User:Abigail M. Fogg/Notebook/Biology 210 at AU
Embryology and Zebrafish Development
INTRODUCTION
The purpose of this lab was to learn about the stages of embryonic development, and to design an experiment to study how environmental conditions affect embryonic development. In this lab the effects of caffeine on embryonic development were observed. My partner and I were not present for the first week of the study, so we started the study at one week with the healthiest fish that had already hatched and developed.
METHOD
Zebra Fish Experiment Procedure
1. Make an hypothesis, prediction, and experimental plan.
2. Observe the zebrafish embryos carefully and determine their developmental stage for the first time reading.
3. Set up the control group and the caffeinated experimental group in covered petri dishes.
4. Use 20 mls of Deerpark water and 20 healthy translucent embryos per dish. Use a dropper to transfer the eggs to the dishes with the appropriate water. The variable tested involves an additive to the water meaning every time the water was changed caffeine was added.
5. Organize the observation schedule and procedure. Make observations and carefully record those over the next two weeks (the procedure for the observations is below)
6. When embryos are 4-5 days old, remove 10 mls of water with any empty egg cases and add 25 mls fresh water. (The empty egg cases will grow mold.)
7. Save any dead embryos in paraformaldehyde that will be provided.
8. One week after the experiment has begun, remove 5 mls water with any egg cases and add 5 mls fresh water or test solution. Preserve 1 to 3 embryos from the control and the experimental groups in paraformaldehyde.
9. Between 1 week and 2 weeks incubation time, remove 5 mls and add 10 mls.
10. Feed two drops of paramecium starting after one week. This can be continued just when the water is changed.
11. Make final observations and measurements at two weeks. At that time the surviving embryos will be collected and placed in an aquarium for safe keeping.
12. Prepare tables and graphs and any calculations of observations.
Making Measurements and Observations
To make observations use dissecting scopes and depression slides to determine the developmental stages at each time point in the experiment. (Refer to hand-out for stages) Fix up to three larvae from week one, three from week two, and save any dead. To fix the larvae, place them in the 0.02% tricaine solution to anaesthetize them. The will be fixed in a vial of 4% formaldehyde.
Zebrafish and Larvae Observation
1. In each dish on each observation day, count the following:
i. number of dead eggs ii. number of living embryos still in egg cases iii. number of living hatchlings (larvae) iv. number of dead hatchlings* After day 4 always keep track of the number of larvae that die.
2. On day 4: Remove 10 mls of water, any empty egg cases and dead embryos.
i. add 25 mls of test solution to test dishes ii. add 25 mls of water to the control dishes
3. On day 7: Add one drop of food (Paramecium) to each dish.
4. Days 4 and 7 and 11: Observe representative embryos/larvae from each plate with the compound microscope. Place a few drops of sample with the organisms in a depression slide and use the 4X objective. Follow the diagram given in class and record the following:
i. degree of body and tail pigmentation (melanophores) ii. eyes and eye movement iii. heart and possibly heart rate iv. pectoral fin development v. yolk sac size (absorbed by day 5-7) vi. development of the swim bladder vii. development of the mouth (protruding jaw) viii. general movement; assign a quantitative score based on a rubric you develop.
You may do this by observing a response to external stimuli like a poke or a shake of the dish (startle response).
5. On day 7: Fix up to three larvae from the control and three from the test dish (number depends on the number surviving and consult with your TA). Use a dropper to get them into the tube. Add 1 drop of tricaine solution per ml of water. Your TA will add the paraformaldehyde and store the samples. You will do another fixation on day 14 and make measurements described in step #7.
