Griffin:Antibody Basics: Difference between revisions
No edit summary |
No edit summary |
||
| Line 3: | Line 3: | ||
[[Image:antibody1.jpg|thumb|right|The basic structure of the heterotetrameric, disfulfide linked, Immunoglobulin gamma (IgG)]] | [[Image:antibody1.jpg|thumb|right|The basic structure of the heterotetrameric, disfulfide linked, Immunoglobulin gamma (IgG)]] | ||
[http://en.wikipedia.org/wiki/Antibody Antibodies] are B cell (lymphocyte) derived proteins produced by the immune system in order to address the immunological needs of a mammalian system over time. The most common type of research antibody is the IgG that | [http://en.wikipedia.org/wiki/Antibody Antibodies] are B cell (lymphocyte) derived proteins produced by the immune system in order to address the immunological needs of a mammalian system over time. The most common type of research antibody is the IgG that contains 2 heavy chains (~50 kDa each) and 2 light chains (~25 kDa each). The general structure of all antibodies is very similar, a small region at the tip of the protein is extremely variable, allowing millions of antibodies with slightly different tip structures to exist. This region is known as the hypervariable region (Fab). Each of these variants can bind to a different target, known as an antigen. This huge diversity of antibodies allows the immune system to recognize an equally wide diversity of antigens. The unique part of the antigen recognized by an antibody is called an epitope. These epitopes bind with their antibody in a highly specific interaction. | ||
===Fc fragment=== | ===Fc fragment=== | ||
Revision as of 22:22, 12 August 2009
Antibody
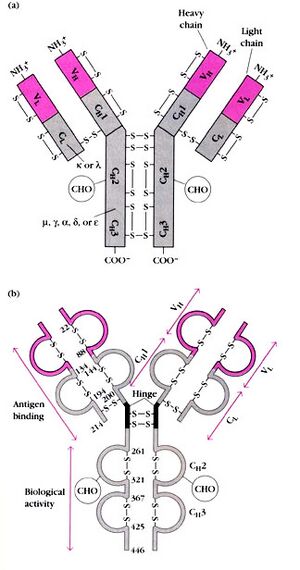
Antibodies are B cell (lymphocyte) derived proteins produced by the immune system in order to address the immunological needs of a mammalian system over time. The most common type of research antibody is the IgG that contains 2 heavy chains (~50 kDa each) and 2 light chains (~25 kDa each). The general structure of all antibodies is very similar, a small region at the tip of the protein is extremely variable, allowing millions of antibodies with slightly different tip structures to exist. This region is known as the hypervariable region (Fab). Each of these variants can bind to a different target, known as an antigen. This huge diversity of antibodies allows the immune system to recognize an equally wide diversity of antigens. The unique part of the antigen recognized by an antibody is called an epitope. These epitopes bind with their antibody in a highly specific interaction.
Fc fragment
The 'F' stands for fragment and the 'c' stands for crystallizable or constant. The Fc portion of the antibody is limited in variability and is responsible for the biological activity of the antibody (as opposed to antigen binding). The Fc portion varies between antibody classes (and subclasses) but is identical within that class. The C-terminal end of the heavy chains form the Fc region. The Fc region plays an important role as a receptor binding portion. The Fc portion of antibodies will bind to Fc receptors in two different ways. For example, after IgG and IgM bind to a pathogen by their Fab portion their Fc portions can bind to receptors on phagocytic cells (like macrophages) inducing phagocytosis.
Fab fragment
The 'F' stands for fragment and the 'ab' stands for antigen binding. This is the region where antigen binding takes place. The Fab portion of an antibody is made up of the N-terminal domains of both the heavy and light chains. This domain of the antibody is called the variable (V) domain (VH on the heavy chain, VL on the light chain) because there is such high variability between antibodies in this area. This allows recognition of thousands of different antigens.
Primary antibody production
Immunizing Antigen
A protein is an important element of the cellular world. A fragment of a protein of interest is chosen to develop an immunizing antigen.
Common types of immunizing antigens include;
15-25 amino acid synthetic peptide
Native proteins have traditionally been used to raise antibodies and for some applications there are advantages in using antibodies against the native proteins. However, there are also distinct advantages of using peptides over native proteins and it is for these reasons that peptides are now becoming more commonly used in antibody production.
- Only the amino acid sequence is necessary - This is important under the following circumstances:
The protein has not yet been discovered, but the gene sequence is known, a new protein has been discovered, but only a partial sequence is known, the protein is expressed in very low amount or not available in sufficient quantity, or the protein cannot be successfully expressed by recombinant techniques.
