Rats as Animal Models in Tissue Engineering

Background
Animal models provide simplified versions and instructive representations of the biological functions of a human. The Office of Technology Assessment approximates that 17-23 million animals are used each year in the United States for research purposes [1]. All products, drugs, and inventions must be thoroughly investigated before their use on humans. Animal models allow for studies that are not feasible or ethical to produce on humans. Factors that need to be considered in choosing an animal model include the viability of implanting the tissue, how readily available the animal is, cost, and ethics. Approximately 97% of all lab animals in the United States are mice and rats [2].
Motivation
Why Rats?
Rats (Figure 1) are one of the most commonly used animal models since they are physiologically more similar to humans than other organisms. Some of their advantages include that they are inexpensive, require low maintenance, and do not carry the same ethical issues as larger animals [3]. Rats can cost anywhere from $30 to $100 depending on the weight, age, breed, and the supplier [4]. Maintenance costs approximately $5 to $10 a month for food and living, which is much less than other, larger animals. The short lifespan of rats (approximately two years) also allows researchers to study the effects of aging and other biological factors in a shorter amount of time. For example, rats' bones stop growing at around 6 to 9 months leaving a considerable amount of lifespan to be dedicated for studies of biocompability, fracture, and bone defect repair for tissue engineering and orthopedic research. Rats also serve as a reproducible and practical model due to their minimal genetic variance across several generations of breeding [5]. Despite the many advantages of rat models, many biomechanical and anatomical differences still exist between rats and humans.
Rats vs. Mice
While the primary model of choice has traditionally been the mouse, recent advances of technology have led to the use of rats as animal models in tissue engineering. The model choice is situational to the purpose and goal of the current study. In comparison to mice, rats differ in size, behavior, genetics, and physiological features.
Size
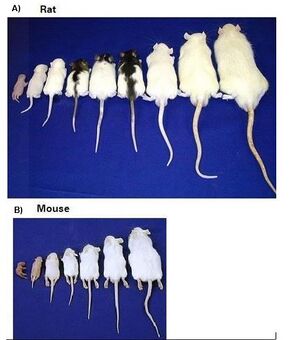
Rats (weighing on the order of hundreds of grams) typically weigh ten times greater in size compared to mice (weigh on the order of tens of grams). Rats are costlier to maintain because they require more food and living space than mice. However, their size also makes surgical procedures easier to perform leading to fewer errors and higher efficiency of money, time, and animal life. In addition, the larger surface area means larger tissues and samples can be taken from the rat. Smaller samples are more vulnerable to variability and error and require sensitive assays. Larger organs also provide greater detail into experimental studies on how much is involved and the distance a drug is travels. Figure 2 demonstrates different sizes of rats (A) and mice (B) at specific ages. The oldest rat was over 11 inches long and weighed over 361 grams, while the oldest mouse was approximately 30 grams and 3 inches long [6].
Genetics
Although rats and mice have very similar features, there are still many genomic differences between them. In 2004, the Brown Norway rat, or "Rattus norvegicus", was sequenced and revealed that some genes, including those involved in immunity, production of pheromones, and the breakdown of proteins, are expressed in rats, but are absent in mice [7]. Almost all the genes found in humans known to be linked with disease have counterparts in the rat genome. For these reasons, rats make a suitable model for disease [7].

Behavior
Although the size can create a challenge in handling rats, rats are typically easier to handle over mice due to their docile behavior. Furthermore, mice are often skittish and inconsistent. For experiments where the variability in behavior would result in unreliable results larger cohorts of mice would be needed. Previous studies have also shown that rats are more social than mice and display empathetic-motivated behavior [8, 9]. In one study, depicted in Figure 3, researchers placed a free rat in an arena with a rat trapped in a restrainer. In all cases, the free rat opened the restrainer for the trapped rat, but never opened empty restrainers [9]. When chocolate was placed in a secondary restrainer, the free rat opened both restrainers and shared the chocolate with the trapped rat [9].