6. On day 7 and day 11: Add 10 mls of water to the control and 10 mls water plus test additives to the test sample.
7. Final day with fixed samples:
i. make observations listed in step #4 ii. measure the length of the tail iii. measure the length of the entire larvae iv. measure diameter of the eyes
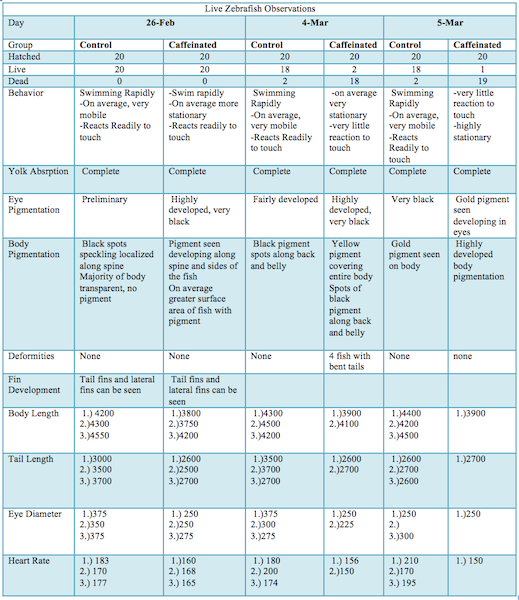
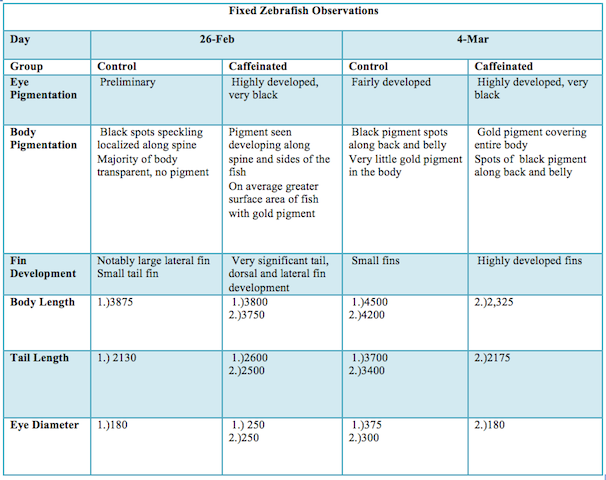
RESULTS
Images
- NOTE: These images are not an accurate representation of pigment development. Images make fish appear darker than they were *
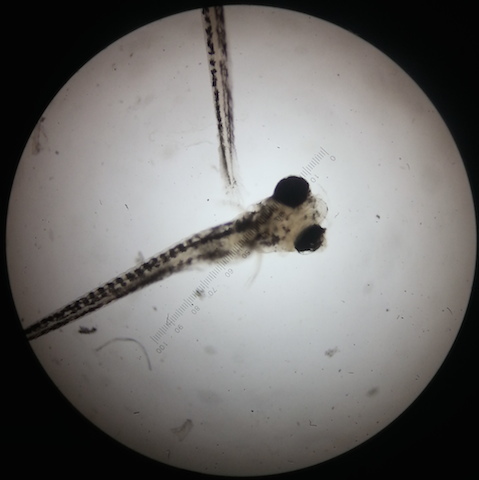
Caffeinated Figure 1: Fixed Feb 26
Caffeinated Figure 2: Fixed March 4
Control Figure 3: Fixed Feb23
Control Figure 4: Fixed March 4
The caffeinated zebrafish showed a higher rate of developing deformities (bent tails, enlarged/undersized eyes), a decreased reactivity to touch, a lower heart rate, and a smaller body size in comparison to the control group. Approximately two weeks into the study there was also a massive die off of the caffeinated group.
