Physics307L:People/Franco/Rough Draft for Final Report
SJK 19:14, 6 December 2008 (EST)

Hey Manuel, Overall I think this was a very good first draft. You will see a lot of comments from me below, and I hope those help you to fill in the gaps. Some of your sections were very good (e.g. the materials and methods) while others are going to require much more work. Some of these criticisms (such as references) we've already discussed in person. Please ask about clarifications!
Balmer Series Analysis and the Rydberg Constant
SJK 14:56, 6 December 2008 (EST)

This is good Title and Author information. The title could be improved slightly by being more specific, such as calculation of the Rydberg constant from the hydrogen balmer series...but it's pretty good as you have it. Room number is not necessary. On a published paper, contact information is so someone in principle can contact you about the paper.
Author: Manuel Franco Jr.
Experimentalist: David Eduardo Sosa and Manuel Franco Jr.
Location: Albuquerque, New Mexico, 87131, United States
University of New Mexico Department of Physics and Astronomy
800 Yale Blvd. NE
Room Number: 116
E-mail: eMJeF_2004@hotmail.com
Abstract
In this experiment, we determined the Rydberg Constant, compared hydrogen and deuterium, and analyzed the Balmer series. We compare our experimental value to the accepted value R = 1.0967758 × 107 m−1.SJK 15:07, 6 December 2008 (EST)

this second sentence is a result and would typically be close to the end of the abstract. Also, it would be better to report your value for the Rydberg constant and say whether it's consistent with the accepted value.
We did this by using four wavelengths of the spectral line emissions of hydrogen in the Balmer series. These colors "reflect emissions of photons by electrons in excited states transitioning to the quantum level described by the principal quantum number n equals 2." (1)SJK 15:00, 6 December 2008 (EST)

It is very good that you use quotes and cite your source here. However, direct quotations (using exact quote) are extremely rare in the abstract and also rare in the paper. So, you need to paraphrase in your own words.
Hydrogen and deuterium are compared to each other, and their experimental values are also compared to their accepted values. In running through the data concepts from the Balmer serious arise that contribute to the experiment. These concepts consist that "the Balmer series is characterized by the electron transitioning from n ≥ 3 to n = 2, where n refers to the radial quantum number or principal quantum number of the electron." (1)
Introduction
SJK 15:32, 6 December 2008 (EST)

Your introduction as is is a brief history of the Rydberg constant...this is interesting, but it's only a small part of what your introduction should be. You should introduce the physics and importance of the constant (as you have done), then you should include: (1) discussion of ways of measuring the constant and the best methods for currently measuring the constant, along with the current accepted value (you will need citations to refereed publications), (2) a brief introduction to what you tried to do, and (3) a concluding statement, something like "our measurements were slightly inconsistent with the accepted value and we can improve these in future measurements by ..." Just to reiterate, you need to cite several original publications in the introduciton, not just wikipedia articles (which are not considered refereed, and which are considered "secondary" sources, not primary.).
In 1885, Johann Jakob Balmer, a mathematician, discovered this series of colors of hydrogen in the color spectrum, hence the Balmer series. He derived such equation based on his observations: http://upload.wikimedia.org/math/1/2/2/1223c57e48b6b6c0cf038dd69a22db64.png [1]
Three years after the discover of this series, Johannes Robert Rydberg, a physicist, would revise the equation and analyze the details of this phenomena. He would revise the equation to what we commonly known today: http://upload.wikimedia.org/math/0/a/c/0acd60cdab573b571c1a07ee23a8c0d7.png
He used this formula "to predict the wavelengths of photons... emitted by changes in the energy level of an electron in an atom." [2] He would later discover the constant slope in the wavelength over the quantum number transitions.
SJK 15:24, 6 December 2008 (EST)

