Tissue Engineered Pancreas, by Sarah Johnson, Alan Mann
Background
Diabetes
Diabetes is a disease in which the body does not properly produce and/or use insulin. Insulin is a hormone that is necessary to convert food, particularly sugar, into energy accessible for the body. There are two main types of diabetes: type 1 and type 2 [23]. Type 1 diabetes is commonly known as juvenile-onset diabetes, and as the name suggests, usually affects children. Those with type 1 diabetes are insulin-dependent, and need a reliable source of insulin in order to survive [23]. It is an immune disorder in which the insulin-producing beta cells in the pancreas are attacked by the body itself, rendering the pancreas incapable of producing enough insulin, which creates a build up of glucose in the blood. This accumulation of glucose wreaks havoc on all organ systems unless the diabetes is treated quickly and properly. Type 1 diabetes cannot be prevented [26]. Type 2 diabetes is caused either by the body’s inability to produce enough insulin or the cells inability to properly use the insulin, often called insulin resistance. This causes a dangerous and damaging accumulation of glucose in the blood. Type 2 is commonly known as adult-onset diabetes and, unlike type 1, may be prevented or delayed with lifestyle changes such as weight loss, healthy diet, and exercise [23]. A staggering 29.1 million people in the US have diabetes, adding up to an estimated $245 billion in annual expenses in 2012. Untreated or mismanaged diabetes may result in blindness, kidney failure, heart disease, stroke, loss of limbs, and of course death [25]. Type 1 and Type 2 diabetes are currently treated with insulin injections via a pump, syringe, or pen. Type 1 patients must administer insulin throughout the day, starting with one or two injections in the morning, but type 2 patients may only need insulin with meals or as necessary. Blood glucose levels must be monitored throughout the day and especially before and after meals. These methods of insulin administration and blood glucose level monitoring are not only inconvenient for the patient, but allow for error that could be deadly [23]. Islet cell transplantation may be beneficial in treating patients with type 1 diabetes, as it replaces or supplements their own insufficient pancreatic cells with healthy ones [15]. The use of an artificial pancreas is mostly studied in patients with type 1 diabetes, but has yet to be tested clinically for long-term use [11].

Insulin Treatment
An artificial pancreas is a system, whether living or non-living, that is mainly used to treat diabetes mellitus by releasing insulin into the bloodstream in response to changing blood glucose levels. Current methods of insulin administration such as insulin injections do not mimic natural insulin release by the pancreas within the body. A healthy pancreas continuously measures changes in blood glucose levels, and releases small amounts of insulin in response to these changes. Current insulin injection methods involve administration of large doses of insulin at a time, leading to imbalances between blood glucose and insulin levels. Thus, a successful continuous “closed-loop” insulin delivery system design will greatly improve current methods of diabetes treatment. Insulin pumps have been a topic of study since the 1970s, and new models are coming closer to achieving natural insulin release[15]. Artificial pancreases have been tested clinically, but further research must be conducted in regards to issues such as successful insertion of the pancreas into the body, safety, and long-term adverse effects. Likewise, islet cell culture therapies have shown some success in diabetes treatment, and remain the primary focus of future studies in diabetes treatment [17].
History
A general overview of the history of diabetes treatments leading up to modern areas of research includes the following:
• 1921-Banting and Best refine insulin from canine pancreas extract, and successfully treat a child dying from diabetes [15].
• 1935-Roger Hinsworth discovers the difference between type 1 and type 2 diabetes [15].
• 1930s to 50s-new types of insulin improve treatments for various types of diabetes [15].
• 1961-The single use syringe is invented, greatly reducing pain caused by injections and eliminating the need to sterilize a needle for injection [15].
• 1969-The first portable glucose meter is created, enabling patients to test their own blood glucose levels [15].
• 1970s-Research in glucose electrode technology begins [20].
• 1979-The first insulin pump design is invented, delivering continuous insulin supply to the patient [15].
• 1990-For the first time, nine patients are treated with human islet transplantation [22].
