGGGGAATATTGGACAATGGGCGAA AGCCTGATCCAGCCATGCCGCGTGTGTGAAGAAGGTCTTCGGATTGTAAAGCACTTTAAGTTGGG AGGAAGGGTTGTAGATTAATACTCTGCAATTTTGACGTTACCGACAGAATAANCACCGGCTAACTCT GTGCCANCAGCCGCGGTAATACAGAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGC GCGTAGGTGGTTCNTTAAGTTNNATGTGAAATCCCCNGGCTCNACCTGNNAGCTNCNTTCNANAC TGTCNNGCTANANTATGGTANANGGTGCTGGAATTTCCTCTGTNGNNNNNCAAATNNNNANATANA NNATTGAACNNNNNTGNCNANNNGNNNNCCTCCNCTGNTNGNCNANNCNNGTATNCGCNCCNTG NGNGNGAAACACNNGNGGNTNGNCTCCNNCGCCACNGNGTNTNCNANATNTACNANCCTTTTCG NTGNGNTNNNNNNNTATTNCNCNGNCTCNCCNNANANNTNNACNNNCANNNTNNNGNNNNGNCC NCCCTGGGGGCTCNNCNNGNNTTTTTNNNTGNCNNGNGCAAGNAANNNNNGNGGGNGTTGNNG CTNNNTNNNACAAAANNANNNNANCGCCCCNTGGTNNNGNNCNGGNNNANGGNNNNTAANANNT GGNNTGGNNNCCTNCGGGNACCNAAANNNNNGGNNGNANNGNANNNCNNNNNCTCCCNNNCCNANNNNGGTTGG
- Note: The “N”s in the raw sequences represent unidentifiable bases.
Table 1: Identifying the Bacteria
| . | Tube Number | Tube Label | Number of base pairs | Identity of Bacteria | % match? |
| Species 1 | 41 | MB41 | 1386 | Chryseobacterium sp. WR1 | 80% |
| Species 2 | 42 | MB42 | 1397 | Pseudomonas sp. Ata11 | 59% |
Discussion: The 16S rRNA gene has been an effective way to identify and classify bacteria, as well as mark their molecular evolution (Kampfer, et. al., 2003). This gene is the most diverse among bacteria and gives the most accurate method to identifying different species (Kampfer, et. al., 2003).
In figure 1, the ladder displays a bright band at about 1300 base pairs. Lane 2 shows a faint band. Lane 3, which contained sample 2 from the 10-5 nutrient plus tet plate, and lane 4, which contained sample 3 from the 10-5 nutrient plate, both amplified and displayed bright bands at 1250-1300 bp. Lane 5 (sample 4) did not amplify. Thus, samples 2 and 3 were chosen to be analyzed. The raw sequences for species 1 (sample 2) and for species 2 (sample 3) are shown above. While the sequences are not the best, considering the amount of unidentifiable bases there were, the species were could still be recognized with some certainty. Species 1 was identified as chryseobacterium with an 80% match to the known genetic code, and species 2 was identified as pseudomonas with a 59% match.
Chryseobacterium is gram-negative, rod-shaped, does not form spores, yellow-pigmented, and is found in soil, plants, and wastewater (Kampfer, et. al., 2003). In lab 3, sample 2 was identified as coccus-shaped, gram positive, and was orange-yellow in color. The different results could be due to human error in the gram stain and in shape identifying, or it could be a completely different bacteria considering it was only an 80% match.
Pseudomonas bacteria are found in many different environments, including lakes, marshes, swamps, in plants, and in animal tissues (Özen and Ussery, 2011). They are a very diverse group, but studies on the 16S rRNA gene have classified them into two categories: P. aeruginosa and Pseudomonas fluorescens (Özen and Ussery, 2011). Pseudomonas do not form spores, are gram-negative, rod-shaped, and aerobic (Özen and Ussery, 2011). In lab 3, sample 3 was identified as bacillus (rod-shaped), gram-positive, and pale-yellow, white in color. The rod-shaped feature is consistent with pseudomonas bacteria, but the gram stain results are different. This could be due to error in interpreting the gram stain results, or it may be different bacteria considering there was only a 59% match.
References:
Kampfer, P., Dreyer, U., Neef, A., Dott, W., & Busse, H. (2003, January 1). International Journal of Systematic and Evolutionary Microbiology. Retrieved March 4, 2015, from http://ijs.sgmjournals.org/content/53/1/93.full
Özen, A., & Ussery, D. (2011, August 3). Defining the Pseudomonas Genus: Where Do We Draw the Line with Azotobacter? Retrieved March 4, 2015.