- Enhanced specificity of the antibody against the protein – This offers the following benefits:
Eliminates or minimizes potential cross-reactivity between structurally homologous proteins, enables the production of different antibodies for different epitopes of the same protein, enables the production of antibodies for proteins with post-translational modifications.
- Convenient and cost-effective purification using the peptide affinity column
Avoids the cost and potential problems of immobilizing whole proteins into affinity columns, eliminates or minimizes conformational and stability problems associated with immobilized proteins, enhances antibody purification since only one peptide ligand is used for affinity binding.
- Conjugation at the N- or C-terminus presents the longest uninterrupted peptide sequence and allows the peptide to assume the correct conformation.
- C-terminal antigen should be coupled to the carrier protein through the N-terminus and, conversely, the N-terminal antigen should be coupled through the C-terminus.
50-300 amino acid purified protein
- E. Coli transformed (Prokaryotic) and purified recombinant protein
- Baculovirus infected SF9 cell (Eukaryotic) derived purified protein
cell culture/in vivo derived enriched fraction
- membrane fractions
- tumor antigens
Carrier Protein
Peptides are designed to be good epitopes by themselves; however they fail to elicit a strong antibody response. The reason for this is that the epitopes are able to bind to the B-cells, but are too small to contain additional motifs required to associate with class II molecules and T-cell receptors. The carrier proteins, once coupled to the peptides, provide these additional motifs and generate a strong antibody response.
- Carrier proteins provide a large number of functional moieties (amino, carboxyl, and sulfhydrul groups) that serve as "anchor points" for peptide conjugation.
- The most common carrier proteins are bovine serum albumin (BSA; 67kDa) and keyhole limpet hemocyanin (KLH; 4.5x105-1.3x107Da). Common examples also include ovalbumin (OVA), rabbit serum albumin (RSA), or bovine thyroglobulin (THY).
Adjuvant
The term “adjuvant” originates from the Latin word adjuvare, which means to help or aid. An immunologic adjuvant is defined as any substance that acts to accelerate, prolong, or enhance antigen-specific immune responses when used in combination with specific vaccine antigens.
Adjuvants in immunology are often used to modify or augment the effects of a vaccine by stimulating the immune system to respond to the vaccine more vigorously, and thus providing increased immunity to a particular disease. The carrier protein linked antigen is mixed with the adjuvant prior to immunizing the host animal.
Freund's adjuvant is an antigen solution emulsified in mineral oil, used as an immunopotentiator (booster of the immune system). The so-called complete form (CFA) is composed of inactivated and dried mycobacteria, usually Mycobacterium tuberculosis (the pathogenic agent of tuberculosis). The so-called incomplete form (IFA) is the same adjuvant, but without the mycobacterial components.
DNA Vaccines Methods and Protocols ISBN 978-0-89603-580-5 (Print) 978-1-59259-688-1 (Online)
Animal hosts
Animal health and well-being during the production of antibodies is of the utmost importance since the isolation of blood and purification of clean antibodies depends on a healthy animal.
Antibody production depends on the humoral immune response of the host animal to produce antibodies toward the antigen much in the same way that a human flu vaccine elicits antibody production in humans. Under both circumstances the immune system of the animal is challenged with an antigen that the body recognizes as foreign and the immune system goes to work.
Monoclonal antibody production
Monoclonal antibodies are produced from a single clone of a B-cell and are derived from a single genetic code. These homogeneous populations of highly specific antibodies recognize only a single epitope and will not recognize other epitopes that may be present in the same antigen.
single epitope=high specificity, low avidity
- Smaller animal conserves materials since the animal eventually gets sacrificed. Mouse or hamster is immunized over time, then the spleen is harvested from the sacrificed animal. Ig producing B Cells from the spleen are immortalized by fusing with a myeloma cell in culture; the B Cell/myeloma fusion is known as a hybridoma and secretes a single clone type of Ig.
- Monoclonals detect a single epitope; hence mono-clonal. High specificity, low avidity.
Ascites
Ascitic fluid (also called ascites) is an intraperitoneal fluid extracted from mice that have developed a peritoneal tumor. For antibody production, the tumor is induced by injecting hybridoma cells into the peritoneum, which serves as a growth chamber for the cells. The hybridoma cells grow to high densities and continue to secrete the antibody of interest, thus creating a high-titered solution of antibodies for collection. Antibody concentrations will typically range between 1 and 10 mg/ml.