DISCUSSION
The results of the study indicated that caffeine alters the development of Zebrafish embryos. The caffeinated zebrafish seemed to show a higher rate of developing deformities (bent tails, enlarged/undersized eyes). The control group did not develop any fish with deformities, while the caffeinated group developed a total of four deformed fish. Figure two shows one of the deformed fish which appears to have one enlarged and one undersized eye as well as a bent tail. The experimental group average body size was also notably smaller than the contol group. The caffeinated fish also showed a decreased reactivity to touch. a pipette was used to touch the fish in order to startle them to see how they would react. At one week of development the reaction of both group was about equal, but throughout the next two observations an extreme decline in reaction was seen in the caffeinated group. The caffeinated group was also seen to be on average more stationary. A lower heart rate was also observed of the caffeinated group. This could possibly be due to the fact that caffeine may potentially retard muscular development (much like that observed in the undersized eye and tail and also the smaller average body size). Approximately two weeks into the study there was also a massive die off of the caffeinated group. There are many reasons why this could have occurred, one being that the hearts of the fish larvae were too under developed to sustain the life of the fish as it began to grow. Likewise, other vital organs could be under developed due to the caffeination and resulted in the death of the larvae. The caffeinated larvae seemed to display a much more rapid development of pigment and fins as compared to the control group, but this growth seemed to be out of proportion with the rest of the larvae`s body (the rest of the body seemed too small for the fins of some fish). This could mean that the caffeine had some effect on the fish`s natural signals for proper growth (cell differentiation and replication). There was clearly too much growth in some areas and not enough in others. This was also seen in the bent tails (one side of the tail is larger than the other) and the deformed eyes (one eye was larger than the other) in figure 2. According to the American Pregnancy Association it is important to remember that caffeine is a stimulant and should be regulated during pregnancy because caffeine will pass through the placenta to the fetus. The fetus` metabolism is still under construction, and it is unable to fully metabolize caffeine. Also this will naturally keep the baby awake and disturb the babies movement pattern. (American Pregnancy Association 2014) In lieu of the zebrafish study, I believe that more research of the effects of caffeine of vertebrate`s development needs to be conducted. There are a few limitations to the study of Zebrafish. To begin, no one model organism will ever be an entirely accurate tool for research regarding the human body. Another disadvantage is that the study of zebrafish is still fairly new. This means that the understanding of this organism in terms of behavior, development, etc. is still very limited. As more research is being done with these organisms scientists understanding is rapidly increasing. And finally, even though zebrafish are vertebrates, their eggs develop outside of the body which differs greatly from the in-utero development of human offspring. (Buske 2014) A future study could investigate the effects of varying concentrations of caffeine would do to the fish embryos. A future study could also use a model organism such as a rat to study the effects of caffeine on offspring in order to study the development of an in-utero specimen.
AF
REFERENCES
Buske, Christine . "Model Organism Week: Research’s Next Top Model." Danio Rerio (Zebrafish): Research's Next Top Model. N.p., n.d. Web. 22 Mar. 2014. <http://www.benchfly.com/blog/model-organism-week-researchs-next-top-model/>.
"Caffeine intake during Pregnancy." American Pregnancy Association. N.p., n.d. Web. 22 Mar. 2014. <http://americanpregnancy.org/pregnancyhealth/caffeine.html>.
Bacteria DNA Analysis and Comparison
INTRODUCTION
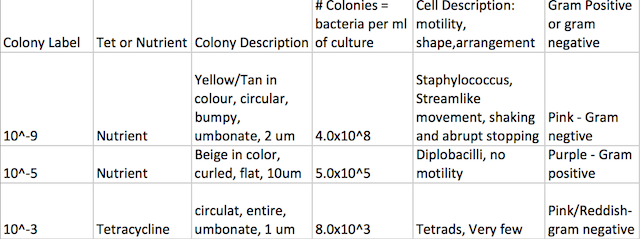
The purpose of this lab was to analyze the DNA that was sent away for sequencing. This analysis will be compared to the observations made of the bacteria found in labs 2 and 3.