Is this really the number Rydberg found? It looks more like a modern accepted value. His number probably had much much more uncertainty. You will need to find a refereed publication (e.g. from that NIST CODATA site we were looking at) as a source of the accepted value.
He found the slope to be: http://upload.wikimedia.org/math/7/b/6/7b625152ea50ad187556f8d158b79aff.png
The constants are as follows:
[math]\displaystyle{ h \ }[/math] is the Planck's constant,
[math]\displaystyle{ m_e \ }[/math] is the rest mass of the electron,
[math]\displaystyle{ e \ }[/math] is the elementary charge,
[math]\displaystyle{ c \ }[/math] is the speed of light in vacuum, and
[math]\displaystyle{ \epsilon_0 \ }[/math] is the permittivity of free space. [3]
Apparently, both Johann and Johannes were Swiss, and both contributed to this discovery.
Method and Materials
SJK 15:57, 6 December 2008 (EST)

You methods section was really good (mainly style issues). Almost everything was there, good job. One missing item is a discussion of analysis methods. This likely won't be long (and maybe you've done it later, I haven't looked yet. Just a bit about what software you use for analysis "Excel (Microsoft, Redmond WA)" and any specific algorithms (such as LINEST).
Equipment
SJK 15:44, 6 December 2008 (EST)

All of the information is in this "equipment" section, very nice job. My comments here are more "style" issues. (1) You don't need the phrase "in this experiment" (2) the customary way to cite companies is to say "...a constant deviation spectrometer (Company name, location (city)" in this case, though, you may not be able to find the company location if it exists anymore (it's maybe Adam Hilger Ltd.). You can link to the website via the references (so put a number and then in the references section give the nice website you found). (3) You don't usually publish that "you were safe" but if there are important safety notes, it is really good to include it. In this case, high voltage safety is common, and you don't have any special advice, so your safety comments would not help the reader. (4) the phrase "turn on the apparatus..." is a "how to" whereas in a scientific report it should be "we did" statments (as in your previous sentence).
In this experiment we used a constant deviation spectrometer with a Pellin-Broca constant-deviation prism [4], a spectrum tube power supply [5], and 3 spectral line tubes of three different elements: Hydrogen, Deuterium, Mercury. Details of the materials used are in links. We began by making sure that all instruments were functioning correctly, and (most importantly)made sure that we were in a safe environment. We next plugged in the power supply, and inserted the Hg tube as instructed by the manual. Turn on the apparatus and allow it to heat for about a minute or so.
Calibration
After setting up the equipment, we first start off by calibrating the spectrometer by using mercury (Hg) and the table of spectral lines of Hg (Lab Manual: Section 4.3). SJK 15:46, 6 December 2008 (EST)

Dr. Gold's lab manual can be one of your numbered references, and then you can say "as in section 4.3 of ref (#)." Also, again here use the past tense for methods "we did this"
Hg Spectral Lines
| Color | Wavelengths |
|---|---|
| Deep Violet | 404.7 nm (hard to see) |
| Violet | 435.8 nm |
| Blue-Green | Skipped (very weak) |
| Green | 546.1 nm |
| Yellow | 577.04 nm |
| Yellow | 579.0 nm |
| Red | 690.75 nm |
We first set the dial one of the wavelengths in the above table. We used the violet color because our data would deviate based on our initial calibration. Since violet is centric it would minimize data instrumental error. We used the other colors to confirm that it's properly calibrated. Then we aligned the colors using the cross hairs in the eyepiece. We adjusted the prism to align the cross hairs with the color as need. Once we calibrated the spectrometer, we adjusted the slit at the other end of the spectrometer to .7 mm. We adjusted the slit as we saw fit later on, but we stayed within the bounds of .5 and 1 mm. The narrower the slit, the color had a higher resolution, but the wider the slit, the color was somewhat blurred. As we were calibrating, Koch mention the us that the gears in the dial have what is called backlash. Backlash is were if you turn the dial clockwise, take a measurement, then counterclockwise there will be gap that will deviate your measurements. So when the spectrometer is calibrated, turn it going counterclockwise or clockwise and take measurements based on that initial direction. If you manage to pass your measurement, go back passing the measurement, and then turn it again stopping at the measurement. Stick to the original direction. All the data measurements have to be taken the same way initially. If we were to take a data point clockwise, or opposite to direction we initially used to calibrate, our data would be off by about 1 nm.SJK 15:49, 6 December 2008 (EST)