CV
Lab 6: Embryology and Zebrafish Development
26 February 2015
Purpose: The purpose of this lab is to identify the stages of embryonic development, compare development in different organisms, and to set up an experiment to study the effects of an environmental condition on embryonic development. It is hypothesized that there will be a dose-response relationship, in which, increased concentration of retinoic acid (Ret A) will produce more deformities in the embryo. If the embryos are placed in Ret A, then abnormalities in the heart, body, and brain will develop.
Materials and Methods: Procedure I: The embryological development of starfish, frogs, chicks, and humans were compared using dissecting and compound microscopes. Procedure II: Two petri dishes were obtained. 20mL of distilled water were placed in one dish, which is the control, and the other dish contained 10mL of Ret A and 10mL of distilled water to create a 1:1 ratio of Ret A and distilled water. Then 20 zebrafish embryos were placed into each petri dish using a cutoff pipet and the embryos were observed under a compound microscope to determine their developmental stage. The embryos are to be observed 4, 7, and 11 days after the start of the experiment.
Data and Observations: Table 1: Comparison of the Embryological Features of a Developing Starfish, Frog, Chick, and Human
| Feature | Sea Star | Frog | Chick | Human |
| Relative Egg Size | tiny | slightly larger | big | huge |
| Fertilization | external | external | internal | internal |
| Amount of Yolk (Holo/Mero) | holo | holo | mero | holo |
| Distribution of Yolk | isolecithal | telocithal | telocithal | isolecithal |
| Gastrulation | 3 germ layers | 3 germ layers + blastoporeneural tube | 3 germ layers+ blastodisk + primitive streak | |
| Larval Stage | motile | tadpole | enclosed in egg | |
| Waste Disposal | disperse into water | disperse into water | allantois | diffusion |
| Protection | motile | motile | shell | mother |
Table 2: Zebrafish Observations