METHOD
Reference lab two and three for previous methods
RESULTS
DISCUSSION
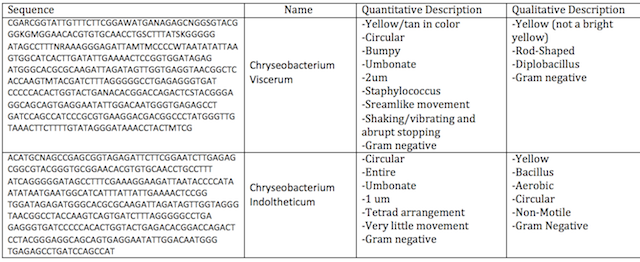
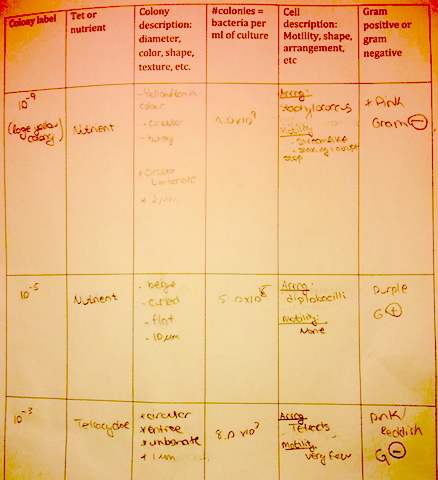
There were many similarities in the comparison of what was viewed in lab two and three, and what was found after DNA analysis and research. The first specimen, which correlated with our sample from agar plate 10^-3 was described to be an identical shade of yellow, also rod-shaped, and gram negative. The second sample which corresponded with our 10^-9 agar plate was also described to be bacillus, non-motile, and gram negative. Although there are a few notable differences there is also room for potential error because the physical description of the colony is reliant on the perception of the viewer. It was notable that there was a presence of many bacterium from the Chryseobacterium lineage in our transect.
References
"Basic Local Alignment Search Tool." BLAST:. N.p., n.d. Web. 1 Mar. 2014. <http://blast.ncbi.nlm.nih.gov/Blast.cgi#364521051>.
"Chryseobacterium indoltheticum - Hierarchy - The Taxonomicon." Chryseobacterium indoltheticum - Hierarchy - The Taxonomicon. N.p., n.d. Web. 1 Mar. 2014. <http://taxonomicon.taxonomy.nl/TaxonTree.aspx?id=99219>.
Dworkin, Martin M., and Stanley Falkow. Proteobacteria: Delta and Epsilon subclasses, deeply rooting bacteria. 3. ed. New York, NY: Springer, 2006. Print.
"Systematized Nomenclature of Medicine - Clinical Terms - SNOMED CT Concept - Classes | NCBO BioPortal." Systematized Nomenclature of Medicine - Clinical Terms - SNOMED CT Concept - Classes | NCBO BioPortal. N.p., n.d. Web. 1 Mar. 2014. <http://bioportal.bioontology.org/ontologies/SNOMEDCT?p=classes&conceptid=http%3A%2F%2Fpurl.bioontology.org%2Fontology%2FSNOMEDCT%2F113550008>.
AF
Invertebrates
INTRODUCTION
The purpose of this lab is to understand the importance of Invertebrates and to learn how simple systems (including specialized cells and overall body plan) evolved into more complex systems. I was absent the day that this lab was conducted so as a substitution I observed a leaf litter sample from a West Virginia soil sample. I also did not observe the live worms.
PROCEDURE
I
1. Using the dissecting scope observe the acoelomate, Planaria. Observe the Planaria as they digest egg yolk. Then look at the cross sectional slide of the Planaria with the microscope. One of the whole mount Planaria slide preparations is stained to show the digestive tract of the organism. Record observations.
2. Observe the nematodes and a cross sectional slide of their pseudocoelomate structure. Again consider their type of movement. Record observation.
3.Observe the coelomate, Annelida. Look at the internal structure and positioning of organs and also the layers of muscle (pink). Record observation.
4.Describe the movements of these three types of worms and how the movement relates to their body structure.
II
1. Break down the Berlese setup and transfer the preservative solution to a Petri dish. Examine under a dissecting microscope. You may use a stirring rod to move the different components of the mixture to see them better.
2. Try to identify the main groups of invertebrates using the two dichotomous keys provided as well as the website:http://www.hope.edu/academic/biology/leaflitterarthropods/
Note: the Berlese funnel method depends on heat and desiccation to force the organisms down through the leaf litter and into the preservative jar. Many of the worms (who dry up rapidly) and insect grubs (who move slowly) will not be captured. In addition, the larger centipedes, millipedes, spiders, ants, and beetles may not get through the screen. You will mostly see the smaller and more mobile species.
