Shivum Desai Journal Week 3
Exploring HIV Evolution: An Opportunity For Research
Purpose
- The first portion of this investigation involves understanding the different ways to view the nucleic acid data associated with the original research paper. The second portion of this investigation involves analyzing a specific sequence of data attained from the original research paper.
Methods
Activity 1/Part 2
- The first step of this investigation started by accessing the original research paper on the PubMed database.
- The next step was to access the amino acid sequences that are stored on the GenBank record website.
- From there data was recorded on S3V1_1 including its accession number, the subject of the study, and from whom the sample was taken.
- From there 5 different sequences were downloaded as a FASTA file and then opened and uploaded to the Biology WorkBench website.
Activity 1/Part 3
- The first step of this part involved uploading all the previously saved sequences in FASTA form to the Biology WorkBench website.
- From there all of the uploaded sequences were selected and then aligned using the ClustalW tool.
- The last step involved comparing the difference in each sequence and analyzing the relation between the sequences, the unrooted tree diagram, and the pairwise similarity scores.
Results
- Activity 1/Part 2
- The accession number of the sequences was recorded as AF089109. The HIV sample for this sequence came from subject 3 in the experiment who was a HIV positive drug user. Then the following sequences were downloaded so that they could be uploaded onto the biology workbench: S3V1_1, S3V1_2, S3V1_3, S3V1_4, S3V3_1
- Activity 1/Part 3
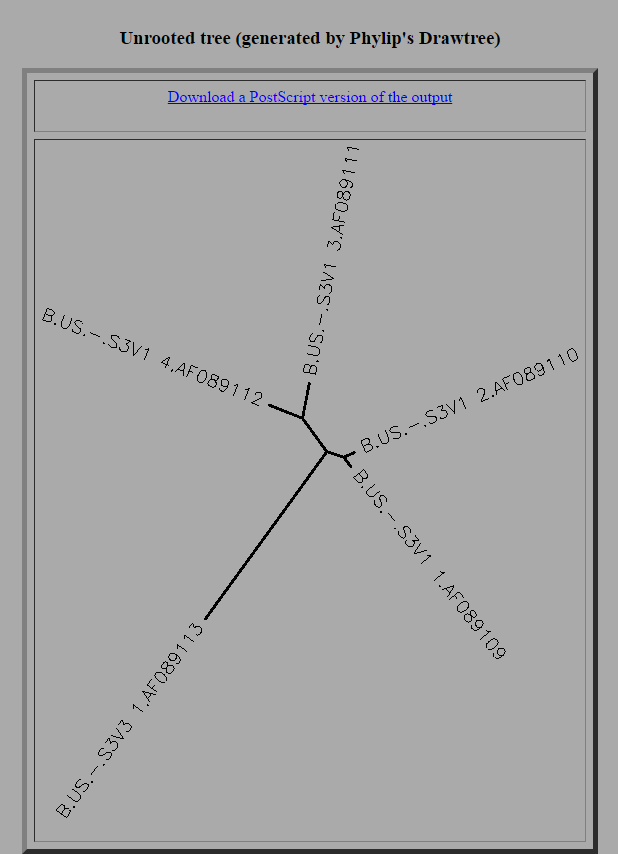
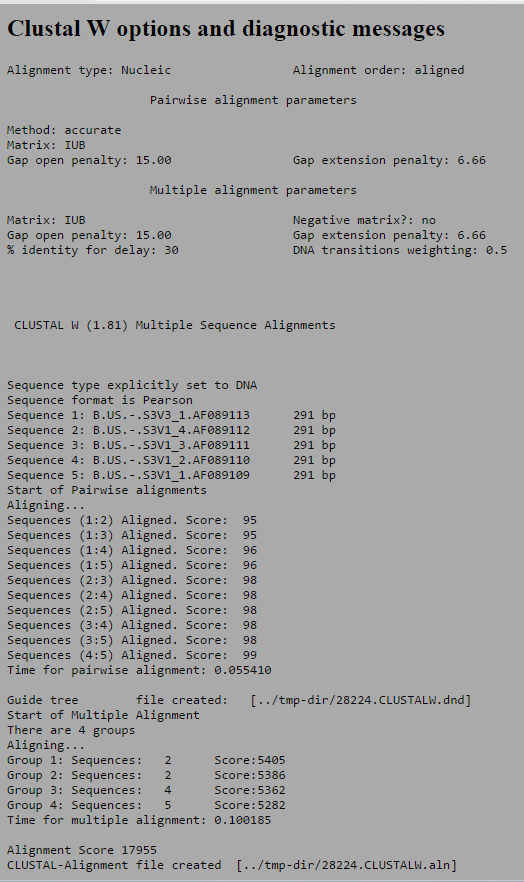
- The sequences were then analyzed by comparing the difference in the sequences along side the topology of the unrooted tree diagram and the pairwise similarity scores. Based upon the difference in the amino acid sequences the tree below was generated to show the similarities between the sequences. Thus, the sequences that have the most distance between them are S3V1_3 and S3V3_1, which means they are the most different sequences and have the most differences in terms of nucleotide bases. Furthermore, all differences and similarities can be see on the unrooted tree chart but one the noticeable characteristic was the similarity between S3V1_2 and S3V1_1, which happens to be the two most closely related sequences in the group. Additionally, the pairwise similarity scores also support the information characterized by the unrooted tree diagram. The difference between S3V1_3 and S3V3_1 is noted by having the lowest similarity score of any of the sequences at 95 while the difference between S3V1_2 and S3V1_1 is resulted in the highest similarity score, 99.
Data Files
References