Brain
Rats have larger brains than mice, and thus are more similar to humans in terms of complexity. Due to their complexity, rats are more likely to express diseases such as Parkinson’s disease, Alzheimer's disease, and autism. Rats have higher cognitive abilities than mice and perform far more reliably on learning and memory tasks.
Types of Rats
A strain is a subgroup of rats with a particular genetic variant. Outbred rats, sometimes referred to as stocks, are used especially for experiments wishing to display genetic diversity. Inbred rats have nearly identical genetics and may play an important factor in experiments that wish to exclude genetics as a factor. There are many different strains of rats, but some of the major strains are listed here.
1. Sprague-Dawley
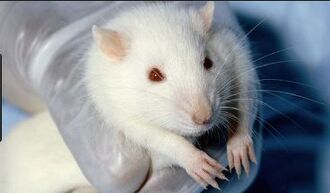
Shown in Figure 4, The Sprague Dawley rat is an outbred albino rat. The advantage of using SDs are their calm demeanor and ease of handling. Ideally for general multipurpose model, safety and efficacy testing, aging, nutrition, diet-induced obesity, oncology, and surgical model[8]. This strain also has an excellent reproductive performance [8].
2. Wistar
The Wistar rat is also an outbred albino rat. These rats tend to grow larger than SD and Lewis (LEW) rats [5]. These rats are characterized by their wide head, long ears, and a tail of lesser length to its body.
3. The Lewis (LEW)
Lewis (LEW) rats are inbred albino rats. These rats possess high insulin and growth hormone levels that contribute to their obesity [5]. They possess docile behavior and low fertility [10]. They are ideal for research about transplantation, induced inflammation or arthritis, inflammation of the brain, and diabetes [10].
4. Long-Evans
The Long-Evans rat is an outbred rat developed from Wistar females and wild gray males. The Long-Evans rat is typically a white rat with either a brown or black hood. This strain of rat is typically used for behavioral and obesity research [5].
5. Ratnude

The ratnude (RNU) rat, shown in Figure 5, is sometimes called an athymic nude rat and is either white, black, or both. Athymic rats lack a thymus, resulting in the reduction of T-cells derived from the thymus. Deficient T-cell activity in the athymic nude rats results in a reduced immune response. The immunodeficient rats are particularly important for permitting tissue transplants without rejection [11].
6. Knockout Rat
A knockout rat is a rat where a single gene is turned off through targeted mutation. Knockout rats are especially useful for studying gene function and drug discovery. The loss of function of a gene allows researchers to study the effects of a gene on a drug. Knockout rats can model diseases such as Parkinson’s, Alzheimer’s, hypertension, and diabetes. Only recently, however, has the knockout rat been developed through new technologies, such as mobile DNA and zinc-finger nuclease [12, 13]. Conventional technology for genetic manipulation relied heavily on the use of embryonic stem (ES) cells, but rat ES cells would did not survive following injection [12].
7. Transgenic Rats
Transgenic rats that have an altered genome, which contains all the genetic material of an organism. Foreign DNA (transgenes) are inserted into the genome allowing for studies of protein function and gene expression within animals. Transgenic rats can be used to model a variety of diseases, such as Alzheimer’s disease and Parkinson’s disease.
History on Laboratory Rats
Animal models, especially rats and mice, have been extensively studied for centuries. Rats, however, were the first animal domesticated solely for scientific research [14].

- 1728-1730 – Wild brown Norwegian rats, known as Rattus norvegicus, were prevalent throughout England. Rat-catchers would capture these rats and sell them for food and rat-baiting. Rat-baiting, shown in Figure 6, was a sport that involved filling a pit with rats and betting on the amount of time a dog took to kill them. Rats were eventually bred specifically for the sport. Albino rats were smaller, more docile, tolerated overcrowding in cages, and better breeders, so these rats were often not included in rat-baiting and instead used in rat shows [15].