• 1999-Medtronic MiniMed becomes the first Continuous Glucose Monitoring (CGM) system approved by FDA [20].
• 2000s-Islet transplantations become more common, techniques more researched, insulin pumps become more advanced. Islet encapsulation and 3D modeling improves islet cell cultures and transplantation [14],[21],[20].
Continuous Glucose Monitoring (CGM) Systems
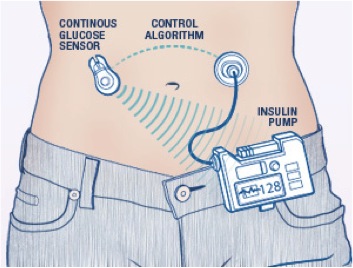
Continuous Glucose Monitoring (CGM) systems have been researched and developed since the 1970s [15], [11]. The vast majority of CGM systems involve the use of a few simple parts [11]. First, a glucose sensor detects changes in blood glucose levels within the body [11]. A transmitter then relays blood glucose information to a receiver [11]. The receiver displays this information on a screen, so that the patient can monitor his or her blood glucose levels continuously [11]. The receiver and screen are part of a device which also contains a cavity filled with insulin, and a tube that is inserted subcutaneously into the patient [26]. The CGM system can be set to a target blood glucose level, and will automatically release the required amount of insulin necessary to maintain the target level of blood glucose [15]. Most CGM systems record blood glucose levels every five minutes automatically [11]. This provides the patient with a better method of continuously monitoring blood glucose levels, since blood glucose monitoring by “finger pricking” is not a feasible method of checking blood glucose levels every five minutes. Many newer models of CGM systems are also equipped with an audible alarm, which sounds if blood glucose levels become too low or too high [24].
Functional Issues
The main functional problems involved with most CGM systems have to do with inaccurate detection of blood glucose levels by the glucose sensor. Blood glucose sensors tend, on average, to over-predict the blood glucose levels of the patient by about 10-16% when in the range of 70 to 180 mg/dL of blood glucose (the healthy “target” range is between 70 and 100 mg/dL) [24]. This presents a considerable risk of hypoglycemia, especially when sleeping or undergoing other long periods of fasting [24]. Additionally, glucose sensors often tend to “lag” when blood glucose levels undergo a rapid change, for example, during the couple of hours after eating a large meal [24], [23]. In this case, blood glucose levels will rise rapidly due to food consumption, and the sensor will under predict the actual blood glucose level of the patient [24], [23]. In order to maintain healthy blood glucose levels, several precautions can be made when using CGM systems. In order to avoid hypoglycemia, patients can adjust the target blood glucose range to a higher level before bed, or before long periods of fasting [24]. This will adjust for error in blood glucose levels recorded by the glucose sensor. Additionally, equipping a CGM with a supply of glucagon has been proposed [24]. Glucagon is a chemical which, when blood glucose levels begin to drop, leads to the depolymerization of glycogen in the liver and the subsequent release of glucose to the body [5]. Thus, it presents a rapid means of delivering glucose in the event that hypoglycemia should occur in the patient. Some studies have shown that to prevent rapid increase in blood glucose levels following a meal (due to the “lag” effect of the glucose sensor) patients can manually inject a small dose of insulin about 15 minutes prior to eating [24]. This will ensure that a higher amount of insulin is being administered to balance out the high amounts of glucose consumed during a mealtime.