Polyclonal antibody production
Polyclonal antibodies are mixtures of antibodies generated from more than one clone of B-cells as a response to specific antigen(s) when an animal is immunized. The antibodies are isolated from the serum as a group, and each antibody recognizes a different epitope of the antigen(s).
multiple epitopes=high avidity, high sensitivity
- Larger animals produce more blood. Goat or rabbit is immunized over time and blood is drawn on a routine basis. Serum fraction of the blood undergoes purification by affinity column or protein A/G column. The total circulating IgG derived from humoral immunity is isolated from the animal blood and recognizes many parts of the antigen.
- Polyclonals detect several epitopes; hence poly-clonal. High avidity means a polyvalent reagent; may be specific, although higher chance of sensitivity.
Common animal types



Rabbit
A large & unique animal with enough blood to draw and isolate total polyclonal IgG. Also suitable for spleen fusions (immortalization) to produce monoclonal Ig. Rabbits have a unique immune system. During T cell-dependent immune responses in mouse and human, Ig genes diversify by somatic hypermutation within germinal centers. Rabbits, in addition to using somatic hypermutation to diversify their IgH genes, use a somatic gene conversion-like mechanism, which involves homologous recombination between upstream VH gene segments and the rearranged VDJ genes.
Goat
A large & hearty, herding ungulate with enough blood to draw and isolate total polyclonal IgG. The immune response typically takes longer to elicit a sufficient titer for purification relative to a rabbit, however the yields can be much higher and the goat is a hearty animal.
Rodent (mouse, rat, hamster)
Small mammal that requires less antigen than the rabbit or goat. Suitable for spleenectomy and B cell fusion to produce monoclonal Ig.
Chicken
A unique non-mammal source for research antibodies, polyclonal IgY from yolk sack is considered more humane than drawing blood. However success has been limited.
Monoclonal Antibody purification
Hybridoma cell cutlure supernatant
Centrifuge cultured medium at 1000 rpm for 5 minutes, collect supernatant into sterile containers, if necessary add sodium azide up to 0.2%. for several month storage, keep supernatant at 4C, otherwise, -20C.
Ascites fluid
Hybridoma cell lines grown as ascites tumors in mice will frequently yield milligram quantities of monoclonal antibody per milliliter of ascites fluid.
Protein G purification
Protein G can be used for the isolation of IgG from serum, ascites, or hybridoma supernatants. Although this specific protocol is geared for large volumes of hybridoma supernatants, the buffers and principles are applicable to other sources of IgG. Most volumes of supernatant can be readily handled using 5 ml of Protein G-Sepharose. This can cost several hundred dollars but can be used for years as long as the column is cleaned and re-equilibrated properly. Important considerations:
- Concentration/purification of antibody is typically performed by affinity chromatography using Protein A- or Protein G-Sepharose beads. These proteins bind to the Fc region of many mammalian species but only for IgG. Therefore, if your hybridoma secretes IgA or IgM this method will not be applicable for purification or concentration.
- While Protein A and Protein G both bind the IgG Fc of many mammalian species, Protein A does not bind very well to the IgG1 subclass of mice. Therefore, to avoid any potential problems it is best to use Protein G.
- Affinity purification of IgG from tissue culture supernatant will co-purify bovine IgG from fetal calf serum. Therefore, if purity is important it is best to use low IgG fetal calf serum or even serum-free media to grow your hybridomas. Serum-free media is also practical since high protein concentrations can make filtering tedious.
Solutions:
- 20 mM Phosphate Buffer, pH 8.0
- 0.05 M NaAcetate, pH 3.0
- 1.0 M NaAcetate, pH 2.5
- 1.0 M Tris Base, pH 10.0
- 20 mM Phosphate Buffer, pH 8.0; 0.05% NaN3
Preparation of cell supernatant:
1. Remove flasks or roller bottles from incubator and pellet the hybridoma cells by centrifugation at 5000g for 15 min.
2. Transfer the supernatant to a clean container and pre-filter using a fluted funnel filter.
3. Filter supernatant through 0.8 micron bottle top filter. It is critical to place a pre-filter disc on the 0.8 micron membrane to delay clogging as long as possible. If flow stops, discontinue the vacuum and replace the pre-filter disc. Re-apply the vacuum and complete filtering.
4. Repeat using a 0.45 micron bottle top filter (remember to use pre-filter discs).
5. Store supernatant at 4°C until ready for protein G chromatography.
Affinity purification of lgG:
1. Wash a 5 ml protein G-Sepharose column with 30 ml PBS.
2. Apply the cell supernatant to the column by setting up a reservoir to allow for continuous flow. Remember slower is better. Shoot for one drop every five seconds. Collect and save all the effluent in case a second application is desired. The reservoir can be as large as several liters. The volume is only limited by the potential saturation of the Protein G column. For example, 1 ml of Protein G Agarose from Gibco-BRL (Rockville, MD) can bind up to 18 mg of human IgG. Therefore a 5 ml gel bed will bind between 90 - 100 mg IgG. Even at the high end of antibody concentration, a 5 ml column should easily handle one liter of hybridoma supernatant.
