Summer 2006 Harvard iGEM work
6/12/06
Incubated DNA nanostructure folding interactions. DNA scaffold and oligonucleotides designed by Shawn Douglas. Protocol available at http://openwetware.org/wiki/IGEM:Harvard/2006/Folding_DNA_nanostructures. Included 2 negative controls, one with water instead of scaffold, one with water instead of oligonucleotides.
Transformed R0010, E0241, and E7104 parts into 15 ul aliquots of chemically competent OneShot Top10 cells.
6/13/06
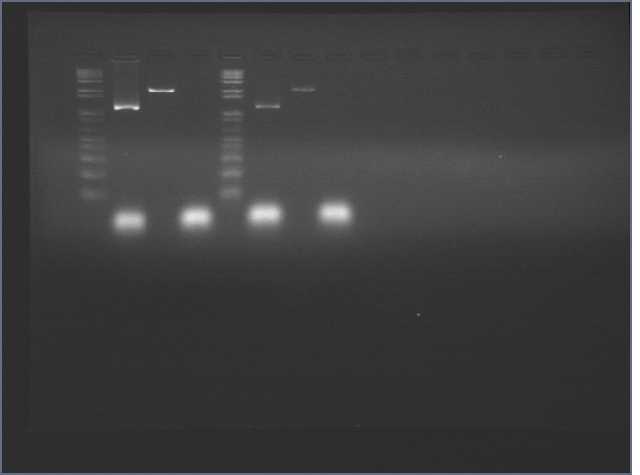
Ran gels from nanostructure incubations yesterday. 2% agarose gel, 130V, 45min. Loaded 10ul sample + 1ul dye, and 10ul 1kb ladder.
Lane 5: 1 kb ladder
Lane 6: DNA nanostructure (+)
Lane 7: (-) control with no oligonucleotides
Lane 8: (-) control with no DNA scaffold
Prepared 2 liquid cultures from each transformation, 5ml LB + 50ul of 5mg/ml ampicillin, shaken at 180rpm and 37dC overnight.
6/14/06
Retrieved liquid cultures (3x2). I accidentally dropped culture #2 from R0010 transf and spilled half the volume. Set aside 1ml of each culture for glycerol stock. Performed miniprep on remaining culture. Eluted with 50ul water.
Prepared digest reactions with 8ul of both minipreps of R0010 (SpeI/PstI) and of E0241 (XbaI/PstI). Incubated 1 hour at 37dC held at 4dC, then incubated at 80dC for 20 min.
Protocols
Gel electrophoresis
For x% gel, mix x grams Ultrapure agarose in 100ml 1X TBE in a plastic flask, heat by microwave and swirl with top screwed on loosely until clear, cool, added with 3ul (with gloves), pour into gel frame, let set for 20 min. Place into gel running box and submerge barely with 1X TBE. Load wells, and run gel at appropriate voltage and time.
Loading dye is BTB dye 50% glycerol + 10X TBE. Dye has been added to 1X 1kb ladder at 1:11 ratio.
Transformation
Chemically competent OneShot Top10 cells. Let cells thaw on ice. Add appropriate amount of DNA, tap gently to mix, and let sit on ice for 20 minutes. Heat shock for 30 sec at 42dC; let cool on ice for 2 min. Add 200ul SOC media, and shake for 1 hour at 37dC. Pipet onto agar plate treated with appropriate drug, and spread. Leave in 37dC overnight with agar side up.
Miniprep
http://openwetware.org/wiki/Miniprep/Qiagen_kit
Digest
x ul DNA
10% by volume buffer (10X, choose buffer best for enzymes used)
1% BSA (100X)
2% enzyme 1
2% enzyme 2
y ul water (to fill to desired mixture volume)