Schumer lab: Embryo dissection and classification
Embryo Staging Systems
We reference two standardized classification systems for the developmental stages of poeciliids:
- Haynes 1995: a system with 11 stages meant to generalize across the diversity of poeciliid development (good for ethanol-preserved embryos where morphological details aren't clear): https://www.jstor.org/stable/pdf/1446809.pdf?refreqid=excelsior%3A5fa51919f5a0bf5dcb394e933ebe93dd
- Tavolga 1949: a detailed system with 26 stages designed specifically with reference to Xiphophorus maculatus and hellerii (good for live embryos or ones where high-quality pictures were taken before preservation): https://www.proquest.com/docview/301824279?pq-origsite=gscholar&fromopenview=true
Embryo Protocols
(Written by Dan VIII-20, added to by Ben)
Gross Dissection of Embryos
General Notes
The best way to avoid damaging embryos when dissecting is to make sure to avoid inserting the point of the scissors too deeply into the body cavity while making the incision. It can be helpful to think of angling the scissors up such that they are actually pointing away from the body cavity; this will create tension that lifts the body wall away from the eggs and gives you more space to cut.
That said, you will definitely pierce a yolk every once in a while, and that's alright. Often our main goal is to avoid damaging the embryos themselves,
Gross dissection of ovary (dissecting stereoscope optional but helpful)
- With the right side of the female facing upward locate the urogenital opening and anus just anterior to the insertion of the anal fin.
- With fine scissors, pierce the ventral body wall between the two openings, taking care to keep the scissor blades as close to the body wall as possible in order to avoid damaging the ovary and embryos.
- Cut upward perpendicular to the main body axis for three quarters of the body depth (roughly to the top of the lateral line).
- Then cut parallel to the main body axis toward the head and through the ribs, stopping before the pectoral girdle, just posterior to and above the insertion of the pectoral fin.
- Then cut downward perpendicular to the main body axis, just posterior to the pectoral girdle (i.e. posterior to insertion of pectoral fin) ending at the most ventral point of the body.
- These incisions create a rectangular flap which can then be carefully folded downward to open the body cavity.
- In pregnant females most of the body cavity will be occupied by the ovary and developing embryos (like a bunch of yellow grapes usually). You may have to carefully peel away the silvery-black peritoneum with a pair of fine forceps.
- Once the ovary is uncovered snip the oviduct that connects the posterior end of the ovary to the urogenital opening using fine dissecting scissors.
- Then remove the whole ovary by grasping the oviduct with fine forceps and gently pulling outward. It will usually come out in one piece. but you may have to continue to pull away the peritoneum with forceps.
Embryo Staging and Photography
Preserved Embryo removal and staging
(High-throughput, quick and dirty version for pre-preserved embryos)
- Under the dissecting stereoscope individually separate the embryos from the ovary starting with those closest to the oviduct.
- Examine each one closely and record its developmental stage following Haynes 1995 (see link at the top of this document) in comparison to the Gambusia affinis illustrations therein.
- In a large petri dish with enough 95% EtOH to maintain moisture but not allow for easy movement, arrange the embryos in columns of five in order from first extracted to last.
- Photograph both sides of the dam along with the embryos, and then the embryos by themselves, making sure to include appropriate labeling and a ruler for scale. This should result in three photos per brood. Note: any extra ovarian tissue, including undeveloped eggs that are often present next to late stage broods, should be placed in the photos as well. See example photo.
- Store embryos individually in 95% EOH in labeled PCR strip tubes or similar containers. Be sure labeling indicates collection (i.e. CALL III-20), dam id (i.e. F 01), and embryo order. Ovarian tissue can be stored in a final tube following the last embryo.
AmScope Microscope camera operation
Both the dissecting microscope in the fish room and the Leica MZFLIII scope in the gel room are compatible with the lab's AmScope Mu1803 18mp microscope camera. The camera inserts into the right eyepiece of the fish room scope, and the upward-facing port coming out of the Leica scope's central column. The camera comes with two adapters that can be fitted around the camera's lens to make it fit each camera's port.
To use the camera, you have to download and install AmScope's proprietary software: https://www.amscope.com/software-download#toup1
- Connect the camera to your computer with the included cord.