- 1856 – Jean Marie Philipeaux was the first to be recognized for using albino rats to study adrenalectomy, the removal of one or both adrenal glands, in France [15].
- 1863 – William Scavell Savory, an English surgeon, studied nutritional quality of proteins in mammals using mixed coat colors of black, brown, and white [15].
- 1877-1885 – In Germany, Hugo Crampe was the first to confirm that genes are inherited using over 15,000 white, grey, and black rats by studying the effects of coat color [14].
- 1894 – Colin Stewart was one of the first to use rats in laboratory setting in the United States. At Clark University, Stewart studied the effects of alcohol, diet, and barometric phenomena on activity of wild and albino rats [14].
- 1906 – Henry Herbert Donaldson, from the Wistar Institute in Philadelphia, carried out selection experiments on albino rats. Donaldson aimed to standardize the rat to create reproducible studies on the growth and development of the rat’s nervous system. The breed, known as Wistar rats, have more than half of all laboratory rats have descended from [16].
- 1908 – First rat colony in America for nutritional studies by Elmer Verner McCollum [17]
- 1909 – Helen Dean King initiated the PA strain of rats, the first inbred strain [16]
- 1922 – Herbert McLean Evans and Joseph A. Long at the Institute for Experimental Biology at University of California, Berkeley studied the reproductive function of rats and developed the Long-Evans strain [18].
- 1925 – Sprague-Dawley strain was bred by Robert Worthington Dawley, a physical chemist at the University of Wisconsin [15].
- 2004 - Brown Norway rat genome was sequenced by the Rat Genome Sequence Consortium (RGSC) led by the Human Genome Sequencing Center at Baylor College of Medicine in Houston [8]
- 2007 - Development of mobile DNA for knockout rats [12]
- 2009 - Development of zinc-finger nuclease for knockout rats [13]
Tissue Engineering Applications
Neurological Disorder
As mentioned above, transgenic rat models hold many advantages over mouse models, such as the modeling of more complex functions within the brain. Alzheimer’s disease (AD) is one of the main causes of dementia, effecting approximately 3 out of 1000 individuals worldwide [19]. AD is characterized by progressive decline of mental processing and functioning.
Treatments for AD are often limited to the blood-brain barrier (BBB), a highly a highly selective semipermeable membrane that protects the brain from pathogens. Nanoparticles are often used as a delivery method for drugs. Polymeric nanoparticles are used as a drug delivery system for treatment of AD because of low toxicity, high drug capacity, and degradation rate is adjustable. In 2013, a polymeric nanoparticle of 120 nm loaded with basic fibroblast growth factor and with lectin coating to target nasal epithelium [20]. The administration of the particles within a rat model demonstrated significant improvements of spatial learning and memory capabilities [20]. The place of injection within the rats, however, would be a much more complex process within humans.
Chimeras
According to the American Diabetes Association, approximately 29.1 million Americans (9.3% of the population) had diabetes in 2012 [22]. Diabetes is still prevalent today and results from the pancreas' inability to produce enough insulin, a hormone that regulates blood sugar levels. Islet cells on the pancreas sense blood sugar levels and secrete insulin to maintain normal blood sugar levels. Islet cell transplantation is used to treat diabetes. However, several challenges limit islet transplantation, such as the severe shortage of available donors, long-term maintenance of blood glucose levels, and immune rejection of the cells. Pancreatic generation is a potential solution using patient-specific induced pluripotent stem cells (iPSCs).
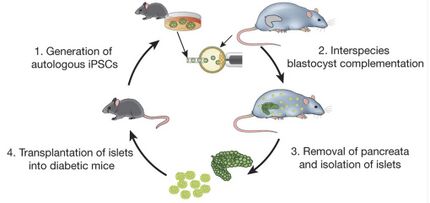
Chimeras are animals that have cells derived from a different species. Dr. Hiromitsu Nakauchi from the Institute of Medical Science in Japan and his colleagues developed a mouse-rat chimera. In 2010, Dr. Nakauchi attempted to grow a rat pancreas in a mouse with some success. Although the pancreas was functional and contained rat-derived cells, the pancreas only grew to the size of mouse's pancreas [23]. Thus, Nakauchi and his colleagues instead decided to grow a mouse pancreas within a rat.