Markham, R. B., Wang, W., Weisstein, A. E., Wang, Z., Munoz, A., Templeton, A., . . . Yu, X. (1998). Patterns of HIV-1 evolution in individuals with differing rates of CD4 T cell decline. Proceedings of the National Academy :of Sciences, 95(21), 12568-12573. doi:10.1073/pnas.95.21.12568
HIV Sequences Database. (2016, July 21). Retrieved September 13, 2016.
Higgins, D.G., Bleasby, A.J. and Fuchs, R. (1992) CLUSTAL V: improved software for multiple sequence alignment. Computer Applications in the Biosciences (CABIOS), 8(2):189-191.
Subramaniam, S. (1998). The Biology Workbench--a seamless database and analysis environment for the biologist. Retrieved September 13, 2016, from http://workbench.sdsc.edu
Donovan S and Weisstein AE (2003) Exploring HIV Evolution: An Opportunity for Research. In Jungck JR, Fass MR, and Stanley ED, eds. Microbes Count! West Chester, Pennsylvania: Keystone Digital Press.
______________________________________________________________________________________
Preparation For Week 4 Journal Club
Outline of: Patterns of HIV-1 evolution in individuals with differing rates of CD4 T cell decline
- Purpose is to observe different strains of HIV-1 to see what types of forces influence the virus to evolve over time and therefore see how the virus adapts to these influencing forces.
- Past attempts by experiments similar to this have not been successful due to viral variants exceeding the capacity of a selective force such as immune response to control or influence the virus.
- This essentially means that the viruses were most likely in their perfect host environment and were therefore unable to be affected by the immune response.
- Previous techniques did not involve examination of sequence patterns. Unlike this experiment, which will conduct analysis of each sequence take from all 15 subjects.
- 15 subjects were analyzed in this experiment over a period of four years.
- Nonprogressor, moderate, and rapidly progressing subjects were all analyzed.
- These are all different types or categories of HIV patients (subjects).
- Nonprogressor, moderate, and rapidly progressing subjects were all analyzed.
- Shows that high levels of genetic diversity is not associated with more rapid CD4 T cell decline.
- Past attempts by experiments similar to this have not been successful due to viral variants exceeding the capacity of a selective force such as immune response to control or influence the virus.
- Methods
- Study population
- 15 participants were selected from cohort of injection drug users participating in the AIDS linked Intravenouse Experiences study (ALIVE) in Baltimore, MD.
- The indiviuals were checked up every 6 months at which times blood was obtains for virologic and immunologic studies.
- They were followed from point of HIV-1 seroconversion and had different levels of CD4 T cells.
- Rapid progressors, moderate progressors, and nonprogressors.
