SBB11Ntbk-Helen Shi
~~!~~
Helen Shi 14:38, 17 March 2011 (EST)
Today, my group (group #3) and I analyzed all the sequences for plasmids. My plasmid pBjh1601KC-sbb1136 has a perfect sequence that matches my template. My other plasmid pBca1766-jtk2936 has many insertions and deletions and was difficult to read overall.
Helen Shi 14:38, 15 March 2011 (EST)
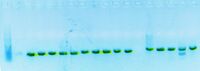
Today, I submitted my plasmids for sequencing. For plasmid pBca1766-jtk2936 samples 1-4, I digested the plasmid with EcoRI and BamHI again before re-running the gel.
pBca1766-jtk2936 samples #1-4 (~1000 bp)

After looking at this gel, I submitted samples 3 and 4 of plasmid pBca1766-jtk2936 for sequencing, and I also submitted samples 3 and 4 of plasmid pBjh1601KC-sbb1136 for sequencing.
Helen Shi 14:55, 10 March 2011 (EST)
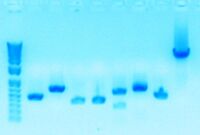
Today, I ran a gel to verify that plasmid pBjh1601KC-sbb1136 samples 1-4 are the correct plasmid.
030811-Gel1

Lanes 5-8 contain plasmid pBjh1601KC-sbb1136 samples 1-4 (~500 bp).
I also did mini-prep for the 4 colonies picked from the plate for plasmid pBca1766-jtk2936, and then digested the miniprep plasmid before running a gel. However, the gel samples were lost so I was unable to run the gel today.
Helen Shi 15:16, 3 March 2011 (EST)
Today, I got my plate of transformed pBjh1601KC-sbb1136 E. coli. The kanamycin plate had a modest growth of colonies (~30-40 colonies) of which 2 were pink in color. Four colonies had been picked from my plate and grown in 5 mL of LB overnight. None of the picked colonies were red/pink so I proceeded with miniprep with these 4 colonies. A Qiagen miniprep kit and protocols was used to elute 50 uL each of concentrated plasmids/DNA for the 4 colonies picked.
I also performed a ligation with T4 ligase and the pBca1766 vector and transformation for my jtk2936 part to produce the pBca1766-jtk2936 plasmid. I transformed the pBca1766-jtk2936 plasmid into the lefty competent cells, and then plated the entire tube of transformed cells (~120 uL) on a spectinomycin plate.
Helen Shi 13:58, 1 March 2011 (EST)
Today, I ligated my ss1136 part with its pBjh1601KC vector backbone. After adding the ligation mix in a half mL tube containing T4 ligase, I incubated it for 30 minutes before proceeding to transformation of my ligated plasmid into lefty competent cells. For transformation, I added 70 uL of thawed lefty competent cells and KCM to the pBjh1601KC-ss1136 plasmid. I then added 100 uL of rescue LB before incubating the cells for an hour. After an hour, I transferred all of the LB with cells and KCM (~150 uL) onto a kanamycin plate and spread the cells around the plate. The plate will be incubated at 37 degrees Celsius over night.
On Thursday, I will ligate the jtk2936 part with the pBca1766 vector and then transform the plasmid into lefty competent cells.
Helen Shi 13:51, 24 February 2011 (EST)
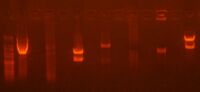
Today, I digested my jtk2936 part from the pBjk2741-jtk2936 plasmid with EcoRI and BamHI, and then I ran a preparation gel after an hour of digestion:
022411-PrepGel1, Lane 3 (1095 bp)

The jtk2936 part is 1095 bp; the other two bands are around 2000 bp and 3000 bp so the 3000 bp part may be the undigested DNA. Then I cut out my band and melted the gel in 600 uL of ADB buffer. I then proceed to complette the gel purification process.
I also did a Zymo gel purification of my previously digested sbb1136 part. I eluted the sbb1136 digested part with 8 uL of water. I should be doing the ligation and transformation on Tuesday.
Helen Shi 14:19, 22 February 2011 (EST)
Yesterday, I found out that I used the AW Zymo wash buffer for all of my Zymo cleanups and gel purifications when I should have used the A4 wash buffer. Thus, my PCR for part sbb1136 and digestion for part jtk2936 must be redone. Chris redid the PCR for part sbb1136 on Monday and analyzed the gel:
022111-AnalGel2, Lane 12 (531 bp)
Today, I did a Zymo cleanup for part sbb1136 using the AW wash buffer, and then I digested the purified DNA with EcoRI and BamHI restriction enzymes and put them in the thermocycler at 30 degrees C for an hour. Afterward, I ran a preparation gel for my digested PCR product:
022211-PrepGel2, Lane 1 (~500 bp)
I then cut the ~500 bp band in lane 1 from the gel and added 600 uL of ADB buffer before melting the gel at 55 degrees C for about 5 minutes. The gel was then stored in the fridge to be ligated and transformed on Thursday.
Helen Shi 15:49, 18 February 2011 (EST)
Today, I performed a gel cleanup for my digested part, jtk2936, for which I ran a preparation gel yesterday. I now have 8.5 uL of jtk2936 DNA. Next class, I will digest this part, ligate it with my plasmid vector, and transform it into E. coli.
For my other part, sbb1136, I should also digest it next class before ligation and transformation.
Helen Shi 13:38, 17 February 2011 (EST)
Today, I got my PCR back and then I ran an analytical gel to check that the size of my PCR product is 531 basepairs. From the gel picture, my PCR product appears to be about 500 basepairs, which is correct. Thus, I continued on to the purification of my PCR product with a Zymo Cleanup; I eluted 33 uL of my sbb1136 part. Tomorrow, I will digest my PCR product with EcoRI and BamHI.
021711-AnalGel1, Last column (~500 bp)
I also digested the plasmid pBjk2741-jtk2936 with EcoRI and BamHI restriction enzymes to cut out the HlyE part. After an hour of digestion, I am performing a preparation gel to extract the digested HlyE part (1095 bp). Tomorrow, I may ligate and transform my HlyE part.
021711-PrepGel2, Column 3 (~1000 bp)
Helen Shi 13:44, 15 February 2011 (EST)
Today, I set up the PCR for my sbb1136 part using the prediluted oligos ss63r and ss63f (10 uM). Since the template for my part came from the MG1655 genome, I used the Expand buffer and polymerase for my PCR. I set up my PCR reaction with 1 uL each of my 10 uM oligos. From my construction file, my predicted PCR product has a size of 531 bp so the PCR file to be used is 2K55.
Helen Shi 04:29, 8 February 2011 (EST)
Hi!