EDTA: Difference between revisions
From OpenWetWare
Jump to navigationJump to search
illustration |
illustration |
||
| Line 1: | Line 1: | ||
[[Image:EDTA.png|right|thumb|200px|chemical structure of '''EDTA''' - ethylene-diamine-tetraacetic acid]] | [[Image:EDTA.png|right|thumb|200px|chemical structure of '''EDTA''' - ethylene-diamine-tetraacetic acid]] | ||
[[Image:EDTA with ligand.png|right|thumb|200px|'''EDTA''' (black) with coordinate bonds to a metal ion (red)]] | |||
'''EDTA''' stands for '''ethylene-diamine-tetraacetic acid'''. It chelates divalent cations and is therefore used in many buffers. Its relative [[EGTA]] has a higher affinity for calcium than EDTA. | '''EDTA''' stands for '''ethylene-diamine-tetraacetic acid'''. It chelates divalent cations and is therefore used in many buffers. Its relative [[EGTA]] has a higher affinity for calcium than EDTA. | ||
Revision as of 15:23, 15 June 2009
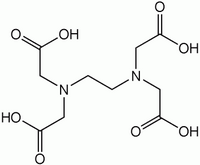
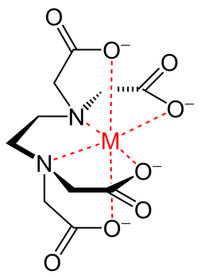
EDTA stands for ethylene-diamine-tetraacetic acid. It chelates divalent cations and is therefore used in many buffers. Its relative EGTA has a higher affinity for calcium than EDTA.
Purpose
- EDTA is a synthetic amino acid and chelating agent for divalent metals.
- Most enzymes that synthesize or modify nucleic acids (e.g. polymerases, ligases, kinases, nucleases) are Mg2+-dependent. The addition of EDTA is a convenient way to stop these reactions.
- EDTA is a component of many buffers used to store DNA, such as TE buffer, where the EDTA removes the metal cofactors (typically Mg++ required for activity of DNAses and other DNA damaging enzymes.
Procurement
- Usually sold as the disodium salt (CAS 6381-92-6) (Sigma E1644).
Use
- Molecular Weight, disodium salt dihydrate: 372.24
- EDTA is essentially insoluble in water, and will only dissolve when neutralized with sodium hydroxide to a pH = 8.0
- Standard stock solutions are 0.5M at pH 8.0. A 1M solution cannot be made. Autoclave.
- Making the standard solution from disodium EDTA (the typical form sold) requires approximately 1 molar equivalent of NaOH
- pK1 = 1.99
- pK2 = 2.67
- pK3 = 6.16
- pK4 = 10.26
EDTA stock solution recipes
0.5 M EDTA stock
- 16.81 g EDTA (Sodium Salt)
- dH2O to 90 ml
- adjust pH to 7.0
- adjust volume to 100 ml
0.5M 500ml pH 8.0 with NaOH pre-calculated
- 93.05g of Na2.EDTA (FW 372.2)
- 10.14g of NaOH (FW 40)
- 500 ml dH2O
Storage and stability
- store at room temperature
- stable for years
Interesting facts
- Here's an article that speaks of the wonders of EDTA and how it can help treat Aunt Edna's cardiovascular disease, enhance your enjoyment of the Friday night happy hour, and act as a "get out of jail free" card.
- EDTA can prolong the life of your motorcycle battery!
- EDTA helps you determine the hidden contents of your hard water.
Safety
?
References
- Here's a great page about EDTA, including formation constant (Kf) values for metal-EDTA complexes. Note that many heavy metal ions (like Fe3+, Co2+, and Zn2+) are chelated much more strongly than Mg2+. A little bit of EDTA in your reaction will go a long way to keep these evildoers out of trouble and away from your precious biomolecules without interfering with your Mg2+-dependent reactions.