Tissue Engineered Vascular Grafts, by Tyler Vlass and Leonard Brandon
Vascular Grafting Background
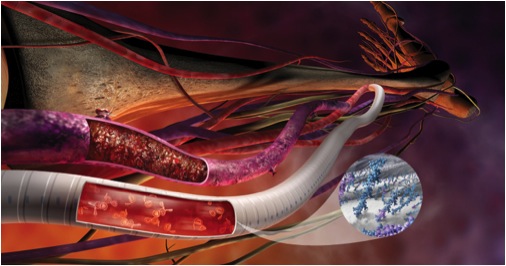
Vascular grafts are used as replacement veins and arteries. These grafts can be manufactured from biomaterials or cultured cells.
Vascular grafts are widely used for vascular access during hemodialysis, a blood filtration technique. During hemodialysis, wastes such as creatinine and urea as well as excess water are separated from the blood, which is then pumped back into the person’s body. Separation is achieved by the use of concentration gradients and semi-permeable membranes. This procedure is used in patients who have acute or chronic kidney failure and is performed 350,000 times annually. [1]
There are three ways of connecting a patient’s blood circulation to a dialysis device. The first method is called an arteriovenous fistula, which is a vein attached to an artery, usually in the forearm. This procedure allows for blood flow to bypass the capillaries, increasing its flowrate. During hemodialysis, a needle is attached through the fistula and waste is removed from the blood. Fistulas are generally the best option for patients and exhibit the least negative side effects. The second method is a venous catheter. This procedure is only temporary, and consists of a tube (catheter) inserted into patient’s larger veins, usually in the neck, chest or leg. The catheter flows two ways and needles are not necessary for access. Catheters often clog, become infected, or cause the veins in which they are placed to become narrow. The final method is
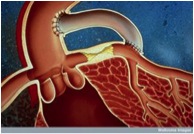
an arteriovenous graft. This graft is similar to a fistula except that it uses an artificial tube placed under the skin to indirectly connect an artery to a vein. This tubing can be used like a vein and punctured repeatedly with a needle for dialysis treatment. These grafts can be used quicker after placement than a fistula, but are more prone to infection and blood clotting (thrombosis). [2]
Another major use for vascular grafts is a coronary artery bypass graft (CABG). CABG is used to treat angina (chest pain) and coronary heart disease (CHD) caused by atherosclerosis. Veins and arteries are usually taken from the patient (autograft) and are used to bypass plaque filled coronary arteries, improving blood circulation to the myocardium (heart muscle). During the procedure, the heart is stopped and the patient is put on cardiopulmonary bypass. Coronary arteries are only suitable for grafting if they are larger than about 1.5 mm. The coronary arteries must also be on top of the heart muscle, not inside it. [13]
History
-1940 – Dr. Willem Kolff filled sausage casings with blood and incubated in a saltwater bath as the first form of dialysis for kidney failure [4]
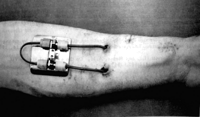
-1943 – Dr. Willem Kolff invents first dialysis machine [4]
-1960 – Scribner Shunt used to connect arteries to veins to allow for ease of connection to dialysis machines [5]
-1970 – Artegraft’s vascular graft approved by the FDA [6]
-1986 –Weinberg and Bell produced cell seeded collagen tubes trying to create novel blood vessels in vivo[7]
Current Tissue Engineered Vascular Grafts
When autografts cannot be used in a patient, replacement vasculature can come from other sources. Grafts can come from other animals (xenografts), cadavers (allografts), or they can be made from synthetic materials, commonly PTFE (Polytetrafluoroethylene). Xenografts and allografts can have serious complications such as aneurism (rupturing of vessels), calcification (hardening of vessels) and thrombosis. Synthetic biomaterials are also prone to these complications and can also change in size due to blood pressure or cell growth inside the graft. Patient specific vascular grafts are the latest in grafting technology and take 6-9 months to produce. These grafts are formed by seeding autologous bone marrow cells onto polymer scaffolds, or by expanding fibroblasts and endothelial cells in a culture. These methods usually produce sheets, which are then rolled around stainless steel into tube structures of varying diameter. The inner cell layer of the tube structure is dehydrated and seeded with endothelial cells from the patient. This procedure can cost upwards of $15,000. [3]
Companies
There are many different kinds of grafts produced by different companies. Some are listed below.