| Day | Control/RetA? | Number of Dead Eggs | Number of Living Embryos | Number of Larvae | Number of Dead Larvae | Developmental Stage | heartbeat of Lavrae | Observations | Body Length | Length of Tail | Diameter of Eyes | |
| 1-Feb-19 | Control | 0 | 42 | n/a | n/a | 30% epiboly | n/a | n/a | n/a | n/a | ||
| 1-Feb-19 | RA | 0 | 27 | n/a | n/a | 30% epiboly | n/a | n/a | n/a | n/a | ||
| 4-Feb-23 | Control | 21 | 0 | 21 | 0 | 48 hours/36 hours | 24 beats/10 sec | 1.6 mm | n/a | n/a | ||
| 4-Feb-23 | RA | 14 | 4 | 8 | 1 | 48 hours/36 hours | 18 beats/10 sec | 1.9 mm | n/a | n/a | ||
| 7-Feb-26 | Control | 0 | 0 | 21 before spill, 5 after spill | 0 before spill, 16 after spill | 36-60 hours | 24 beats/10 sec | irregular heartbeat, bulging side | 2.7mm | n/a | n/a | |
| 7-Feb-26 | RA | 4 | 0 | 8 | 0 | 48-60 hours | 27 beats/10 sec | white dots around the eyes | 3.1mm | n/a | n/a | |
| 11-Mar-02 | Control | 0 | 0 | 5 | 0 | 60-72 hours | 25 beats/10 sec | 3.3 mm | n/a | n/a | ||
| 11-Mar-02 | RA | 0 | 0 | 7 | 1 | 60-72 hours | 23 beats/10 sec | 3.5 mm | n/a | n/a | ||
| 14-Mar-05 | Control | 0 | 0 | 4 | 1 | >72 hours | 25 beats/10 sec | 3.4 mm | ||||
| 14-Mar-05 | RA | 0 | 0 | 0 | 7 | >72 hours | 0 beats/10 sec | 3.7mm |
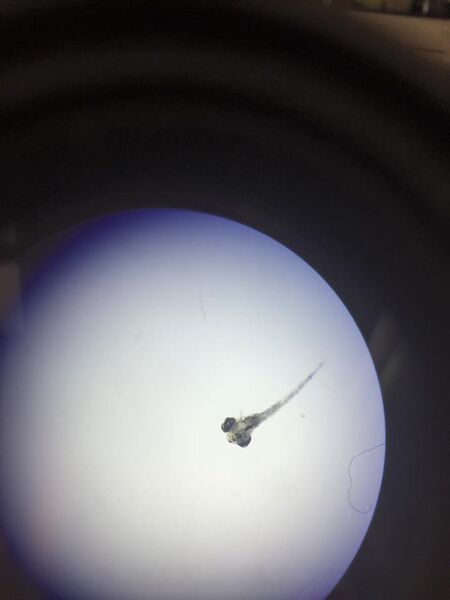
Figure 1: Day 1 Control Petri Dish (left) and Ret A Petri Dish (right)
Figure 2: Day 4 Control Fish (4x Magnification)
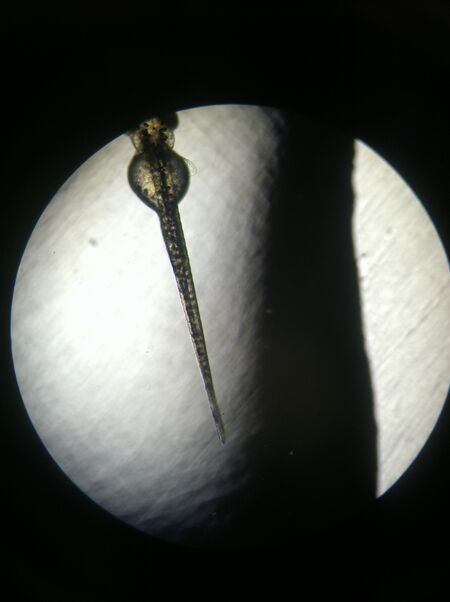
Figure 3: Day 4 Ret A Fish (4x Magnification)
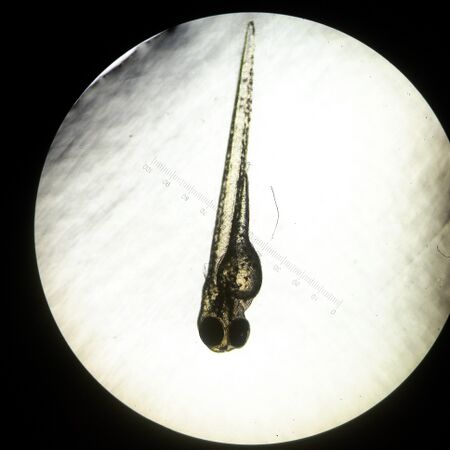
Figure 4: Day 4 Ret A Fishes (Dissecting Microscope)
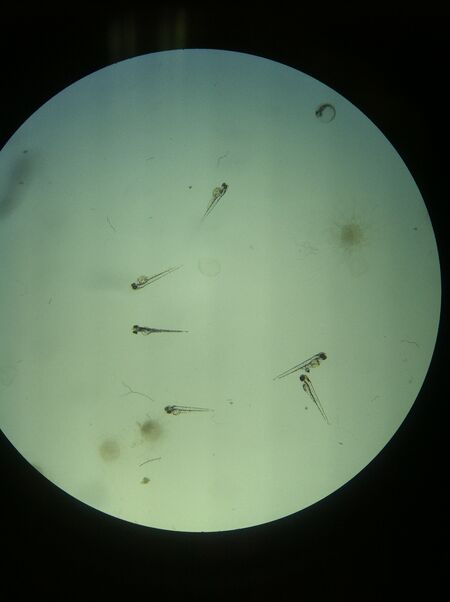
Figure 5: Day 7 Control Petri Dish (left) and Ret A Petri Dish (right)
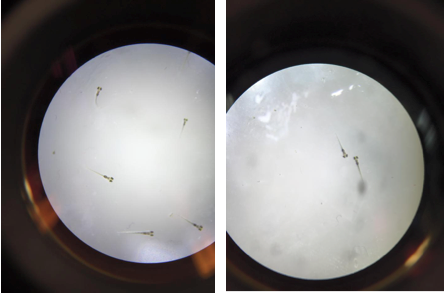
Figure 6: Day 7 Control Fish (4x Magnification)
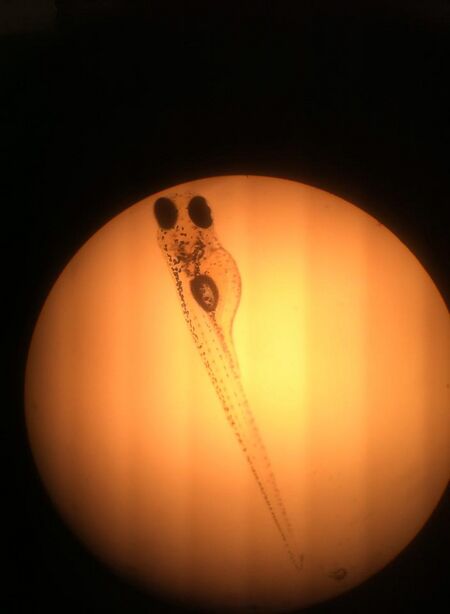
Figure 7: Day 7 Ret A Fish (4x Magnification)

Figure 8: Day 11 Control Fish (Dissecting Microscope)
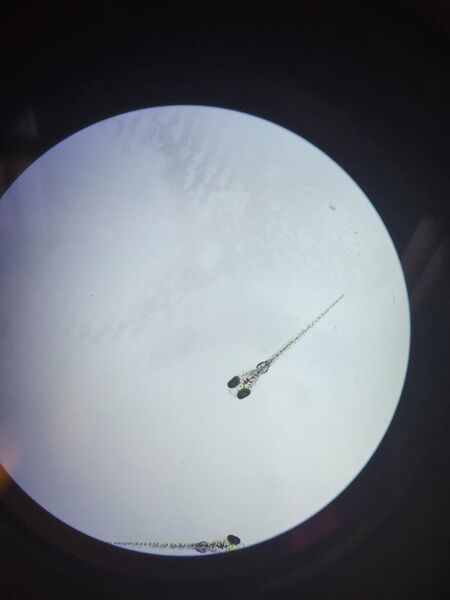
Figure 9: Day 11 Ret A Fish (Dissecting Microscope)
Conclusion and Further Discussion:
Overall, the hypothesis that deformities will be present in the embryos treated with retinoic acid was supported. There was no difference in the length of the larvae, however there was a huge difference in eye diameter. On day 11, the eyes of the treated larvae were much larger than that of the control group. Also, the heartbeat of the treated fish varied from week to week, ranging from 18 beats per 10 seconds to 27 beats per 10 seconds; whereas, the heartbeat of the control group stayed consistently around 24-25 beats per 10 seconds. By day 11, all the treated larvae died and the control group remained healthy. In future studies, it would be useful to test the fish at different concentrations of retinoic acid to see the effects on the embryo at various concentrations. This would help determine a safe dose of vitamin A for drugs and would provide information of how much vitamin A can be ingested before there are detrimental effects on the embryo.
CV
2.20.15
Very good entry. Well organized.
Food web is too simple.
SK
Lab 5: Invertebrates
19 February 2015
Purpose: The purpose of this experiment was to identify the importance of invertebrates and to learn how simple systems evolved into more complex systems. It is hypothesized that the manicured grassland (transect 5) will show many types of arthropods. If there are many different arthropods in transect 5, then when the Berlese funnel is examined, it will show various types of arthropods.
Materials and Methods: Procedure I: A acoelomate (Planaria), nematode, and coelomate (Annelida) was observed using the dissecting scope to identify the type of moment of this worm and how it reflects the simplicity of the organism. Then the cross section of a Planaria and nematode were observed with a microscope. Procedure II: Organisms in each of the five major classes were observed: arachnida, diplopoda, chilopoda, insect, and crustacean. Procedure III: The Berlese Funnel was deconstructed and half the liquid and organisms were poured into one petri dish and the other half was poured into another petri dish. Both dishes were examined using a dissecting scope. Any arthropoda invertebrates were identified and classified. Procedure IV: Any vertebrates that inhabit and pass through the transect (transect 5) were observed and noted.