Figure 7 demonstrates a schematic representation of the researchers' procedure. Mouse pluripotent stem cells (PSCs) were injected into rat blastocysts, a structure in the early development of the rat. The blastocyts lacked the "Pdx-1" gene needed for pancreas formation [23]. Following injection of mouse PSCs and subsequent pancreatic formation, mouse islet cells from the pancreas were isolated and transplanted into diabetic mice. Immunosuppressive drugs were used to prevent rejection of residual rat cells from transplantation. About 10 months later, the researchers inspected the islets and found that mouse's immune system removed eliminated any remaining rat cells. For over a year, blood sugar levels in the mice were remained normal.
Although the process shows promising results for organ generation and recovery of diseased tissue, the process has some limitations. The process would require organs from animals of closer size to humans. Contaminated cells deeply embedded within larger organs might also be more difficult to remove after transplanting islet cells.
Cardiovascular Tissue
Cardiovascular tissue is specialized muscle tissue consisting of the heart and blood vessels. The heart is the muscular organ that pumps oxygen and nutrient-rich blood throughout the body. Blood vessels are tubular structures that extend throughout the body and carry oxygen and nutrients to, and waste products away from, the tissues and organs.
A. Tissue-engineered Heart
In the United States, approximately three thousand people are waiting for a heart donor [22]. Currently, the only treatment for end-stage heart failure is heart transplantation. However, donor organs are scarce and transplants often run the risk of rejection from the recipient’s immune system. Hypertension, diabetes, and renal failure are the most common risks that come with heart transplantation and could potentially be avoided using a bioartificial heart [22].
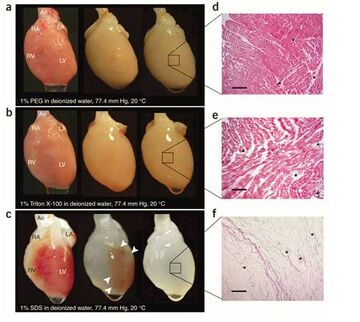
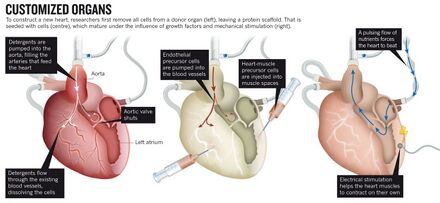
In 2008, Dr. Doris A. Taylor and her team of researchers at the Texas Heart Institute in Houston engineered a rat heart by decellularization of a cadaveric rat heart and recellularization with neonatal rat cardiac cells [24]. Decellularization was done by perfusion of detergents within the coronary, or the arteries that surround and supply the heart. The three detergents used were polyethylene glycol (PEG), Triton-X-100, and sodium dodecyl sulfate (SDS). The detergents washed away lipids, sugars, soluble proteins, DNA, and most other cellular material within the heart. As seen in Figure 8, SDS demonstrated the best results with complete decellularization leaving only the extracellular matrix. Collagen, laminin, and fibronectin were only left after the cellular material washed out from first the right ventricle, then the atria, and finally the left ventricle [24].
The scaffolds were then reseeded with cardiac cells from newborn rats and cultured under controlled physiological conditions to promote organ growth [42]. The hearts were recellularized by injection of the cardiac cells and perfusion of endothelial cells. Endothelial cells line the entire circulatory system, including the heart and the blood vessels, and are important for prevention of clotting or leakage. By day 9, the recellularized heart showed signs of contraction and was drug responsive [24]. A bioreactor was then used to imitate how the heart might function within the body of a rat. A pulsating flow of nutrients was pumped into the heart and caused the heart to beat, while electrical stimulation synchronized the beating. After 8 days, the heart was beating and pumped small amount of blood [24]. Once implanted into live rats, the transplanted hearts were not immediately rejected. Long-term studies and observation of possible side effects are, however, limited in a rat model.
