SBB11Ntbk-James Macaulay
James Macaulay 13:23, 15 February 2011 (EST)
Construction of composite part red/recA basic part sbb1142 Digest sbb1142 from pBca9145-jtk2979 (EcoRI/BamHI, 3287+2057, L) Sub into pBca1766 (3287bp, EcoRI/BamHI) Product is pBca1766-sbb1142 [red/recA]
Bglbrick Construction of P_hdeAB basic part sbb1128 PCR jm50f/jm51r on MG1655 genomic DNA (431bp, EcoRI/BamHI) Sub into pBjh1601KC (Bglbrick vector: 3140bp) Product is pBjh1601KC-sbb1150 [P_hdeAB] ------ jm50f Forward cloning of P_hdeAB Bglbrick basic part CGATAGAATTCATGAGATCTTAAGAAGAAAATCCCCTGC jm51r Reverse cloning of P_hdeAB Bglbrick basic part CCATTGGATCCCCAACTGCAGTTGGCTGG
- Set up PCR for part sbb1142 with 2K55 protocol.
James Macaulay 13:44, 17 February 2011 (EST)
- Set up an analytical gel for part sbb1142, which I renamed as jm54 to differentiate from the mass of other parts with the sbb prefix.
- No visible bands showed up, need to set up new PCR.
- New PCR setup:
- jm54-W
- PCR_Protocol -- 2k45 PCR cycle
- jm54-D
- PCR_Protocol -3.3µL ddH20 + 3.3µL DMSO. -- 2k45 PCR cycle
- New PCR setup:
- No visible bands showed up, need to set up new PCR.
- Set up a digest of pBca9145-jtk2979 with EcoRI/BamHI.
- Used 5µL of PCR product to be digested instead of 8µL.
James Macaulay 16:25, 18 February 2011 (EST)
- Submitted an analytical gel for jm54-W and jm54-D, re-PCR'd from previous day.
- Looking for 431 bp bands in lanes 1 and 2:
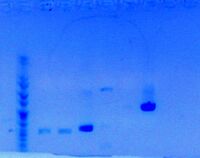 Successful? Y
Successful? Y- - Since successful, moving on to perform regular zymo cleanup.
- Performed a Zymo Gel Purification for part jtk2979, labeled JAM jtk zymo and placed in Box C.
James Macaulay 13:10, 22 February 2011 (EST)
To do today:
- Digest PCR Products
- Excise out band in lane1 (431 bp)
then Gel Purify, then ligate. The ligation step will probably be saved until Thursday (2/24).
- The cut-and-paste part, jtk2979 needs to be redone because the AW buffer was used instead of A4 buffer. We need to re-digest and gel-prep.
James Macaulay 13:50, 24 February 2011 (EST)
Zymo Gel purification of part sbb1128.
- Used 8µL for DNA preparatory gel.
- Placed in box A
- labeled "JM28 pur 24".
- JM=initials, 28 refers to the part number suffix, 24 refers to the day last manipulated, eg., 2/24.
EcoRI/BamHI digest of pBca9145-jtk2979
- Only had 4µL of DNA, so replaced the other 4µL with ddH2O.
- Gel purification My sample is placed in lane 2
- Looking for 3287bp segment of the digestion.
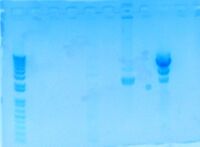
- Notes for next time
- Used 4µL of DNA for digestion, so ELUTE using 4µL ddH2O when performing final step of zymo purification.
- labeled: JM2979, and placed in box: A
James Macaulay 12:46, 1 March 2011 (EST)
All cell parts go into "Lefty comp cells" 1mL, insert, buffer, water, last: .5mL ligase. Vectors: 1mL of vector material
Let ligation sit for 30 min on bench.
Then do transformation, then assemble into groups of 3. Don't wait until ligation is done to start setting up: KCM + water to competent cells, then however much of cells (protocol says). -do this in half mil tubes. (fits in thing)
after 30 min, put ligation tube in ice (1 min), squirt in competent cells (with kcm + water), then do heat-shock for (1-1.5 min), then rest on LB.
At this point take things up stairs, put piece of tape around it(tape it to some shaker)
After rescue (45min-1hr), all the promoters are KC, so they can be done on Kan or Chloramphenicol. -make sure to write on bottom of plate; yourname, righty or lefty? then put name on board.
Zymo Gel Purification of jtk2979
labeled: jm2979, placed in box A
James Macaulay 14:15, 3 March 2011 (EST)
- Miniprep of part sbb1128
- Elutions Labeled: jm42 (1) through (4), placed in box A
- Cut and Paste Part
- labeled: jm2979, placed in box A
- needs ligation
James Macaulay 12:02, 8 March 2011 (EST)
- Mapping of sbb1128
- Minipreps placed in box C
- samples are last 4 lanes in one of the gels.
- Ligation and Transformation of jtk2979 (red/recA)
- Plated on Spec resistance with Lefty cells
- Labeled with my name and part name, and date of transformation
- ligation product placed in box C
James Macaulay 12:53, 10 March 2011 (EST)
- Cut and Paste Part jtk2979
- plated and considered good