Data and Observations: Table 1: Soil Invertebrates from Transect 5:
| Organism | Length (in mm) | Number in Sample | Description of Organism |
| Springtail X (arthropod) | 5.0 mm | 3 | from top half of water, small antennae, large body |
| Proturan X (arthropod) | 3.0 mm | 2 | from bottom half of water, long antennae on both sides of body, 6 legs, long body |
| cochineal | 4.0 mm | 1 | antennae, huge wings, looks like a gnat, small body, huge back antennae (from different group) |
Table 2: Vertebrates from Transect 5:
| Vertebrate (common name and Phylum) | Genus and Species |
| Black Squirrel (Chordate) | Sciurus carolinensis |
| Brown Squirrel (Chordate) | Sciurus carolinensis |
| Field Sparrow (Chordate) | Spizella pusilla |
| American Tree Sparrow (Chordate) | Spizella arborea |
| Cottontail Rabbit (Chordate) | Sylvilagus floridanus |
Figure 1: Vertebrate Food Web found in Transect 5:
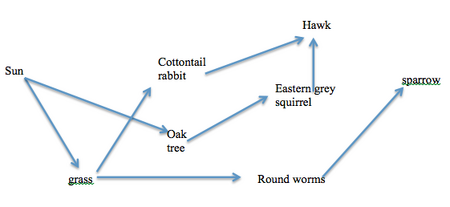
Conclusion and Future Discussion: The hypothesis that there will be many different types of arthropods in transect 5 was not supported. There were only two different organisms identified in this lab section of transect 5 and another group’s experiment had to be used to increase the diversity. There were many types of the same arthropods, however. For example, 3 springtails and 2 proturans were discovered. The springtails were found in the top half of the liquid and the proturans were found in the bottom half. This is due to the fact that the springtails found were larger organisms and rested in the leaf litter, whereas the proturans were smaller so they lied closer to the soil, or in the soil. In this lab, acoelomates, nematodes, and coelomates were examined. The acoelomates are the simplest form of worm and are known as flatworms. Coelemates, like Annelida, are the most complex type of worm because of their complete coelem, or interior body cavity. The acoelomates are limited in structure and movement and thus, do not move as often. The nematodes and coelomates move by whipping their bodies back and forth because they have segmented and structured bodies that allow for that movement. Going back to the transect, there were 5 vertebrates identified that inhabit the area. All the organisms were of the phylum Chordate and are commonly found in green spaces. The species that were identified were two types of sparrows (American Tree and Field), a cottontail rabbit, and two types of squirrels (one black and one brown). The biotic factors in the transect that would benefit these species are the grass, the tree, sticks, and leaves. Rabbits feed on grass, the tree acts as shelter for the squirrels and sparrows, and the sticks and leaves allow the sparrows to build nests in the tree. Also, the tree is an oak tree, so it produces acorns in the spring, which is food for the squirrels. The abiotic factors that would benefit the animals are the water and concrete. The water is a necessity for life and the concrete allows people to walk directly past the transect. The people walking by could drop things, like food or objects that could be used in nests. CV
2.20.15 Very good notebook entry. Nice table and nice to see a drawing! SK
Lab 4: Plantae and Fungi
12 February 2015
Purpose: The purpose of this experiment is to observe the characteristics and diversity of different plants and to understand the importance and function of fungi. It is hypothesized that there will be mostly angiosperms in the manicured grassland. If there are more angiosperms than any other type of vascularization, than more angiosperms will be observed.
Materials and Methods: Procedure I: Two Ziploc bags were obtained and transect 5 (manicured grassland) was revisited. In the first bag, about 20 percent of the bag was filled with soil and the rest was filled with leaf litter. This will be used to set up a Berlese funnel. In the second bag, five plants that represented the transect were collected. Photos were taken of the trees in the area and any seeds, pinecones, and flowers were collected and brought back to the lab. Procedure II: The moss, Mnium, was observed and compared to the height of a lily plant. Then, the cross section of the lily stem was examined under a microscope and the xylem and phloem layers were identified. Procedure III: The leaves of moss were examined using a dissection scope and the different parts were identified. Procedure IV: The moss, Polytrichum, was examined and the male and female gametophytes and sporophyte. Then a lily flower was dissected and the anther, stigma, and style of the flower were identified. Finally, the seeds from the transect were dissected and the parts were identified. Procedure V: A fungus was observed underneath a dissecting scope. Procedure VI: 25 mL of 50:50 ethanol/water solution was poured into a 50 mL conical tube. A piece of screening material was cut and placed into the bottom of the funnel and the sides of the screen were taped. The leaf litter collected from the transect was put into the top of the funnel and then the funnel was placed in a ring stand. The conical tube was then taped to the funnel and the setup was placed under a 40-watt lamp with an incandescent bulb about 1-2 inches away from the leaf litter. Everything was covered with foil. The Berlese funnel will be examined in a week.