- Sequencing of HIV-1 env Genes
- PCR was used to obtain env gene from blood.
- External env primers were used and the BamHI or EcoRI restriction sites were included for cloning purposes.
- PCR was conducted and the amplified sequences were cloned into pUC19 and was then sequenced using the Sanger chain termination method with the previously listed primers.
- Only five samples were subjected to second round PCR because they were at the lowest cellular dilution.
- Plasma viral load.
- This was determined by reverse transcription-PCR.
- Generation of phylogenetic trees.
- Trees were generated using the MEGA computer package.
- Taxon labels indicate the time at which each strain was isolated and the number of identical replicates sampled.
- Taxa are colored according to the time point at which they were observed.
- Correlation analysis was conducted to see the connection between genetic diversity or mutual divergence and CD4 T cell count after a year’s time.
- dD/dN ratios were used to compare each sequences with each subsequently observed strain.
- High genetic diversity in strains from subjects 9 and 15
- Leads to thoughts that the individuals maybe infected with two different viruses.
- Both were HIV-1 seronegative up to 7 months before the first is it
- Exclusion of subject 15 due to multiple recombination events did not change the conclusions of the dS/dN analysis.
- Comparison of the Rate of change in Divergence and Diversity.
- Each individual was fit with a regression line of divergence/diversity over time and summarize it with the slope for each individual.
- Averages of the slopes for each of the 3 groups was measured by the decline of CD4 T cell counts.
- Study population
- Results
- Patterns of CD4 decline were variable amongst the 15 subjects.
- T cell number changes ranged from 53 cells per year to 593 cells in a year.
- Nonprogressor group have low viral loads at early points, the moderate progressors were indistinguishable from the rapid progressors.
- Genetic sequence analysis was focused on the viral env region around the third hypervariable domain. This is an important site of host virus interaction and tolerates frequent mutations. 6-21 env sequences were amplified by PCR from each individual visit, resulting in the total of 873 clones that were sequences and analyzed.
- Changes in HIV 1 sequences over time were quantified by 2 parameters
- The genetic diversity at each visit
- Defined as the mean number of nucleotide differences between any two clones from the visit
- Ranged from -2.94 to 5.10 nt per clone per year
- Divergence
- Quantified as the median percentage of nucleotides per clone at a given visit that differed form those present in the consensus env sequence form the first visit of that subject.
- Ranged from .13% to 2.09% of the nucleotides per clone per year.
- The genetic diversity at each visit
- Further examination of subjects 9 and 15 excluded the possibility of a dual infection, development of recombinant virus or mistiming of the point of seroconversion.
- Both diversity and divergence increased over time in all 3 progressor categories
- Rapid progressor group had significantly higher rate of increase in divergence over time than the nonprogressor group.
- Non significant difference in the p between moderate and rapid progressors in terms of their increase in divergence.
- But there was significant differences in slope of the rate of increase in diversity between rapid and nonprogressors and nonprogressors and moderate progressors. But no such trend between moderate and rapid progressors.
- Subjects who’s virus showed greater genetic diversity or divergence at a given visit were likely to have a greater CD4 t cell decline over the next year.
- Phylogenetic trees from 10 of 15 subjects showed no evidence of predominance of a single strain throughout a period of times.
- in these 10 subjects, host factors selected against clones that predominate any given visit but they are not effective against full range of viruses.
- Discussion
- Higher levels of genetic diversity and divergence in the HIV 1 variants were associated with a decline in CD4 T-cells.
- Viral strains from nonprogressors showed possible selection against amino acid change, while those from progresses showed selection for such change.
- Selection against nonsynonymous mutations as seen in the nonprogressors may lead to appearance of more competent viruses that would be recognized by the host immune system.
- But would essential lead to other variants escaping the immune response.
- A similar study was conducted by McDonald et al. in which the diversity of the rapid progresses was a lot less than what was observed in this experiment. The best explanation of this result was that the McDonald study did not follow its subjects from the point of seroconversion and there were many less time points of evaluation through the study itself.
- Another study by Wolinshy et al. also yielded different results in regard to two of their subjects which had less genetic diversity even though they had the most rapidly declining CD4 T cell counts.