The method developed by Taylor and her team of researchers, summarized in Figure 9, paved the way for generating organs, such as the liver, lung, kidney, and pancreas. In addition to whole organ regeneration, partial organ generation have many important applications, such as for patches, valves, vessels, and substrates that can be used in combination with cell therapies [24]. Current trends in engineering hearts include using 3D printing technology and developing the organ-on-the-chip.
B. Tissue-engineered vessels (TEV)
Vascularization is often a significant challenge when engineering or implanting tissue. Engineered blood vessels can be difficult because they must be able to endure a variety of mechanical properties, including varying pressures, shear stresses, sufficient regulation blood flow, and proper immune response. Human blood vessels range in size with microvessels (<1 mm), small vessels (1-6 mm), and large vessels (>6 mm in diameter) [26]. Rats are useful for studies involving small diameter (1-2mm) and length (1 cm) TEVs [26].
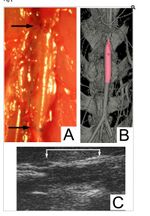
Various tissue engineering methods for creating blood vessels include 3D printing, decellularized native vessels, synthetic biopolymers, and cell sheets that create tubular structure with various degrees of success [27]. Previously engineered autologous engineered arteries had poor mechanical properties. Furthermore, the aged smooth muscle cells of previous models have less proliferative capacity and collagen production. The process of decellularization of native vessels, however, maintains mechanical properties and produces less immunologic responses. In one study, the process of decellularization was used to create an allogeneic matrix. Human smooth muscle cells were seeded onto a scaffold poly-glycolic acid (PGA) mesh over a 1-mm diameter silicone tube using a perfusion system. The human tissue-engineered vessels were decellularized and implanted into nude rats, shown by various imaging techniques in Figure 10, for the evaluation of the vessel as an arterial graft [27]. The human TEV presented similar mechanical properties to that of a typical human blood vessel. Burst pressure, or the point at maximum pressure before the blood vessel breaks, was similar to humans (1,567 ± 384 mm Hg (n = 3) versus 1,680 ± 307 mm Hg for human vein [27]. Histological anaylsis also demonstrated the growth of of endothelial and smooth muscle cells [27]. Transmission electron microscopy further demonstrated the growth of elastin in the neointima and collagen fibers [27]. Limitations of this study, however, are the small sample size and short-term studies. Long-term results are needed to investigate any late side effects, such as stenosis, or the abnormal narrowing of the blood vessel.
C. Microvascularization
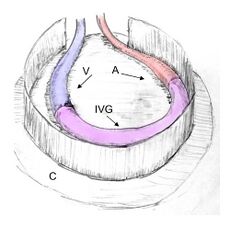
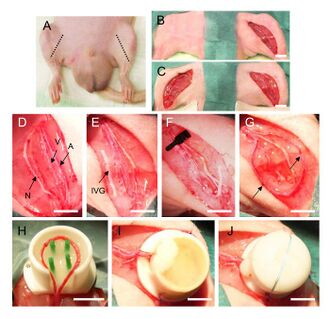
In another study, a rat model was designed to demonstrate intrinsic growth of microvessels by an arteriovenous loop (AVL) [28]. As shown in Figure 11, the loop connects the vein (V), the interpositional venous graft (IVG), and the artery (A). The loop was encased in a cylindrical chamber filled with extracellular matrix and tissue-specific cells such as muscle, cardiac, or bone cells. The chamber shape and size can be easily adjusted to fit the defect. The scaffold is then transplanted into the defect site, as shown in Figure 12. Under controlled conditions, the grafts demonstrated angiogenesis, or the growth of blood vessels, and lasted 112 days [28].
