Shreffler:Arah2 tetramers
Overview
This protocol is adapted from those published on the NIH tetramer core website with input from Jim Moon here at CIID
Materials
Equipment
- test
Reagents
- test
Procedure
Biotinylation Assay
Background
To gauge whether SA is in excess. Can be used to modify step below. Non-reducing gel required to determine uncoordinated Arah2 and SA by MW
Procedure
- Add 5 µl of the Arah2-biotin monomer sample (0.5 mg/ml) to 12 µl of 1X PBS
- Label three Eppendorf tubes as "2X", “1X”, and "-" and add 5 µl of the Arah2-biotin monomer preparation.
- Add 5 µl of 0.8-1mg/ml Streptavidin to the tube labeled "2X".
- Add 2.5 µl of 0.8-1mg/ml Streptavidin and 2.5 µl of ddH2O to the tube labeled “1X”
- Add 5 µl of ddH2O to the tube labeled "-".
- Mix completely but gently.
- Incubate samples at room temperature for one hour.
- Add 10 µl of 2X loading buffer (non-reducing) to each sample.
- Do not boil or add DTT to the samples.
- Prepare and run samples on a 12% SDS-page gel.
- Stain the gel with 20% acetic acid and 0.1% Commassie blue in 100% methanol.
- Destain the gel using 40% EtOH and 10% acetic acid.
Tetramerization
Background
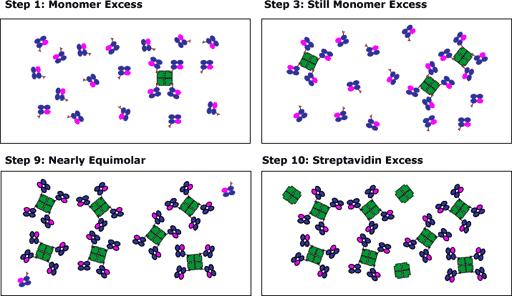
The following information is a generalization of how much labeled streptavidin to add to the biotinylated monomers at the last stage of the tetramer preparation. The actual amount of streptavidin to be added depends both on the molecular weight of the monomer as well as the percent biotinylation. The protocol above should help give a gross empiric measure of whether SA excess is reached.
The procedure of adding the streptavidin over 10 time intervals is designed to maximize tetramer formation. At early additions, the biotinylated monomer will be in excess and will saturate all of the avidin. At the conclusion of the streptavidin addition, there may be an excess of avidin, however, the assumption is that most of the monomer will have gone into tetramers. If the streptavidin was added at one time and was in excess, the resulting solution would be more likely to exist as a mixture of monomers/dimers/trimers.
Comments on stretpavidin addition protocol
- After step 1, biotinylated monomer is present in vast excess, and all the biotin binding sites on the added streptavidin are occupied with monomer
- After step 2, biotinylated monomer is still present in excess, and all the biotin binding sites on the added streptavidin are occupied with monomer.
- After step 9, the concentrations of biotinylated monomer and biotin binding sites on the streptavidin are nearly identical. Note that for this example, we have consciously overestimated the concentration of biotinylated monomer. Normally, we try to shoot for "equimolarity" at step 10.
- The addition of streptavidin in steps 10 adds excess streptavidin, which remains free. The excess streptavidin will not bind to cells when used in staining reactions. Of course, it could increase the staining background, but it won't be in vast excess, so its contribution to the background noise will be minimal.
Theory
Our standard storage conditions for biotinylated monomer stocks are 100 µl at 2 mg/ml, for a total of 200 µg of protein. For the purposes of calculation of how much streptavidin to add, we usually assume 100% biotinylation. Even if we are wrong, the worst that can happen is that you'll waste a bit of streptavidin; it probably won't add much to the background. We exclusively use streptavidin-PE and streptavidin-APC from Molecular Probes. These are sold at a concentration of 1 mg/ml, which refers to the total weight of the conjugate. We assume a 1:1 ratio of streptavidin to phycobiliprotein, and we have calculated the concentration of biotin binding sites in these stocks. This is an estimate, but the addition protocol is devised to provide good results even if our estimates are off. In practice, we find that this procedure is very robust.
Tetramerization Protocol
The addition of streptavidin to the biotinylated monomers is performed at room temperature, with the samples kept in the dark between additions (usually in a drawer). The protocol will probably also work if the samples are kept on ice. The key is to add the streptavidin in small subaliquots, and to mix thoroughly (but gently) after each addition. The streptavidin volumes listed below are appropriate for 100 μL aliquots of biotinylated Arah2.
- Streptavidin-AlexaFluor (MW ~70kD) at 2 mg/ml (~14 mM) to Ara h 2-biotin (MW ~18kD) assuming Ara h 2 concentration of 0.5 mg/ml (~30 mM) in 100 µl:
Add 10 µl of marker to the monomer solution every 10 minutes for a total of 10 times to give a 1:1 volume in the end (2 fold molar excess of biotin binding sites to biotin-protein)
Discussion
References
Contact
- Who has experience with this protocol?
- Email Wayne Shreffler through OpenWetWare