Artegraft – Produces a bovine carotid artery graft (BCA) since 2004. This graft is made from a biological fibrous matrix, providing long-term patency (remains open) and flexibility. Compared with ePTFE (expanded PTFE), BCA grafts exhibit fewer complications. [6]
Cytograft – Produces the LifelineTM vascular graft using sheet-based tissue engineering to culture fibroblast cells, promoting production of collagen. Once enough collagen is produced, the sheets are strong enough to be rolled into varying sized grafts. Growth is finished in a bioreactor. [8]
Bionova – Produces Ominflow II, a biosynthetic vascular graft used as a patch. This graft is produced from cross-linked collagen with a polyester mesh endoskeleton. The graft produced has radial elasticity similar to natural vessels and exhibits excellent biocompatibility, reducing infection and other complications. [9]
Gore – Produces Propaten, a vascular graft that uses the anticoagulant properties of heparin. This graft uses a bioactive surface to anchor heparin to the vasculature. This allows thromboresistance, while retaining the bioactive properties of the graft. [11]
Latest Research
Electrospinning
Electrospinning is a relatively inexpensive way to produce scaffolds for cell seeding. Polymer solution is drawn into a spinneret and charged with large electric potentials. Polymer is then injected through the electric field, and as solvent evaporates fibers are created. Size of the fibers can be manipulated from 50 nm to 10 um by mandrel RPM or solvent concentrations. These nanofibers can be manufactured to regulate the microscopic mechanics of the scaffold, which translates to increased cell differentiation and proliferation. These engineered grafts also exhibit increased functional longevity. Polymers that can be used for this application include: Polylactic acid (PLA), Polyurethane (PU) and polycaprolactone (PCL). Electrospun nanofiber scaffolds can be manufactured into vascular grafts in one step, with living cells being seeded into the fibers as they are made. This method decreases the time needed for biofabrication of patient specific vascular grafts. [12]
Magnetic Force Driven Tissue Engineering
Cells can have magnetic iron oxide nanoparticles attached inside them, allowing them to be driven by magnetic forces to a desirable location. This technique has been used to seed endothelial cells onto PTFE scaffolds for vascular grafts. These cells remain healthy and the magnetic forces used accelerate proliferation, adhesion and monolayer assembly. The cells are also visible through magnetic resonance imaging (MRI), which allows for noninvasive monitoring of vascular grafts.[12]
References
[1] McAllister, Todd. "First Human Use of an Allogeneic Tissue Engineered Vascular Graft." American Heart Association, 27 June 2011. Web. 05 Mar. 2013. <http://my.americanheart.org/idc/groups/ahamah-public/@wcm/@sop/@scon/documents/downloadable/ucm_428738.pdf>.
[2] "Vascular Access for Hemodialysis." National Kidney & Urologic DiseasesInformation Clearinghouse (NKUDIC). US Department of Health and Human Services. Web. 05 Mar. 2013. <http://kidney.niddk.nih.gov/kudiseases/pubs/vascularaccess/>.
[3] Dahl, Shannon L. "Readily Available Tissue-Engineered Vascular Grafts." Science Translational Medicine 3.68 (2011). PubMed. Web. 05 Mar. 2013. <http://stm.sciencemag.org.silk.library.umass.edu/content/3/68/68ra9.full>.
[4] Blakeslee, Sandra. "Willem Kolff, Doctor Who Invented Kidney and Heart Machines, Dies at 97." The New York Times, 12 Feb. 2009. Web. 05 Mar. 2013. <http://www.nytimes.com/2009/02/13/health/13kolff.html?pagewanted=all>.
[5] "Pioneers in Kidney Dialysis: From the Scribner Shunt and the Mini-II to the "One-Button Machine"" Pathbreakers. University of Washington, 1996. Web. 05 Mar. 2013. <http://www.washington.edu/research/pathbreakers/1960c.html>.
[6] Bovine Carotid Artery Graft. Artegraft, 2012. Web. 05 Mar. 2013. <http://www.artegraft.com>.
[7] L'Heureux, Nicholas. "Technology Insight: The Evolution of Tissue-Engineered Vascular Grafts: History of Tissue-engineered Vessels." Nat Clin Pract Cardiovasc Med 4.7 (2007): 389-95. Medscape. Web. 05 Mar. 2013. <http://www.medscape.com/viewarticle/559495_2>.
[8] "Tissue Engineered Blood Vessel (TEBV)." Cytograft. 2009. Web. 05 Mar. 2013. <http://www.cytograft.com/tebv.html>.
[9] "Omniflow II for Vascular Repair." Bionova. Storm Worldwide, 2011. Web. 05 Mar. 2013. <http://www.bionova.com.au/bionova-products/714-2/>.
[10] Roh, Jason D. “Tissue-Engineered Vascular Grafts Transform into mature Blood Vessels via an Inflammation-mediated Process of Vascular Remodeling.” PubMed Central. 9 Mar. 2010. <http://www-ncbi-nlm-nih-gov.silk.library.umass.edu/pmc/articles/PMC2842056/?tool=pubmed>.
[11] "Gore Propaten Vascular Graft." Gore Medical. 2012. Web. 5 Mar. 2013. <http://www.goremedical.com/propaten/>.
[12] Rathore, Animesh, Muriel Cleary, Yuji Naito, Kevin Rocco, and Christopher Breuer. "Development of Tissue Engineered Vascular Grafts and Application of Nanomedicine." WIREs Nanomed Nanobiotechmol 4 (2012): 257-72. Print.
[13] "What is coronary artery bypass grafting?" NIH. Feb. 2012. Web 05 Mar. 2013. <http://www.nhlbi.nih.gov/health/health-topics/topics/cabg/>.
[14] Udelsman, Brooks, Mark Maxfield, and Christopher Breuer. "Tissue Engineering of Blood Vessels in Cardiovascular Disease: Moving towards Clinical Translation." Heart (2013): n. pag. Print.