Artificial Pancreas System
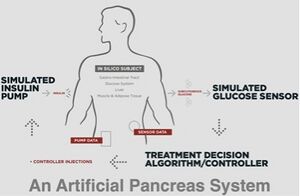
The fully automated, closed-loop artificial pancreas system is a feedback-controlled device with an algorithm that automatically adjusts the rate of insulin delivery by an insulin pump, based on real-time continuous glucose monitoring [25]. The utilization of an implanted artificial pancreas has been shown to decrease the error in inaccurate blood glucose measurement and would work throughout the night to reduce nocturnal hypoglycemia. The MD-Logic is a wireless, fully automated, closed-loop artificial pancreas system that uses algorithms and has a personalized alert system, to ensure safety. The algorithms for alerts integrate data derived from past glucose levels, insulin delivery, and models of insulin pharmacodynamics. Models created using software such as MATLAB and MathWorks monitor current glucose readings to ensure that the artificial pancreas system is working properly and the patient is safe [19]. The Diabetes Wireless Artificial Pancreas Consortium was established to test the MD-Logic system, and overnight youth camps were created to monitor the system in an out-of-the-hospital setting. The study was conducted on 56 type 1 diabetes patients from the ages of 10 to 18. The conclusions of the study demonstrated that on nights when the artificial pancreas was used, as compared to nights when the sensor-augmented insulin pump was used, there were significant decreases in the number of episodes in which the glucose level reached below normal. There were no significant between-treatment differences in the median overnight glucose levels than there were with the senor-augmented pump. There were significant improvements in the many measures of glycemic control and glucose variability with the artificial pancreas, and total overnight insulin doses were significantly higher during nights when the artificial pancreas was used in place of the sensor-augmented pump. Over time, glucose levels remained significantly more stable with the artificial pancreas, and there were fewer false hypoglycemia alarms. Patients experienced adverse effects when using both systems, but fewer of these effects were experienced nocturnally with the use of the artificial pancreas [19]. Regardless of the promising results from this overnight camp study, much more research is necessary before artificial pancreases can be implanted in the body [19].
Islet Cell Culture
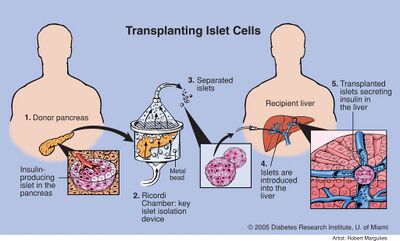
Islets of Langerhans are regions in the pancreas that contain endocrine cells that produce the hormone insulin. Patients with type 1 diabetes may benefit from islet cell transplants because the cause of their diabetes is damaged, inefficient, and/or lack of insulin-producing pancreas cells. Islets of Langerhans are comprised of four different cell types that produce the following hormones: glucagons (α cells), insulin (β cells), somatostatin (δ cells), and pancreatic polypeptide (γ cells). The release of glucagon by α-cells, combined with the release of insulin by β-cells, allows the pancreas to continuously monitor and stabilize blood glucose levels [26]. In an islet transplantation procedure, islet cells are taken from the pancreas of a patient or suitable donor, cultured in a lab, and subsequently transplanted back into the patient, usually to the liver [26], [20]. The transplantation is performed using a simple catheter, which is inserted into the portal vein of the liver [26], [20]. Islet cell aggregates travel into small capillaries throughout the liver, where they become “stuck” due to size limitations [20]. There, the cells act as they normally would in the pancreas, releasing insulin (and glucagon) in response to changing blood glucose levels [26], [20]. Since insulin secreted from the pancreas normally travels to the liver, the liver has been shown to be the most suitable location for islet cell transplantation [20].
Microengineeirng 3D islet models
In vitro research is conventionally conducted in a monolayer cell culture, but with new the development of micro- and nano-technologies, many microengineering methods have been developed to construct three-dimensional islet models. These models better mimic islet cells in the pancreas in vivo. Microengineered in vitro 3D models indicate potential to provide superior experimental platforms to elucidate islet behaviors under physiological and pathological conditions, such as the molecular mechanisms of diabetes and clinical islet transplantation. These new advances in islet cell cultures should ultimately aid researchers to better understand the behavior of real islet cells and recreate the machinery and behavior in engineered islet cells [20].
