Synthetic Reaction Diffusion Patterning
Example Patterns
Specifications
| Theoretical | Empirical | |
|---|---|---|
| Pixel Dimension | 100 um X 100 um | |
| Pattern Size | 5 cm X 5 cm | |
| Accuracy | ||
| Precision |
Mechanism
Synthetic biologists use an intercellular diffusive molecule and intracellular reactions to create patterns. These patterns are created based on the French Flag model in which a lawn of identical cells differentiate based on the concentration of local signal observed. I have not found any projects that have tried to create Turing patterns in which at minimum 2 diffusive molecules are used: one short range activator, and one long range inhibitor. Most patterns are made up of Escherichia coli and use acyl-homoserine lactone (AHL) as the diffusive molecule(1). There have been two classes of patterns made. I call one class pictures because they are time-invariant patterns while the other class I call animations because they are patterns that vary over time.
Pictures:
The first synthetic pattern was created in 2005(2). In this project, sender cells sending AHL were seeded in the middle of a plate while receiver cells were seeded everywhere else. The receiver cells held a genetic circuit such that upon a low concentration of AHL, GFP expression is repressed and upon a high concentration of AHL, GFP expression is repressed. Only at intermediate concentrations of AHL is GFP expressed. This band-pass filter created a three-ring pattern. A similar work was published a few years later with maybe a small improvement in resolution(3). A third project created a high-pass filter rather than a band-pass filter(4).
Tabor et al. used synthetic reaction diffusion signaling in a clever way to make a genetic edge detector. In essence, all cells emit AHL(5). Once light hits cells in the plate, they become receiver cells and these receiver cells carry a high-pass filter. Thus, only cells that have been hit with light and that receive a high concentration of AHL (those at the edge) produce lacZ.
Animations:
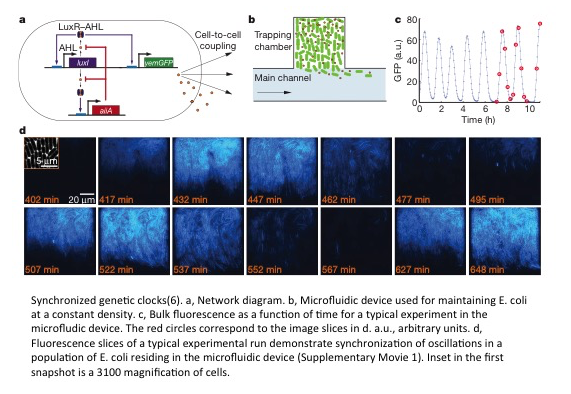
Synthetic biologists have created systems that oscillate signal in a synchronized fashion. The first system was made by Danino et al(6). They oscillated signal of cells by using an AHL quencher that is up regulated by the presence of AHL and they synchronized oscillations using AHL. Prindle et al. later built off of this work and synchronized cells across a 1 cm X 2 cm device using the gaseous diffusive molecule hydrogen peroxide along with AHL(7). The device they made is an arsenic sensor with a visual display! Chen et al. was able to synchronize the oscillations of an activator population and a repressor population using two different AHL molecules(8). I wonder how the synchrony compares with the Danino et al. oscillations where they essentially have two activating populations.
Reference/Resources
- W. Bacchus, M. Fussenegger, Engineering of synthetic intercellular communication systems. Metab. Eng. 16, 33–41 (2013).
- S. Basu, Y. Gerchman, C. H. Collins, F. H. Arnold, R. Weiss, A synthetic multicellular system for programmed pattern formation. Nature (2005).
- T. Sohka et al., An externally tunable bacterial band-pass filter. Proc. Natl. Acad. Sci. U.S.A. 106, 10135–10140 (2009).
- K. Brenner, D. K. Karig, R. Weiss, F. H. Arnold, Engineered bidirectional communication mediates a consensus in a microbial biofilm consortium. Proceedings of the National Academy of Sciences. 104, 17300–17304 (2007).
- J. J. Tabor et al., A Synthetic Genetic Edge Detection Program. Cell. 137, 1272–1281 (2009).
- T. Danino, O. Mondragón-Palomino, L. Tsimring, J. Hasty, A synchronized quorum of genetic clocks. Nature. 463, 326–330 (2010).
- A. Prindle et al., A sensing array of radically coupled genetic “biopixels.” Nature. 481, 39–44 (2011).
- Y. Chen, J. K. Kim, A. J. Hirning, K. Josić, M. R. Bennett, Emergent genetic oscillations in a synthetic microbial consortium. Science. 349, 986–989 (2015).