User:Andy Maloney/Notebook/Lab Notebook of Andy Maloney/2010/03/17/ReAutomation
Purpose
I've decided to go ahead and reautomate the data that I took last week. The reason I want to do this is because when I initially did it, I didn't use Larry's super awesome "Ignore things close to edges" and so I got a lot of useless data in the automation. Also, the default size limitation when tracking was set to 190 pixels. This corresponds to microtubules that are about 35 µm in length. This is huge and I want to track smaller things as well. So I've reduced the pixel limit to 55 which corresponds to microtubules that are about 10 µm in length. I should get more data to use especially for the whole and alpha casein.
- Steve Koch 03:24, 26 March 2010 (EDT): At some point (and maybe not all the time), you may want to use the "shearing" method to make more uniformly sized MTs. This is not a must, though, as I am not sure that sheared would act the same as not-sheared. The doublecortin method would also be very cool, but too difficult for now.
2010-03-18 Update
Well, the automation is still going. There are a tremendous amount of microtubules to track and it is taking a bit longer than I would have expected but, it is trucking along and I absolutely love the automation software. Hopefully it will be done tomorrow and I can look at the new generated data soon.
2010-03-25 Update
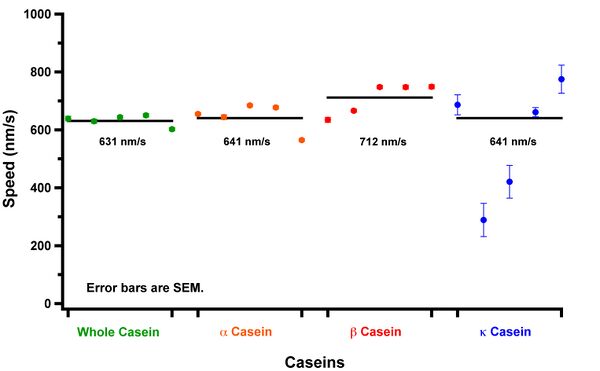
I have finally finished the reautomation on the data obtained on March 4th. Below are my results in graph form.
I'm curious about the beta casein. Koch and I have an idea as to how being close to the tape in my flow cells may cause the speed of the microtubules to be reduced. I will have to investigate this in my next attempt at this experiment.
As of right now, there are some conclusions that can be made from this data.
- Whole casein and alpha casein are remarkably similar. Both allow for really tiny microtubules to exhibit motility. We believe that this may be caused from motor densities on the casein surface. There is literature that describes how gliding speeds can depend on kinesin motor densities. I will have to look into this at some point. Their speeds are also remarkably similar.
- Beta casein appears to allow for faster microtubules. With the above hypothesis about motor densities, this may mean that the density of kinesin on the surface is less than it is for whole casein and alpha casein since they move faster. To further justify this idea, I have noticed that beta casein will support tiny microtubules in the assay, however, they do not exhibit motility. If tiny ones do get attached to the surface and glide, they inevitably fall off rather quickly. This does need to be investigated more.
- Kappa casein does not support tiny, small or even medium sized microtubules. Plus, it takes over 400 seconds to see motility in the assay as opposed to the 30 seconds or so it takes to see it in the other assays.
To explain more about the 3rd point above, after sealing my flow cell, I time how long it takes me to observe motility (from sealing to the first region of interest in the microscope). In relative terms, it takes kappa casein passivation a longer time to show microtubule motility than it does the other caseins. This could be because of motor density. If it is, then this would explain why I cannot get motility on a kappa casein surface with "small" (sorry, I know small is a relative term I will define later) microtubule.
- Steve Koch 03:29, 26 March 2010 (EDT): Do you see small MTs in the kappa casein samples? I remember when you first showed me the (verma? et al.) study that I assumed kappa casein was the best because it didn't "break apart" the MTs. Is that still possible. I.e., that there's contaminants in the other 3 caseins that causes MTs to break into littler ones? Judging by what you've said and what I've seen, I wouldn't think so, but just putting that out there. BTW: This page is awesome! I love how much and how clean of data you can get and analyze with the combination of your assays and technique and Larry's software.
2010-03-29 Update
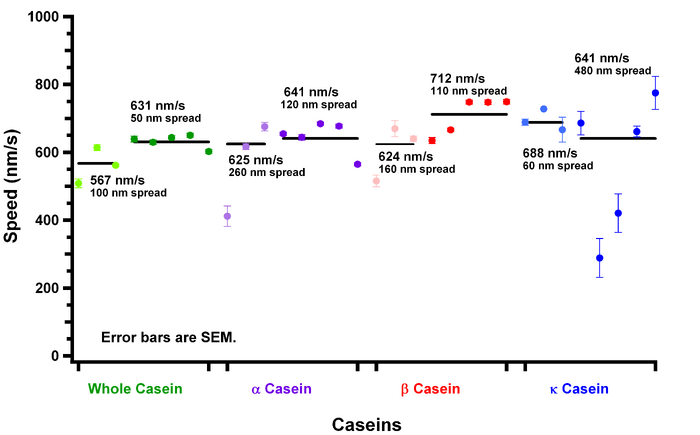
I finished looking at speed data for the beginning 3 regions in the experiment. For a comparison of above, I graph the data below.
There are several interesting things to note here.
- Kappa casein is not like the other caseins. How it can allow for motility is still a mystery. The following remarks are not including kappa casein.
- Early regions show slower speeds.
- Early regions also have a larger spread in data.
- Alpha casein speeds for early times averages to later times more quickly. This is seen in the fact that in the first 3 regions (light purple), the average speed is very similar to the last 5 regions (dark purple) I looked at.
I do believe that I have definitive results stating that the speed people observe is dependent on passivation and the time that they observe the speed at. Of course, this speed difference could be an artifact from the temperature of the flow cell. This is something to be investigated as well.
I should also note that the speed variation from one region to the next could be dependent on how close I am to the tape of the flow cell.