Islet Encapsulation
Islet encapsulation addresses the problem of immune response rejection of transplanted islet cells. The encapsulation material must meet two important requirements: it must isolate the encapsulated islet cells from the immune system, and it must allow the mass transport of small molecules such as glucose and nutrients into the islets, and of course allow the diffusion of insulin and waste products out of the islets. This encapsulation is designed to limit or eliminate an immunological response to the non-host islet cells. The encapsulation uses an immune-protective biomaterial to create a permselective membrane around a group of islet cells, which is often referred to as a bioartificial pancreas. The membrane allows the islets to regulate blood glucose levels through insulin release but excludes other molecules such as proteins based on their larger size. Several materials, including alginate and ploysulphone have shown promise in effective encapsulation. Alginate is a natural derived from kelp, and is often augmented with materials such as polyethylene glycol (PEG) and ploy-L-lysine (PPL) to increase effectiveness. There are three general methods of encapsulation that have been studied for inslet transplantation: intravascular macrocapsules, extravascular macrocapsules, and microcapsules [15].
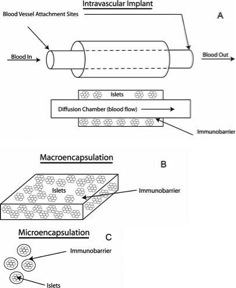
Intravascular implants are perfusion chambers that are directly connected to the vascular system, where blood flows through hollow fibers. These implants allow for adjustable mass-transfer rates but risk damaging blood vessels during implementation. Macroencapsules contain a large mass of islet cells within a diffusion chamber, and are extravascular. They are implanted in the peritoneal cavity and subcutaneously, and are easily implanted and retrieved, but are limited in oxygen diffusion and nutrient transport. Microencapsulation is the spherical encapsulation of single islets, or small groups of islets. Methods of constructing the microcapsules include emulsion, photopolymerization, micro-machined nanoporous microsystems, and electrified coaxial liquid jets. These microcapsules allow for increased oxygen diffusion and nutrient transport, but are difficult to remove if necessary [15].
Complications of Islet Cell Therapy
In a 2005 study involving 65 patients who underwent islet cell transplants, only about 10 percent of the patients were insulin independent after 5 years [26]. A 2006 study showed that after 6 months, over half of the 225 patients involved were insulin independent [26]. At the 2-year mark, however, that number dropped to one-third [26]. Several complications give rise to the above results. The biggest issue facing patients who have received islet cell transplants is an immune response [23], [26]. Research shows that in large animals such as dogs and humans, an immune response to the transplanted cells is the largest cause of loss of cell viability with time [23]. Over the course of a few months, patients may need to return to manual methods of insulin administration. In order to prevent an immune response, several methods have been tested, leading to further complications. Immunosuppressive drugs have been used to prevent an immune response, but have led to other dangerous health complications [23], [26]. Also encapsulation of islet cells in man-made (usually polymer) membranes has been proposed to prevent an immune response [23]. This method, while fairly successful at preventing an immune response, has in the past eventually led to decreased islet cell viability due to poor diffusion of oxygen and other nutrients to the center of the islet cell aggregates [23].
Future Research
The Hanuman Medical Foundation hopes to create an encapsulated islet cell therapy using a macroscopic islet cell “sheet” release insulin in response to changing blood glucose levels [15]. By using a thin sheet to reduce problems with diffusion, Hanuman hopes to provide an islet cell therapy that enables canines to remain insulin independent for longer than an average of 93 days [15]. Bernard, Lin, and Anseth (2012) have produced a microwell cell culture platform that enables β-cells to form 3D aggregates [17]. Upon further study, they have found that these β-cell aggregates provide more natural cell-cell contact, and that this enables a higher percentage of the cells to remain viable over time [17]. Further studies in this area may lead to sustained viability of transplanted islet cell cultures in diabetes patients.