Using 3D imaging techniques (MRI, micro-CT) and immunohistology, the vascularization can be analyzed over time [28]. The AVLs were successful in demonstrating how different cell types or growth factors contribute to angiogenesis without disturbances from environmental factors, such as growth factors or invading cells. These AVLs have also demonstrated success in larger animals, such as an ovine model [28]. One limitation of this model, however, is the complexity of the surgery. The surgery is a two-step procedure and requires microsurgical skills. Infection of the inner chamber might also occur without noticing, thus sterility of whole operation and administration of antibiotics must be done with extra care. In addition, there is possible risk of thrombus formation and closure of the blood vessels.
The research group is now working on studying the effects of tumor angiogensis in mouse models. Future work will also focus on gaining a greater understanding of the biomechanical properties, how TEV grafts interact with the host after implantation, and how TEVs grow into a vascular network.
Limitations
Two of the main limitations of small animal models is that researchers cannot observe the animal model long term and the size of the animal makes surgery difficult [5]. Although rats are physiologically more similar to humans than other species, there are still many remaining anatomical and genetic differences that affect direct comparison of results. There are many differences even between strains of rats. For instance, WIS rats grow larger than LEW and SD rats [5]. Thus, when studying differences in mass and growth rate of tissue the values will vary.
Ethics
Animal models have remained a controversial issue for many years. Many individuals are concerned with the humane treatment of animals. Ideally, animal models will be replaced by better, more accurate models. The Animal Welfare Act (AWA) is a federal law that regulates the treatment of several animals for research, excluding mice, rats, and birds [29]. Furthermore, the U.S. Department of Agriculture is not required to check up on these animals or include their numbers in annual welfare reports sent to Congress [30].
Conclusion
Rats have played a critical role as animal models for various tissue engineering applications. Although many differences still exist that limit their use and reliability, rats are physiologically more similar to humans than other species. Ease of handling, availability, low cost, and low maintenance of rats are all advantages of using rats as animal models. Future studies using rats as well as other models will help to further understand biological phenomena of humans.
References
[1] U.S. Congress, Office of Technology Assessment. (1986). Alternatives to Animal Use in Research, Testing, and Education. "Washington, D.C.: Government Printing Office.
[2] Foundation for Biomedical Research. [www.fbresearch.org] (accessed Mar 12, 2017).
[3] Liebschner, M. A. (2004). Animal Models for Tissue Engineering Application Biomechanical considerations of animal models used in tissue engineering of bone. “Biomaterials”. 25(9): 1697-1714.
[4] Current Price List. “The University of Adelaide”. [7] (accessed Apr 10, 2017).
[5] Patrick, C., Uthamanthil R.,Beahm, E., and Frye, C. (2008). Animal Models for Adipose Tissue Engineering. “Tissue Engineering. Part B, Reviews”. 14(2): 167- 178.
[6] Mouse/Rat Size Chart. "Reptile Forms UK". [8] (accessed Apr 26, 2017)
[7] 2004 Release: Scientists Compare Rat Genome. “National Human Genome Research Institute (NHGRI). [9] (accessed Apr 12, 2017).
[8] Sprague Dawley. "Charles River". [10] (accessed Apr 15, 2017).
[9] Inbal, B. B., Decety1, J., Mason, M. (2011). Empathy and Pro-Social Behavior in Rats. "Science Magazine". 334 (6061): 1427-1430.
[10] Lewis Rats. "Charles River". [11] (accessed Apr 15, 2017).
[11] Festing, M. F. W. Athymic Nude Rats. “Immunologic Defects in Laboratory Animals 1”. 267-283.