Data and Observations: Table 1: Identity of Plants from Transect 5 (Manicured Grassland)
| Transect Sample Plants | Location and # in transect | Description | Vascularization | Specialized Structures | Mechanisms of Reproduction |
| 1 | Rose bud | ~3.5 cm tall | bundles in a ring, taproot | dicot, network of veins | angiosperm |
| 2 | Long Grass | ~30 cm tall, thin grass, green | scattered bundles, fibrous root | monocot, parallel veins | angiosperm |
| 3 | Weed 1 (from edge of transect) | ~8.0cm tall, parts into 3 flowery-leaves | bundles in a ring, taproot | dicot, network of veins | angiosperm |
| 4 | Weed 2 (from middle of rose bush in soil) | ~11.0 cm tall, green, leafy, 3 leaves | scattered bundles, fibrous root | monocot, parallel veins | seedless plant |
| 5 | Leaf (from oak tree overhead) | ~11.5 cm tall | bundles in a ring, taproot | dicot, network of veins | angiosperm |
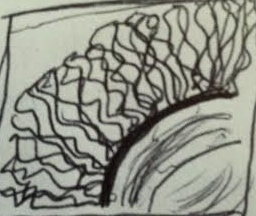
Figure 1: Observation of Fungi Through a Dissection Microscope
Conclusion and Future Discussion:
Overall, the hypothesis that there would be more angiosperms than any other type of vascularization was supported. Four out of the five plants observed from the transect were angiosperms. This could be due to the fact that transect 5 is a well-maintained manicured grassland, so more flowering plants would be placed there for aesthetic purposes. Figure 1 displays a part of mushroom seen through a dissection microscope. The mushroom is a Basidiomycota fungi because of the branches found underneath the cap, also known as basidia. Black-bread mold was also observed under a microscope and the mycelium and sporangia were viewed. Sporangia are black sacks that contain spores, which allow fungi to produce more fungi. In the future, it would be useful to observe the transects in the spring or summer, when the plants are alive and in full bloom. It would allow for more plants to be seen that might not have been viewed in the winter
CV
2.10.15 Excellent notebook entry. Good description of gram staining and PCR set up. Nice data tables. SK
Lab 3: Microbiology and Identifying Bacteria with DNA Sequences
5 Feb 2015
Purpose: The purpose of this lab is to understand the characteristics of bacteria and observe which are resistant to antibiotics. Also, it is important to see how DNA sequences are used to identify species of bacteria. It is hypothesized that the agar plates without the tetracycline will have more colony growth then the plates with tetracycline. If the nutrient agar plates without tetracycline has more growth than the plates with, then more colonies will be observed on the no-tet plates.
Materials and Methods: Procedure I: The growth on the agar plates (set up the previous week) from the Hay Infusion Culture was observed. The number of colonies on each plate were counted and documented. Procedure II: Wet mound Procedure: A drop of water was placed onto a slide. Then, a loop was sterilized over a flame and a tiny amount of the growth from a selected colony was scraped with the loop and mixed into the drop of water. A slipcover was placed over the mound and oil was put on top of the slip. The wet mound was then observed using the 10x and 40x objective.
Gram Stair Procedure: A drop of water was placed onto a slide, and then a loop was sterilized and used to scrape a tiny amount of growth from a selected colony. The sample was mixed into the water on the slide and the area was circled with a red wax pencil underneath the slide. Holding the slide in a test tube clamp, the slide was passed through a flame three times with the bacterial smear side up. Once the slide was dried and cooled, it was placed in a staining tray and covered in crystal violet for 1 minute. After the minute, the stain was rinsed using a wash bottle filled with distilled water. The slide was then covered with Gram’s iodine mordant for 1 minute and rinsed using the wash bottle filled with water. To decolorize the smear, 95% alcohol was placed on the slide for 20 seconds and then rinsed gently with water until all the color has drained from the slide. Next, the slide was covered with safranin for 20-30 seconds and rinsed using water. Finally, the excess water was blotted away using a kimwipe and the smear was viewed under 40x and 100x oil immersion objectives. Procedure III: A single colony of bacteria was transferred to a sterile tube filled with 100μL of water. The tube was incubated at 100°C for 10 minutes in a heat block. The samples were then centrifuges for 5 minutes at 13,400 rpm. While the samples are in the centrifuge, 20μL of primer/water mixture were added to a labeled PCR tube. The tube was mixed gently to dissolve the PCR bead. Finally, 5μL of supernatant from the centrifuged samples were added to the 16S PCR reaction and the tubes were placed in the PCR machine.