OBSERVATIONS
DISCUSSION
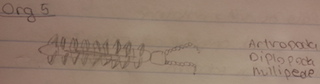
The organisms that were observed ranged from 28.5mm being the largest, to 0.2mm being the smallest. The Diplopoda Centipede seemed to be considerably larger than the Insecta that were observed. The organisms that reside in leaf litter are fairly diverse because the dead organic matter releases nutrients into the soil. It also keeps the soil moist and serves as protection for the individuals residing under it. Some organisms commonly found in this environment are worms, decomposers, snails, and spiders. Different types of fungi and bacteria will also be present as we discovered in lab 3. (Lin,2012)
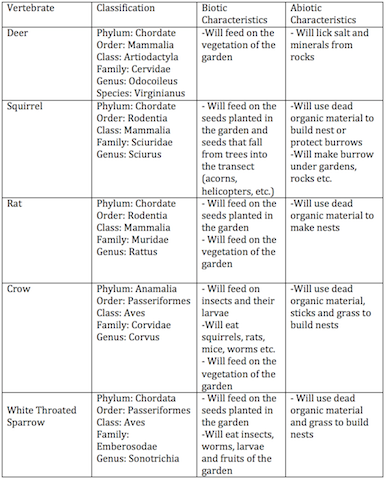
Potential Vertebrates
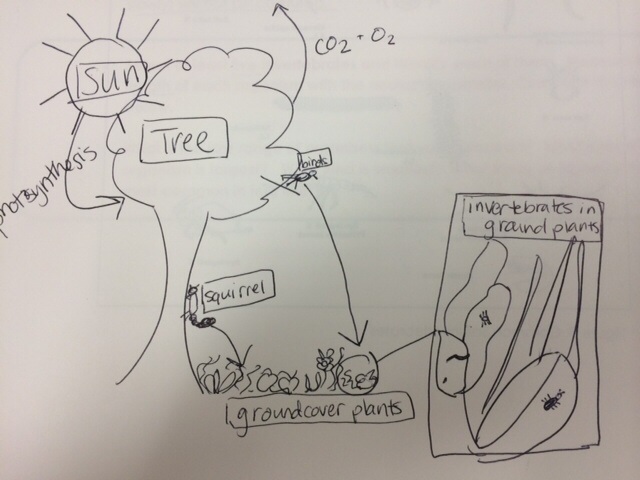
Food Web
CONCLUSION
The purpose of this lab is to understand the importance of Invertebrates. The invertebrates were observed and classified which allowed me to connect the significance of each organism within the transect. Vertebrates rely on invertebrates and organic matter for their sustaining of life, and invertebrates rely on plants, bacteria and fungi. Each element that we have studied throughout the various labs is very significant to the ecosystem of this transect. The fabrication of a food web and speculation of potential vertebrates was a very effective method of increasing my understanding of the importance of invertebrates.
References
Lin, Kevin. "Seasonal Science: What Lurks in the Leaf Litter?." Scientific American Global RSS. N.p., n.d. Web. 2 Mar. 2014. <http://www.scientificamerican.com/article/bring-science-home-leaf-litter-biodiversity/>.
"White-Tailed Deer." White-Tailed Deer. N.p., n.d. Web. 2 Mar. 2014. <http://www.theanimalspot.com/whitetaileddeer.htm>.
"Animals in Colorado." Animals in Colorado. N.p., n.d. Web. 2 Mar. 2014. <http://donerickson.com/animals/animals.html>.
"Birds Directory." : Crow [Kingdom: Animalia, Phylum: Chordata, Class:Aves, Order: Passeriformes, Family: Corvidae, Genus: Corvus]. N.p., n.d. Web. 2 Mar. 2014. <http://birdsdirectory.blogspot.com/2011/06/crow-kingdom-animalia-phylum-chordata.html>.
AF
Plantae and Fungi: Returning to the Transect
Wednesday,February,6
INTRODUCTION
The purpose of this lab is to understand the characteristics and diversity of Plants and to appreciate the function and importance of fungi. This was achieved through collecting and analyzing various plant samples from our transect.
PROCEDURE
1. Bring three Ziploc bags and one pair or gloves and a trowel to the transect
2. Using one bag, the gloves and the trowel, obtain a leaf litter sample. Take the top layer of the soil and the leaf litter and place 500 grams into a Ziploc bag.