This turned into a major "how to" section as opposed to "this is what we did" as it should be. You have too much about backlash...you can condense it by saying, "all measurements were taken by approaching the spectral line while turning the dial counterclockwise to eliminate problems from backlash." Backlash is a very common thing nowadays, so you don't need a citation.
Hydrogen Spectrum Tube
Once we calibrated and backlash was reduced SJK 15:50, 6 December 2008 (EST)

backlash is never permanently eliminated it...rather, you avoid it throughout by only turning in one direction
, we turn the power supply off. Then we change the Hg tube with the H tube. (CAUTION: The test tubes will get hot after extended periods of use. Minor burn is possible when handling them!) We turned the power supply back on. We will now record the wavelengths seen through the eye piece based on the Hydrogen. There are four bright and distinct lines: red, turquoise, bright violet, and a diminished violet. We saw something similar to this: SJK 15:54, 6 December 2008 (EST)

Again, you have to much "how to" in this section. In this case, the safety note is possibly helpful to someone. You would not use bold text or "caution". Instead, you would say something like "An important note is that the bulbs get very hot and caution is necessary when changing bulbs to avoid burns."
Also I like your inclusion of the photo. In a "real" publication, you would probably have copyright problems using someone else's image (you'd need permission). The image you found is likely licensed for free use...but you should cite the wikipedia user who created it or the source, which you can find by clicking on the image on the wikipedia page.
Also, all figures need captions (they need to be numbered and have a descriptive caption so the reader knows what the figure is showing, even without reading the rest of the paper.)
Deuterium Spectrum Tube
After taking several measurements of the wavelength from hydrogen, we repeat the process of replacing the test tubes. This time we took measurements from deuterium. The same colors appear as in hydrogen, and the wavelengths are about the same.
Data
SJK 18:55, 6 December 2008 (EST)

Typically you do not have a "data" section. In your case, you can get rid of this section, and these tables can be number (e.g. "Table 1") and given descriptive captions, (e.g., "Table 1 4 measurements of spectral line wavelengths for hydrogen...) Then you refer to these tables in the Results, "...as shown in Table 1..."
The following measurements are the wavelengths for hydrogen and deuterium. The data was taken counterclockwise.
Hydrogen Measurements
| Color | Measurement 1 (nm) | Measurement 2 (nm) | Measurement 3 (nm) | Measurement 4 (nm) |
|---|---|---|---|---|
| Red | 657.8 | 659.9 | 659.5 | 659 |
| Blue/Green | 486 | 486 | 486.1 | 485.9 |
| Violet | 433.8 | 434 | 434.2 | 434.4 |
| Dim. Violet | 409.7 | 409.8 | 409.3 | 410.1 |
Deuterium Measurements
| Color | Measurement 1 (nm) | Measurement 2 (nm) | Measurement 3 (nm) | Measurement 4 (nm) |
|---|---|---|---|---|
| Red | 659 | 659.9 | 659.5 | 658.1 |
| Blue/Green | 485.9 | 485.9 | 486 | 486 |
| Violet | 434.2 | 434.2 | 434.2 | 434.2 |
| Dim. Violet | 409.8 | 409.7 | 409.8 | 409.3 |
Results and Discussion
SJK 18:59, 6 December 2008 (EST)

A lot of your results section would go in the "analysis" part of the methods that I talked about above. Also, you will need to number the table and put a descriptive caption under it.
SJK 19:03, 6 December 2008 (EST)

