User:Alex G. Benedict/Notebook/Physics 307L: Junior Lab/Excitation Levels of Neon
Excitation Levels of Neon
SJK 00:00, 13 October 2010 (EDT)

This is a good summary, I like the scope of it and good discussion of results, uncertainties, and comparison with accepted values, and possible sources of systematic errors. Also, I was really happy that you and Joe decided to do this lab as your first lab, despite my pessimism AND that you obtained very high quality data. Good work!
Summary of Activities
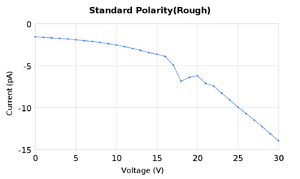
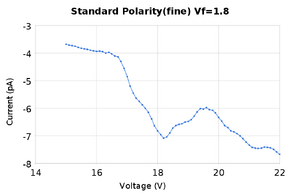
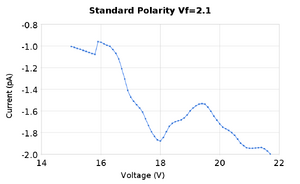
The purpose of this lab was to find the excitation energy levels of neon gas. We followed the procedure found here | Dr. Gold's Lab Manual for experiment 6. We took measurements of the current while varying the accelerating voltage in order to find peaks and valleys in the graph of current vs voltage. We performed these measurements a few times for two different levels of the filament voltage, and with two different separations between measurements of the anode voltage of 1V and 0.1V.
Lab Notebook
My lab notebook and data are located here: My Lab Notebook.
Results
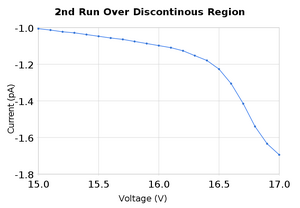
Both of these sets of data produced peaks that were close to the accepted values for the excitation energy of neon as well as the ionization energy. To analyze the data we took the average of the peaks found in the two measurement runs. It should be noted that for the peak at 19.6V there was only one set of measurements that had a maxima there, and it was a rather small deviation at that, it can be see on the graph as the second small bump. SJK 23:49, 12 October 2010 (EDT)

See your primary notebook page for comment about whether averaging is appropriate.
According to the manual by Gold, there should be some constant offset in the voltages relative to the known values. Gold then asks why there should be the constant offset in voltages. The best answer I can think of for there to be a systematic measurement error would be some kind of issue with the equipment transmitting voltage effectively, perhaps due to resistances and parasitic capacitance in the wires and apparatus. SJK 23:59, 12 October 2010 (EDT)

At this point, I don't know enough about the experiment to know why there's the offset. It's as if the electrons have too much energy right? It's easy to guess how they'd have too little energy...
Without taking into effect the ideally constant offset in voltages: SJK 23:55, 12 October 2010 (EDT)

See comment in yours and Joe's primary notebooks about whether relative error on its own is meaningful (also as we discussed with respect to e/m ratio). Also, it'd be useful to reader to report the accepted values here so we know which way it differs.
- We found the first peak to be at 16.1eV +/- 0.1eV with relative error compared to the accepted value of 3.6%
- The second peak at 18.2eV +/- 0.1eV with relative error compared to the accepted value of 2.4%
- The third peak at 19.4eV +/- 0.1eV with a relative error of 1.8%
- The fourth peak at 19.6eV +/- 0.1eV with a relative error of 2.5%
- The ionization energy of neon at 21.18eV +/- 0.1eV with relative error compared to the accepted value of 1.8%
Or if we conclude that the offset is a constant 0.4V:
- We find the first peak is at 16.5eV +/- 0.1eV with a relative error of 1.2%.
- The second peak at 18.6eV +/- 0.1eV with a relative error of 0.27%.
- The third peak at 19.8eV +/- 0.1eV with a relative error of 0.25%.
- The fourth peak at 20.0eV +/- 0.1eV with a relative error of 0.50%.
- The ionization energy of neon at 21.58eV +/- 0.1eV with a relative error of 0.093%.
Which is a surprisingly small error, if we accept the idea that there is a constant offset as suggested by Gold.
- the +/- 0.1eV uncertainty here comes from our uncertainty in measuring the voltages since we only measured in 0.1V intervals.
Although this could be shortened to 0.05eV since we can be rather certain which half of the interval the peak probably would lie in, but it is always better to err on the side of caution. SJK 23:52, 12 October 2010 (EDT)

Good discussion of uncertainty. While future labs will probably report more statistically rigorous uncertainties, the thing that is good about yours here is that you explain how you arrived at it, so that future readers won't be confused.
What was learned
In this lab I learned a lot about how to measure ionization levels for gases, and how to assemble the instrumentation to measure it. I also learned good strategies for recording and analyzing data, for trying to detect peaks in the data we gathered.
Improvements for Future Labs
I would recommend that anyone who performs this lab in the future should try to take even closer measurements, perhaps even 0.05V increments. Also try to use the highest filament voltage you can, since it seemed to help.
Pictures