3. Take a representative sample from 5 different plants in a way that is minimally damaging. Make sure to represent as many different plants as possible. Build a map of the transect and record where each sample was taken from. Place the samples in a zoploc bag.
4. Collect any seeds, pine cones, flowers, etc. from the plants and bring them back to lab.
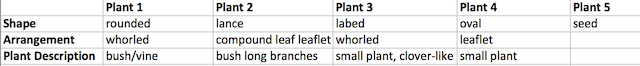
5. Describe the plants that were extracted and where they were found in your notebook and record this data in table 1.
II
1. Observe the moss and lily stem in the lab. Determine the appearance of Xylem and Phloem. Determine the difference between vasucularization between moss (bryophytes) and Lilly (vascularized plants).
2. Observe the extracted plant samples and describe the vascularization of each plant. Record information on Table 1.
3. Describe the physical attributes of each sample. Record this in Table 1.
4. Identify the seeds brought back from transect as either monocot or dicot. Record this information in Table 1.
OBSERVATIONS
Location of samples
Plant Samples
Plant Sample Observations
Fungi Sample
This individual can be identified as a fungus because sac spores (the black dots) can be seen Fungus Sporangia are fruiting bodies that are essential in the process of asexual production of spores. This particular fungus is a mould so it is likely from the Phylum Ascomycetes (sac fungi)
Topsoil Sample
CONCLUSION
The purpose of this lab was to understand the characteristics and diversity of Plants and to appreciate the function and importance of fungi. Plant samples were collected from the transect, they were observed, characterized and categorized. Fungi was also viewed under a dissecting microscope. My understanding of teh diversity and the complexity of plants has definitely broadened and seeing the fungal spores was very interesting as I connected it to what we had learned in class. In a future lab samples could be viewed with a more powerful microscope in order to see the functional components of both plants and fungal spores.
AF
Hay Infusion Observation II
Wednesday,January,30
INTRODUCTION
One week prior to this lab, plates a serial dilution of a sample from the hay infusion culture was prepared so that it could be observed. In lab today the various plates were observed in order to determine the characteristics of bacteria. The plates that contained an antibiotic (tetracycline) were also compared to plates that had no antibiotics in order to observe antibiotic resistance. A PCR preparation was also started in order to illustrate the significance of DNA sequencing in the identification of species.
PROCEDURE
1. Take qualitative observations of Hay Infusion Culture
2. Qualitative observations of the Agar plates.
Gram Stain
1. Label the slides. Use a sterilized loop to obtain a small portion of bacteria. Transfer it to its respective slide.
2. Heat fix the sample of the bacteria to the slide by passing it through a flame three times with the bacterial smear side up
3. Working with a staining tray, cover the smear with crystal violet for 1 minute.
4. Rinse the stain off using a wash bottle filled with water.
5. Cover the smear with Gram's iodine mordant for 1 minute. Rinse gently.
6. Decolorize by flooding the smear with 95% alcohol for 10-20 seconds. Rinse gently. Decolorization has occurred when the solvent flows colorlessly from the slide
7. Cover the smear with safranin stain for 20-30 seconds. Rinse gently.
8. Blot excess water carefully with a paper towel and air dry.
9. After the gram stain is done, focus in on the sample at a low magnification, then observe the slide under 40X and the oil objective. (coverslips are not used with gram stains).