After moving the analysis parts to methods, you're going to find that this section is pretty short. Your results section should include clear explanations of all the tables and graphs (yes, there will be duplication between information in the figure captions and the text). Also, you include your final value(s) of the constants. And you should discuss whether your final values are consistent with accepted values. Discuss features of your data (e.g. do some data points look more off than others? What are the major sources of random and systematic error? What could be done to improve it?)
Wavelengths of H and D
This is the Spread Sheet 1 in where I did all my data analysis for hydrogen and deuterium. The following table shows the colors of each wavelength with the averages and the uncertainties. I took the data and found an average for all corresponding wavelengths. Also found their S.E.M. (standard error of the mean), by taking the standard deviation of the mean and dividing the square root of the total number of data points.
| Color | Wavelengths for H | Wavelengths for D |
|---|---|---|
| Red | 656.1 +/- .5 nm | 659.13 +/- .03 nm |
| Blue/Green | 486.00 +/- .04 nm | 485.95 +/- 0.03 nm |
| Violet | 434.1 +/- .1 nm | 434.2 +/- 0.1 nm |
| Dim. Violet | 409.7 +/- .1 nm |
409.65 +/- .12 nm |
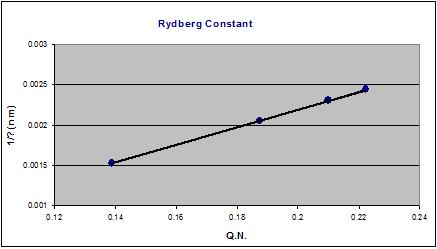
Determining the Rydberg constant
As for the Rydberg constant, I found the constant in this word Spread Sheet 2, and I obtained this graph for hydrogen. The calculated slope is 1.0975 (+/- .0003) × 10^7 m^-1.
I used this equation to find the slope http://upload.wikimedia.org/math/5/a/0/5a05645dc256a0aca0ab1c9cf97b6525.png [7].
I went through and calculated the individual slopes of each hydrogen wavelength, and compared it to the accepted value.
Conclusions
SJK Steve Koch 19:05, 6 December 2008 (EST)

Percent difference is not very meaningful without a comparison to the size of your uncertainty. Also, you can conclude by saying what the next experimental steps may be. In a "real" paper, you'd want to conclude by saying your most important conclusions of the work and by pointing out the impact this will have in your field of science and beyond.
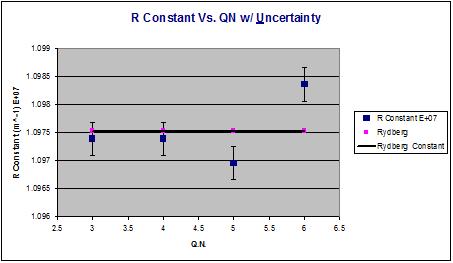
The experimental value for the Rydberg constant is 1.0975 (+/- .0003) × 10^7 m^-1, and the accepted value is 1.0967758 × 10^7 m−1. The percent difference between the values is .06%.
Koch, I did not calculate the value in for deuterium in the first lab of the Balmer series. So, when I take the new and improved data, I will calculate it for deuterium here in this spot.
- Steve Koch 19:01, 6 December 2008 (EST): Sounds good. Investigating Deuterium will be good for one of your follow-up experiments.
References
SJK 19:09, 6 December 2008 (EST)

As I mentioned above you're going to need references to "real" scientific publications--that is, primary research papers that are peer reviewed. Yes, some of these are going to be very old and hard to find! I sent you an email with a link to the NIST CODATA site, which should help you a lot.
(1) Wikipedia article on Balmer Series
Wikipedia article on Rydberg Constant
Acknowledgments
SJK 19:08, 6 December 2008 (EST)

In terms of acknowledging people who came before you, you do a good job. However, the custom is to only thank people who directly contributed to the work. So you don't need to thank Balmer and Rydberg here, because you would have cited their work in the text. Aram would be appropriate (and your lab partner if he's not an author). Also, any other students that helped you (or maybe students from last year's class).
I just want to thank Johann Jakob Balmer and Johannes Rydberg for make this discovery, for without this discovery I would not being doing this. I also wanna thank my my mom, my dad, my brothers, my dog. I want acknowledge Dr. Kock (you can't call me that until you've passed :) ) and Aram for taking the time to instruct us and show us how to go about this and other experiments. I want to thank the department of physics and astronomy, a.k.a. PandA, for providing the materials for this experiment.
Links
Question for Koch
- Why does Rivera take several measurements for mercury in his spreadsheet?
- Steve Koch 19:17, 6 December 2008 (EST): I didn't look at his spreadsheet, so I'm not sure. Perhaps to get better precision? You can always email him to ask him!
Further data
Steve Koch 19:17, 6 December 2008 (EST): Your idea to pursue Deuterium some more is a good one. Please also talk with me about what you plan to do!