References
[1] American Diabetes Association, “Economic Costs of Diabetes in the U.S. in 2012”, March 6, 2013. Alexandria, VA. Retrieved from [1]
[2] American Diabetes Association, Statistics About Diabetes, June 2012. National Diabetes Statistics Report, 2014. Retrieved from [2]
[3] Beck, Jonathan. Islet Encapsulaion: Strategies to Enhance Islet Cell Functions, 2007. Tissue Engineering, Vol 13, No. 3, 2007, Mary Ann Liebert, Inc. Retrieved from [3]
[4] Bernard, Abigail et al. A Microwell Cell Culture Platform for Aggregation of Pancreatic beta-Cells, 2012. Mary Ann Liebert, Inc., Tissue Engineering: Part C, volume 8, number 8, 2011. Retrieved from [4]
[5] Bowen, R. Glucagon, June 1999. Retrieved from [5]
[6] Cefalu, William T. Tamborlane, William V. The Artificial Pancreas: Are We There Yet?, May 2014. American Diabetes Association, Diabetes Care, volume 37, no. 5, p. 1182-1183, 2014. Retrieved from [6]
[7] Centers for Disease Control and Prevention, 2014 Diabetes Statistics Report, October 24, 2014. Atlanta, GA: National Center for Disease Prevention and Health Promotion, Division of Diabetes Translation. Retrieved from [7]
[8] Diabetic care services, Diabetes Basics: Types of Diabetes…The Causes and Symptoms, 2015. Eastlake, OH: Care Services, Inc. Retrieved from [8]
[9] Diabetes Health, The History of Diabetes. Retrieved from [9]
[10] Diabetes Research Institute Foundation, Downloadable Islet Cell Transplant Images, 2014. Retrieved from [10]
[11] Diabetes.co.uk. Insulin Pumps. Diabetes Digital Media Ltd, 2015. Retrieved from [11]
[12] Dorian, Randy et al., Designing a Definitive Islet Sheet Study, April 2012. San Francisco, CA: Hanuman Medical Foundation, 2015. Retrieved from [12]
[13] The Epsilon Group, Pre-Clinical Assessment of Artificial Pancreas Systems, 2015. Retrieved from [13]
[14] Gao, Bin et al. Engineering of microscale three-dimensional pancreatic islet models in vitro and their biomedical applications, Febrary 2015. London: Informa healthcare, 2015. Retrieved from [14]
[15] Leiva-Hidalgo, Alberto de. From pancreastic extracts to artificial pancreas: History, science and controversies about the discory of the pancreatic and antidiabetic hormone, 2011. Spain: Av Diabetol, volume 27, p. 27-38. Retrieved from [15]
[16] National Diabetes Information Clearinghouse (NDIC). Pancreatic Islet Transplantation, September 2013. National Institute of Diabetes, Publication 13, 2013. Retrieved from [16]
[17] Nazario, Brunilda. Artificial Pancreas on the Horizon, February 2004. MedicineNet.com, Retrieved from [17]
[18] Nichols, Hannah., Medical News Today, Diabetes: What is the difference between type 1 and type 2?, September, 2014. Bexhill-on-Sea, UK: MediLexicon International Ltd. Retrieved from [18]
[19] Phillip, Moshe et al. Nocturnal Glucose Control with an Artificial Pancreas at a Diabetes Camp,February 2013. The New England Journal of Medicine, p. 824-833. Retrieved from [19]
[20] Rajotte, Ray., Senior, Peter. CIHR Video: Dr. Ray Rajotte, PhD & Dr. Peter Senior, MD, PhD, September 2011. Insidermedicine, YouTube. Retrieved from [20]
[21] Steil, G. M. et al., Closed-loop insulin delivery-the path to physiological glucose control, February 2004. Advanced Drug Delivery Reviews, Volume 56, Issue 2, p. 125-144. Retrieved from [21]
[22] Tzakis, Andreas G. Pancreatic islet transplantation after upper abdominal exenteration and liver replacement, August 1990. Lancet, p. 402-405. Retrieved from [22]
[23] Wang, Taylor et al., Successful Allotransplantation of Encapsulated Islets in Pancreatectomized Canines for Diabetic Management Without the Use of Immunosuppression, 2008. Transplantation, volume 85, p. 331-337. Retrieved from content/uploads/2012/03/Wang-et-al.-2008-SuccessfulAllotransplantation-of-Encapsulated-Isl.pdf
[24] [23]
[25] [ http://www.jdrf.org/files/General_Files/Research_ENews/Frontline%203-07.pdf]
[26] [24]