Data and Observations: Table 1: Agar Plate Dilution Results
| Dilution | Agar Type | Colonies Counted | Conversion Factor | Colonies/mL |
| 10^-3 | nutrient | 1000 | x10^3 | 1,000,000 |
| 10^-5 | nutrient | 85 | x10^5 | 8,500,000 |
| 10^-7 | nutrient | 6 | x10^7 | 60,000,000 |
| 10^-9 | nutrient | 0 | x10^9 | 0 |
| 10^-3 | nutrient + tet | 250 | x10^3 | 250,000 |
| 10^-5 | nutrient + tet | 8 | x10^5 | 800,000 |
| 10^-7 | nutrient + tet | 1 | x10^7 | 10,000,000 |
| 10^-9 | nutrient + tet | 0 | x10^9 | 0 |
Table 2: Bacteria Characterization
| Colony Label | Plate Type | Colony Description | Cell Description | Gram +/- |
| 1 | nutrient + tet (10^-3) | orange-red, 0.6 cm diameter, circular-undulate, raised convex | coccus (<1μm) | - |
| 2 | nutrient + tet (10^-5) | orange, 0.7 cm diameter, circular-hard edge, raised convex | coccus | + |
| 3 | nutrient (10^-5) | pale yellow, 0.5 cm diameter, circular-hard edge, flat | bacillus, flagella to move (1μL) | + |
| 4 | nutrient (10^-5) | white, 1.0 cm diameter, circular-undulate, flat | spherical coccus | + |
Conclusion and Future Discussion: Furthermore, the hypothesis is supported because the plates without tetracycline produced more growth than the plates with tetracycline. This is due to the fact that tetracycline is an antibiotic, and thus, kills off bacteria without the resistance gene and shows less growth. Along with the amount of colonies, there were also differences in the colony size, shape, and color. The +tet plates contained bigger colonies that were yellow, orange, and red in color, and had more of a raised convex shape to them. The no-tet plates had smaller colonies that were many different colors (blue, white, red, yellow, orange), and were very flat. A fungus was also observed on the no-tet plate. Based off of the results, it is predicted that the antibiotic affected the bacteria in the white and blue colonies on the no-tet plates because they were not observed on the +tet plates. Although the types of bacteria are unknown, it is certain that no archaea bacteria was observed because they only inhabit extreme environments, like hot springs, and none of the samples were taken from an extreme enough environment. Also, the Hay Infusion Culture was once again observed, and smelled like rotten eggs, as opposed to a swamp in the week before, and more water had evaporated, so the water became darker. One reason the culture could have smelled worse this week is because the bacteria had more time to grow and reproduce, causing a more pungent smell. In the future, I would like the chance to redo the gram stain to get a better picture when looking through the microscope. It would also be beneficial to observe colonies on all the plates to see if the bacteria are in fact the same throughout. -CV
2.4.15 Good start. You need to include some pictures and I think you were trying to include a table. To translate an excel table into OWW use: http://excel2wiki.net. Make the table in excel, copy and paste it into the website and then copy and paste the output into your notebook. SK
Lab 2: Identifying Algae and Protists
29 January 2015
Purpose: The purpose of this lab is to use a dichotomous key to identify unknown algae and protists and to understand the characteristics of each to be able to identify species from the transect. It is hypothesized that the Hay Infusion Culture will contain more protists than algae. If there are more protists than algae, then the number of protists observed will be greater than that of algae.
Materials and Methods: Procedure I: A wet mound of known organisms was made on a slide and observed with a microscope at 4x and 10x. The sizes of the organisms were recorded using the ocular micrometer and a dichotomous key was used to identify the known organisms. The key was then used to identify as many different organisms as possible from the Hay Infusion set up last week.
Procedure II: 4 tubes, each containing 10 mLs of sterile broth, were labeled 10-2, 10-4, 10-6, and 10-8. 4 nutrient agar and 4 nutrient agar plus tetracycline plates were obtained and labeled. One plate from each group was labeled 10-3, 10-5, 10-7, and 10-9. Then the Hay Infusion Culture was swirled and 100 μLs were drawn and placed in the 10-2 tube. That tube was then swirled and 100 μLs from the 10-2 tube was taken and added to the 10-4 tube. This was repeated to make the 10-6 and 10-8 dilutions. 100 μL from the 10-2 was pipetted onto the nutrient agar plate labeled 10-3. The sample was carefully spread onto the plate. This was repeated for the +tet plate labeled 10-3. This procedure was repeated for the 10-4 dilution on the 10-5 plates, the 10-6 dilution on the 10-7 plates, and the 10-8 dilution on the 10-9 plates. Then the +tet agar plates were wrapped in aluminum foil and all the plates were left out at room temperature to incubate for 1 week.