OBSERVATIONS
Hay Infusion Culture
-Water Level Decreased -Decreased amount of material in jar -Increased transparency of water -No film coating the top of the water -Less potent Smell
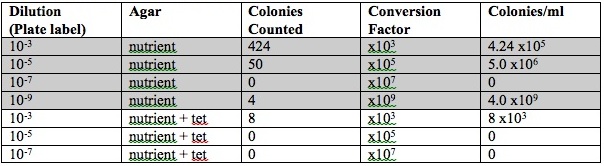
Agar Plates
PICTURES
DISCUSSION
I would not anticipate to find any Archea species growing on the agar plates because these specimen have a preference for extreme environments (ex. extreme halophiles, thermophiles, methanogens etc.). When water has a foul odor this is indicative of the presence of bacteria. Because the smell of the water was particularly potent the first week, and far more fain the second this likely indicates a larger population of bacteria the first week in comparison to the second. A smaller bacteria population would also account for why there was less organic matter seen in the jar the second week. Perhaps the material was consumed and many of the bacteria died due to lack of resources (food). When observing the agar plates it was very noticeable that the presence of tetracycline altered the growth patterns of the bacteria. Plates that only contained nutrient agar had large colonies in comparison to those growing in the presence of tetracycline. Also there seemed to be more bacteria of different color and texture on the strictly agar plates in comparison to the tetracycline containing plates. This indicates that the antibiotic does work to kill bacteria. There were two fungi groups found on the plates. Antibiotics can be used to kill bacteria that will grow fungus but not kill fungus specifically. Antifungals can be used for this purpose. (U.S National Library of Medicine) Tetracycline inhibits protein synthesis of bacteria which causes the death of the organism. A bacteria that is resistant to this antibiotic will develop genes that code for an energy-dependent efflux of tetracyclines, or alternatively be able to create a protein that protects the bacterial ribosomes from the antibiotic. (Chopra 2001)
CONCLUSION
It was seen by observing the hay infusion culture that over time it becomes increasingly difficult for species to survive with very limited resources. It was also seen that antibiotics such as tetracycline greatly limit the growth and are effective in killing many bacteria. The usefulness of the categorization of bacteria based on shape, movement and membrane type was experienced. A future study could compare the use of various antibiotics, and could also compare characteristics of cells that have grown immune to antibiotics.
REFERENCES CITED
"Fungal Infections: MedlinePlus." U.S National Library of Medicine. U.S. National Library of Medicine, n.d. Web. 16 Feb. 2014. <http://www.nlm.nih.gov/medlineplus/fungalinfections.html>.
Chopra, Ian. "Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance." NCBI. N.p., n.d. Web. 16 Feb. 2014. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC99026/>.
AF
Hay Infusion Observation
Wednesday,January,23
INTRODUCTION
In lab today the hay infusion that was created last week was observed. Organisms from the Hay Infusion were observed in order to grasp an understanding of the characteristics of Algae and Protists. A dichotomous key was used to identify/characterize/classify these organisms.
PROCEDURE
1. Qualitative observations were taken (appearance,smell of jar etc.)
2. A micropipette was used to extract a sample of the hay infusion from two different environments (the top and the bottom)
3. The liquid sample was placed on a slide and covered with a cover slip
4. The sample was observed. Observations were recorded.
Procedure II: Serial Dilution (For net weeks lab)
1. Obtain four 10mL test tubes filled with sterile broth
2. Obtain four nutrient agar, and four nutrient agar plus tetracycline plates (make sure they are labelled accordingly)
3. Swirl the Hay Infusion Mixture. Using a micropipette draw 100 microliters of the Hay infusion culture so that it can be transferred to the first 10mL test tube. Swirl the test tube well.
4. Draw 100 microliters from the test tube and place it in the next test tube. Mix well. Continue to do this for the remaining test tubes.
5. Transfer 100 microliters of the solution from test tube one to agar plate. Make sure the mixture is spread well. Do the same for the Agar + Tetracycline plate.
6.Repeat this process for the remaining test tubes.
7. Leave the plates to incubate for one week
OBSERVATIONS
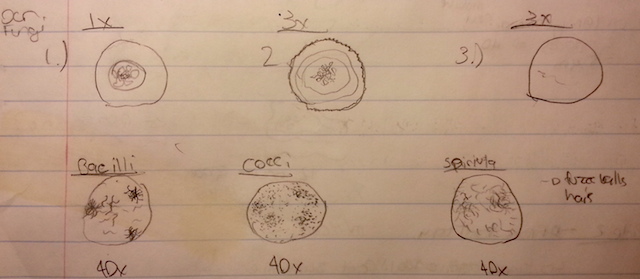
The contents of the jar had a very potent odor. Inside the jar the water was fairly clear with an exception of a brown and green film that coated the top of the water (likely some mold). In the water there were two plants, one that had a thin leafy stalk and one that had a thick white stalk. Organisms near these plants would differ from other organisms because they would likely use the plant as a source of food, oxygen or shelter. There was also a thin layer of sand in the bottom of the jar. A sample was selected from two regions of the jar; the brown film on the top layer of the jar and the bottom of the jar near the leaved plant. Sketches of the organisms observed are located in the third image.