3. When all the media has passed over the column, wash with excess PBS (about 30-40 ml).
4. Prepare tubes for collecting the eluted IgG. Set up 10 boroscilicate glass tubes. Place 25 µL Tris solution into tube #l and 70 µL Tris solution into tubes #2-10.
5. Elute the lgG by adding 20 ml 0.05 M NaAcetate, pH 3.0 to the column. Immediately begin to collect the drops in your glass tubes. Collect 1 ml in tube #l and then 2 ml into each of tubes 2-10.
6. Strip the column by applying 20 ml of 1.0 ml I M Na Acetate, pH 2.5 (collect into a waste container).
7. Re-equilibrate the column in PBS with sodium azide.
Polyclonal purification
Peptide antigen affinity column
Protein A or G affinity column
Polyethylene Glycol Precipitation
Hybridoma supernatants precipitation with polyethylene glycol 6000 (PEG 6000) and finally reprecipitated using an ammonium sulphate procedure. The PEG 6000 treatment causes a density change in the ammonium sulphate immunoglobulin precipitate, and resulted in the formation of a pellicle which contained pure mouse monoclonal antibody. The protocol removed contaminating bovine serum immunoglobulin as well as other serum and cellular protein from the monoclonal antibody preparations.
- Brooks DA, Bradford TM, and Hopwood JJ. An improved method for the purification of IgG monoclonal antibodies from culture supernatants. J Immunol Methods. 1992 Oct 19;155(1):129-32. DOI:10.1016/0022-1759(92)90279-3 |
Ultrafiltration
Monoclonal antibodies (mAB) continue to gain importance as therapeutic and diagnostic agents for many types of cancer. The process of screening hybridoma libraries for candidate mABs is both time consuming and labor intensive. Once a hybridoma cell line expressing a suitable mAB is established, a bench-scale purification methodology (e.g. 50-500 mL) must be developed to produce sufficient mAB for further characterization. A traditional method for purifying mABs involves clarification of the hybridoma supernatant by centrifugation, followed by an ammonium sulphate precipitation to concentrate the mABs. The precipitate is then recovered by centrifugation, resolubilized and desalted using dialysis. After these steps, the mAB is further purified using Protein A/G affinity chromatography. The purified antibody is desalted and exchanged into a biological buffer using dialysis. The entire process typically requires several days to complete and can be particularly onerous if multiple mABs are to be evaluated in parallel.
We describe a new and simplified method that minimizes the processing time to less than a day to obtain pure mAB. The method involves clarification of the hybridoma supernatant by microfiltration, followed by concentration using ultrafiltration (Saha K, et al, 1992). The mAB is further purified on Protein A/G beads. The purified mAB is desalted and buffer-exchanged using ultrafiltration. This approach was successfully used to purify an anti c-myc antibody secreted by the hybridoma clone 9E10 (Evan G, et al., 1985). The mAB purified by this new method performed comparably to the commercially purified mAB in downstream applications such as western blotting and ELISA.
The data demonstrate that the new protocol is robust and delivers mAB of a high purity and yield as compared to the traditionally purified mAB. The application of ultrafiltration to mAB purification will be of considerable value to any researcher interested in screening hybridoma libraries and accelerating the purification of mABs.
Validation of Specificity
Validation primary antibody specificity is an ongoing topic of the utmost importance to scientists who choose to utilize immunoglobulin-based approaches.
Peptide neutralization
The disappearance of staining in the presence of blocking peptide, i.e. preincubation of the antigen against which the antibody was raised with the antibody in theory should saturate antigen binding fragments and thereby no staining should be observed.
Knockout animals
Disappearance of staining in knock-out animals for the target protein is particularly useful for the validation of immunohistochemical studies.
RNAi
Reduction of staining by knock-down approaches utlizing siRNA, shRNA, Lentivirus. Further validation of the knockdown approach would employ RT-PCR.
In Situ hybridization
Comparative labeling
Comparison of staining in immunoblots or immunocytochemistry for the target protein vs. related subtypes when expressed in the same cell line. The idea that similar gene class should comparative stain similar structures in systems.
Epitope comparison
Comparing antibodies raised against multiple distinct epitopes of a gene product may yield similar staining or banding patterns. However epitope detection may also be under the influence of steric factors (cis or trans) that could influence the hydrostatic access of the antigen.