- The reason for this maybe that the best fit viral strain replicated in those subject’s which is why they were able to emerge so well in their host environment.
- Basically, the HIV viruses in the subjects bodies were found to always overpower or slip by the immune system because the body’s immune system would always attack the most frequent virus but not the full array of viruses. Which, allowed the other variants to basically slip past the body’s threshold activated autoimmune response.
Bibliography For Outline
Nowak, M., Anderson, R., Boerlijst, M., Bonhoeffer, S., May, R. & McMichael, A. (1996) Science 274, 1008–1011. Wolinsky, S. M., Korber, B. T., Neumann, A. U., Daniels, M., Kunstman, K. J., Whetsell, A. J., Furtado, M. R., Cao, Y., Ho, D. D., Safrit, J. T., et al. (1996) Science 272, 537–542. Markham, R.B., Wang, W.C., Weisstein, A.E., Wang, Z., Munoz, A., Templeton, A., Margolick, J., Vlahov, D., Quinn, T., Farzadegan, H., & Yu, X.F. (1998). Patterns of HIV-1 evolution in individuals with differing :rates of CD4 T cell decline. Proc Natl Acad Sci U S A. 95, 12568-12573. doi: 10.1073/pnas.95.21.12568
Definitions
- Seroconversion:The stage in an immune response when antibodies to the infecting agent are first detected in the bloodstream. For example, people infected with HIV typically seroconvert about 4–6 weeks following the initial infection, when antibodies against viral proteins are first produced.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
- T Cell: Any of a population of lymphocytes that are the principal agents of cell-mediated immunity.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
- HIV 1: The retrovirus that causes AIDS in humans. It has a specific affinity for the helper T cells of its host, binding to CD4 antigens on the cell surface and thereby disabling these cells.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
- HIV 2: Two varieties (serovars) are known: HIV-1 and HIV-2. The latter, which is less virulent, is found chiefly in Africa. The clinical course of HIV-2 infection is generally characterized by a longer asymptomatic stage, lower plasma HIV-2 viral loads, and lower mortality rate.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
- CD4: A surface glycoprotein that characterizes helper T cells. CD4 recognizes foreign peptides bound to MHC class II proteins expressed on the surface of B cells, macrophages, and other antigen-presenting cells. It acts as a coreceptor, binding simultaneously with the T-cell receptor to the antigen-MHC II complex and enhancing the T-cell's response. CD4 consists of a single polypeptide chain organized into four immunoglobulin-like domains.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
- Reverse Transcription PCR: Reverse transcription PCR (RT-PCR) is used for amplifying molecules of RNA, by initially converting the RNA into its complementary DNA molecule using the enzyme reverse transcriptase and then following the standard procedure for PCR.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
- Synonymous Mutation: a nucleotide substitution that does not result in an amino acid substitution in the translation product owing to the redundancy of the genetic code.
- Smith, A. D., & Bender, D. A. (2006). Oxford dictionary of biochemistry and molecular biology (2nd ed.). Oxford: Oxford University Press.
- Non synonymous Mutation: a nucleotide substitution in a protein‐coding gene that results in an amino acid substitution in the translation product.
- Smith, A. D., & Bender, D. A. (2006). Oxford dictionary of biochemistry and molecular biology (2nd ed.). Oxford: Oxford University Press.
- Virology: The scientific study of viruses and the diseases caused by them.
- Smith, A. D., & Bender, D. A. (2006). Oxford dictionary of biochemistry and molecular biology (2nd ed.). Oxford: Oxford University Press.
- Monophyletic: In systematics, describing a group of organisms that contains all the descendants of a particular single common ancestor. In cladistics such a grouping is called a clade and is the only type of group regarded as valid when constructing classification schemes.
- Hine, R., & Martin, E. (2015). Oxford Dictionary of Biology (7th ed.). Oxford: Oxford University Press.
Useful Links
Acknowledgments
I would like to thank Dr. Dahlquist for her help on this assignment. As well as the help of Jordan Detamore who was my partner in this experiment. I would also like to recognize the Oxford Dictionary Biology Dictionary for its help with biology definitions. Lastly, while I worked with the people noted above, this individual journal entry was completed by me and not copied from another source.