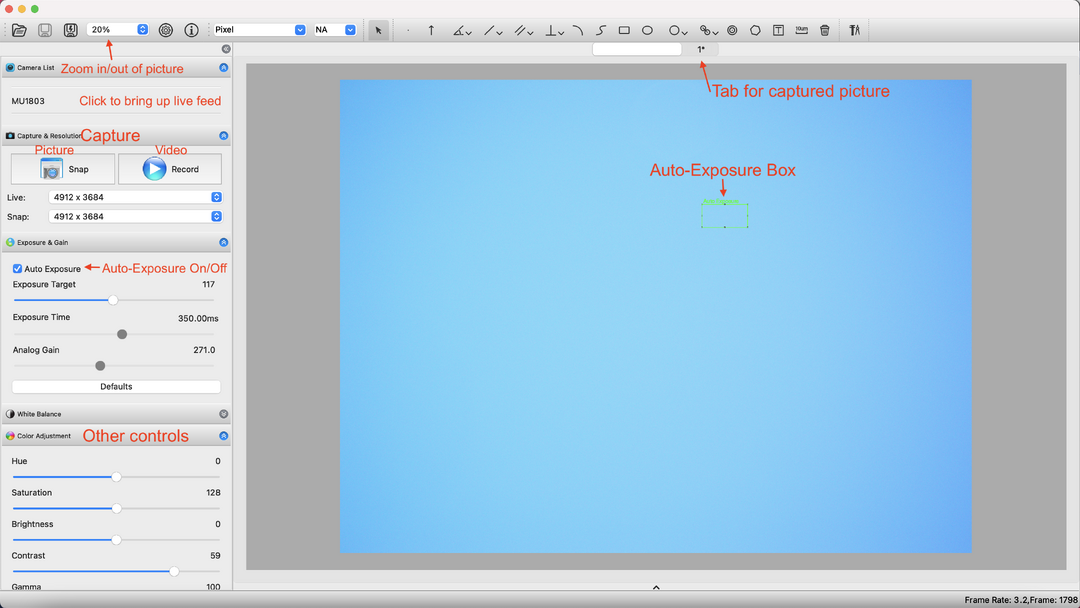
- Click the name of the camera (MU1803) under "Camera List" to start live feed from the camera (see example picture).
- Select "Auto Exposure" to create a green box onscreen from which the software will judge the exposure level.
- Move the box to a part of the image that leads to the desired exposure level, then deselect "Auto Exposure" to lock the camera at this exposure level
(This prevents obnoxious changes in exposure when manipulating things on-camera). - Adjust the contrast, hue, etc. as desired.
- Take pictures by clicking the "Snap" button, or videos by clicking "Record".
- Pictures will appear as tabs above the image, alongside the tab for the live feed.
- Depending on the size of your monitor, you will probably have to "zoom out" to view the entirety of a capture image at once; 20% works well on my monitor.
- Close pictures by double-clicking their tab; you will be prompted to save them if you have not already done so.
Embryo removal and staging with photography
(Slower version, recommended for live embryos)
- Take a picture of the mother pre-dissection, then follow gross dissection instructions above.
- Place the ovary in a petri dish with tank water, and individually separate the embryos from the ovary under the dissecting stereoscope.
- Attach the microscope camera to the dissecting scope, as described above.
- For each embryo:
- Transfer to a clean petri dish with a micrometer (included in the MU1803 microscope camera box) underneath and centered in the dissecting scope's field of view.
- Capture one or more pictures of the embryo with the chorion intact.
- Next, remove the chorion to allow the embryo's body to straighten:
- Using two sets of fine-tipped forceps, grab two adjacent points on the chorion. The chorion is easiest to see where the embryo's body pushes it up away from the surface of the embryo, but this tension also makes it hard to grab at this point.
- Gently pull apart with the forceps until the membrane ruptures - in early-stage embryos, there is a significant pressure inside the membrane that will cause the yolk to squeeze out when the chorion breaks. Continue teasing off the chorion and the yolk should return to a more normal shape (if you've managed to break the chorion without rupturing the yolk underneath, which is not always the case)
- Use forceps to orient the embryo while taking at least one picture from the side, and one or more from the top and/or bottom - these can be used for detailed staging and morphometrics later (see example photos).
- Save photos with names that minimally include the dam id (i.e. F 01), embryo id, and photo number, and be sure to include information on the collection in either the file name or in the directory structure.
- Store embryos individually in 95% EOH in labeled PCR strip tubes or similar containers. Be sure labeling indicates collection (i.e. CALL III-20), dam id (i.e. F 01), and embryo order. Ovarian tissue can be stored in a final tube following the last embryo.