Data and Observations: Hay Infusion Characteristics: • Smells like a swamp • White mold in one area floating at the top • Surface of the water was covered in a murky, white film
Table 1: Hay Infusion Culture Characteristics Samples taken from: Species type Size Motile? Phosynthesizing? White mold section Pandorina algae 62.5μm Yes - cilia yes Paramecium protist 300μm Yes - cilia no Colpidium protist 70μm Yes - cilia no Bacteria bacteria 1-2μm no no Bottom of jar against colpidium protist 70μm Yes - cilia no the side
Conclusions and Future Discussion: Overall, there were not that many species observed in the Hay Infusion Culture, but there were more protists than algae, supporting the hypothesis. This could be due to the fact that algae grow in wetter areas, and the area the sample was taken from was dry land. The organisms closer to the plant matter would differ from those away from the plant matter because species are adapted to different environments. Some might feed on the plants, while others feed on microorganisms found in soil. In the future, I would want to grow the culture for longer to see how species compete for food to survive. I would also like to sample more areas around AU to see how the algae and protists in one area vary from the rest of the campus. -*Caroline Vill 23:08, 28 January 2015 (EST):
1.27.15 Good first entry. The picture didn't upload correctly but we will work on uploading pictures in lab. SK
Lab 1: Biological Life at AU
26 January 2015
Purpose: The purpose of this lab is to observe the evolution of a member of the green algae group, the Volvocine Line, and understand how natural selection leads to evolution. Also, to observe a specialized niche on the AU campus and characterized the various inhabitants within that ecosystem. It is hypothesized that evolution does not always move towards increased complexity, and that more biotic factors will be identified in the transect then abiotic factors. If there are more biotic factors than abiotic factors in the transect, then more biotic components will be observed.
Materials and Methods: Procedure I: A microscope was obtained and the first alga to be observed was Chlamydomonas, then Gonium was observed, followed by Volvox. The number of cells, colony size, specialization, mechanisms of motility, reproduction methods, and a picture was recorded for each species. Procedure II: 5-20x20 meter transects were placed around the AU campus and marked with Popsicle sticks. Groups of 3 were picked at random and assigned to a transect. An aerial view of the plot was drawn and all the abiotic and biotic in the areas were listed. A sterile 50 mL conical tube was used to take a soil/vegetation sample that is a true representation of the transect. Once back in the lab, a Hay Infusion Culture was made. 10-12 grams of the soil sample were placed in a plastic jar with 500 mLs of Deerpark water. Then 0.1 grams of dried milk were added and the culture was mixed gently for 10 seconds. Finally, the lid was removed and the jar was placed in the lab and allowed to sit for a week.
Data and Observations: Characteristics of the Transect: Transect 5 located on the main quad right outside of Hurst. It was a trapezoid shape, half covered in grass and the rest was soil. The abiotic factors in this area were concrete, a plastic fork, snow (water), and soil. The biotic factors were dead leaves, sticks, grass, rose bushes, and other unidentifiable bushes. Aerial View of Transect 5:
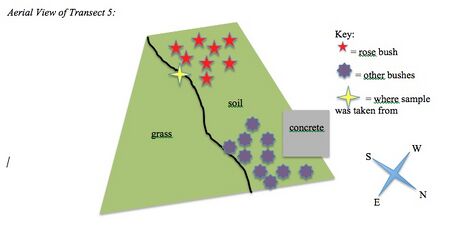
Conclusions and Future Directions: The hypothesis was supported in the fact that there were more biotic factors than abiotic factors in the transect. While the count indicated 5 biotic factors, there are microscopic organisms, like protists and algae, which are unaccounted for. The Hay Infusion Culture will reveal the protists and algae the following week. Factors that might influence the transect is the huge tree located right next to the plot. This would explain all the dead leaves in the area. Also, transect 5 is located in the main quad next to a sidewalk and is in a very high-traffic area. This would explain the plastic fork, as well as the lack of vegetation. Other than grass and aesthetic bushes, there were no other plants and weeds are not able to grow because the area is well maintained. In the future, I would like to examine an area located in a secluded area on campus to note the differences between the factors in each plot.
-CV
19 January 2015
I think I've figured out how to use this site...I hope this works! *Caroline Vill 20:11, 19 January 2015 (EST):