Slide 1-Top Environment
Specimen 1 - A Large brown individual was observed. Did not appear to be mobile. Occupied approximately 10 spaces at 10x (100 um). This cell did not fit the description of any other cell discussed in class.
Specimen 2- An individual observed in very close proximity to the brown individual. Small, mobile, black, vibrating and moving in a circular pattern. Occupied approximately 2 spaces at magnification 40X (5um). This could have possibly been a chlamydomonos cell
Specimen 3- Mobile individual, clear in color. Appeared to have some sort of inner membrane. Occupied 12 ocular spaces at 40x ( 30um)
Slide 2 - Bottom Environment
Specimen 1 - Spherical individual moving quite quickly with flagella. Occupying approximately 62um
Specimen 2 - Very long stick like figure. Occupying approximately 2mm in length
Specimen 3 - Small vibrating spherical organism.
If this hay infusion was observed for another two months I believe that many more organisms would be seen in this environment. I also believe though that with the very limited resources (food, shelter, mates etc.) that natural selection would be very prevalent. The most fit organisms would be those that could find shelter, nutrition and if necessary a mate within their lifetime. It is these individuals that have the greatest probability of reproducing and passing on their genetic information to offspring.
Pictures
Image 1
Image 2
Image 3
CONCLUSION
A future experiment could make a hay infusion that incorporates more organic matter (plants, decaying leaves, etc.) with hopes that this would provide organisms with more means for growth. While observing this hay infusion it was often quite difficult to find organisms. The organisms that were observed were identified/characterized by the dichotomous key.
AF
Transect observation
Wednesday, January, 15
Procedure
1. A transect (20 x 20 space of land on the American University Campus) was assigned to each group.
2. Each group was taken to their transect where they observed the topography, the biotic and the abiotic components of the area.
3. A sample of the transect was then taken using a trowel. A sample that contained some biological life (weeds) was selected.
4. Between 10 and 12 grams of the sample was then placed in a large jar.
5. 500 mL of Deerpark water and 0.1 grams of dried milk were added to the jar
6. The jar was labeled for identification and then shaken for 10 seconds
7. The jar was then left for 1 week to culture
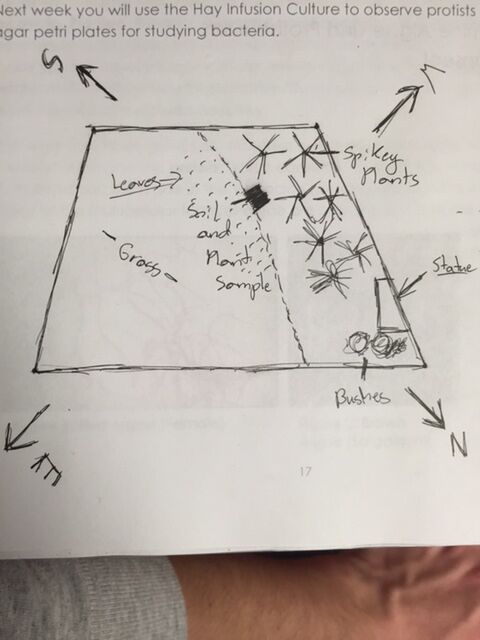
Transect location: AU Community Garden
Biotic Components: Plants, weeds, grass, bugs, grubs. Many larger animals will be in this area during the spring and summer months such as deer and squirrels
Abiotic Components: Wooden planks for boxed gardens, plastic labels, soul, rocks, wood chips. Area not sheltered by trees or buildings so it is exposed to sun, snow, wind and rain.
Topography: Fairly flat. The garden rests at the bottom of one hill and at the top of a large crevasse.
Pictures:
AF