[12] Kitada K, Ishishita S, Tosaka K, Takahashi R, Ueda M, Keng VW, Horie K, and Takeda J. (2007). Transposon-tagged mutagenesis in the rat. Nat Methods. 4:131-133
[13] Geurts, A. M.; Cost, G. J.; Freyvert, Y.; Zeitler, B.; Miller, J. C.; Choi, V. M.; Jenkins, S. S.; Wood, A.; Cui, X.; Meng, X.; Vincent, A.; Lam, S.; Michalkiewicz, M.; Schilling, R.; Foeckler, J.; Kalloway, S.; Weiler, H.; Menoret, S.; Anegon, I.; Davis, G. D.; Zhang, L.; Rebar, E. J.; Gregory, P. D.; Urnov, F. D.; Jacob, H. J.; Buelow, R. (2009). Knockout Rats via Embryo Microinjection of Zinc-Finger Nucleases. “Science”. 325 (5939): 433.
[14] Kuramoto, T., Satoshi, N., Masako, O., Hitoshi, N., Birger, V., and Tadao, S. (2012). Origins of Albino and Hooded Rats: Implications from Molecular Genetic Analysis across Modern Laboratory Rat Strains. “PLOS ONE”.
[15]. Baker H. J., Lindsey J. R., and Weisbroth S. H. (1979) The Laboratory Rat: Biology and Diseases. 1:2-33.
[16] Castle, William Ernest. “Complete Dictionary of Scientific Biography”. [[12] (accessed Apr 13, 2017).
[17] Day, H. G. (1974). Elmer Verner McCollum. “Biographical memoirs. National Academy of Sciences (U.S.)”. 45: 291.
[18] Evans, H. M. and Long, J. A. (1922). The oestrous cycle in the rat and its associated phenomena. “University of California Press”.
[19] Darya, H. and Poot, A. A. (2016). Biomaterials for the Treatment of Alzheimer’s Disease. "Frontiers in Bioengineering and Biotechnology". 4(49):1-10.
[20] Zhang C., Chen J., Feng C., Shao X., Liu Q., and Zhang Q. (2014). Intranasal nanoparticles of basic fibroblast growth factor for brain delivery to treat Alzheimer’s disease. Int. J. Pharm. 461, 192–202.
[21] Schrepfer, S. et al. Experimental orthotopic tracheal transplantation: the Stanford technique. Microsurgery 27, 187–189 (2007).
[22] Statistics about Diabetes. "American Diabetes Association". [13] (accessed Apr 25, 2017).
[23] Wu, S. and Hochedlinger K. (2011). Harnessing the potential of induced pluripotent stem cells for regenerative medicine. "Nature". 13: 497–505.
[24] Ott, H., Matthiesen, T.S., Goh, S.K., Black, L.D., Kren, S.M., Netoff, T.I., Taylor, D.A. Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart. "Nature Medicine". 14(2): 213–221 (2008).
[25] Maher, B. (2013). Tissue engineering: How to build a heart. “Nature”. [14] (accessed Mar 20, 2017).
[26] Hwang, S.J., Kim, S.W., Choo, S.J., Lee, B.W., Im, I.R., Yun H.J., Lee, S.K., Song, H., Cho, W.C., Lee, J.W. (2011). The decellularized vascular allograft as an experimental platform for developing a biocompatible small-diameter graft conduit in a rat surgical model. "Yonsei Medical Journal". 2011;52:227–233.
[27] Quint, C., Arief, M., Muto, A., Dardik, A., and Niklason, L.E. (2012). Allogeneic human tissue-engineered blood vessel. “Yonsei Medical Journal“. 52(2):227-33.
[28] Weigand, A., Beier, J.P., Arkudas, A., Al-Abboodi, A., Elias Polykandriotis, E., Horch, E., Boos, A.M. (2016). The Arteriovenous (AV) Loop in a Small Animal Model to Study Angiogenesis and Vascularized Tissue Engineering. "Journal of Visualized Experiments". 117.
[29] National Institutes of Health. "U.S. Department of Health and Human Services". [15] (accessed Apr 25, 2017).
[30] Lab Animal Welfare Overview. "MSPCAAngell". [16] (accessed Apr 25, 2017).