Polyelectrolyte Complexes for Microfabrication - Falcucci, Foster, and Kallin
Background
After initially being developed by photolithographyand etching, the fabrication of microfluidic devices has evolved towards soft lithography. In soft lithography, a polymer is poured onto a glass or silicon master and then peeled off to get the desired design in that polymer. The polymer of choice thus far has typically been poly(dimethyl siloxane) (PDMS) [1]. However, several issues with PDMS have given rise to interest in finding a new material that could be used to make microfluidic devices.
Issues with PDMS
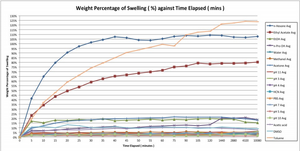
Introduction to Polyelectrolyte Complexes

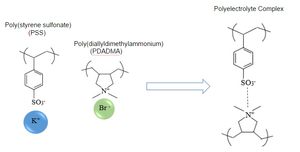
In traditional polymers, covalent crosslinkage is responsible for unique properties. However, in polyelectrolyte complexes(PECs), these crosslinkages are replaced by ionic interactions between oppositely charged polymer chains. As its name suggests, a polyelectrolyte is a polymer consisting of multiple charges due to the charges on the base monomers. For example, DNA is a polyanion. When a polyanion and polycation are mixed, they ionically interact, forming a polyelectrolyte complex. A reaction that demonstrates the formation of a polyelectrolyte complex is shown in figure 3. Table 1 demonstrates some of the most common polyanion and polycations used to form polyelectrolyte complexes. In polyelectrolyte complexes, charges are typically buried within the complex due to the electrostatic attractions between oppositely charged ions. This creates a surface that is not soluble in water. When oppositely charged polyelectrolytes in counter-ion salt solutions are mixed, a coacervate phase can be reached [5]. This ability to form coacervates, and other unique properties of polyelectrolytes give them a promising range of applications, which is continued to be explored.
Properties
PECs prove to be generally very robust materials. By employing physical crosslinking through coulombic charge pairing, PECs are extremely durable materials compared to many polymer samples. The charges form stronger bindings between chains approaching the strength and thermal/reactive resistance of chemically cross-linked polymer networks, while also maintaining the "recyclable" aspects of physical crosslinking. This crosslinking provides many of the interesting phenomena and characteristics associated with the PECs.
Plasticity
Due to the ionic nature of PECs there is a strong electrostatic force that acts between all of the constituent polymer chains and any surrounding materials. In an aqueous environment, these complexes have a ratio of open ionic sites and shielded sites [6]; this ratio is dependent on the number of available counter ions (often from simple ionic salts) and the number of ionic sites along the polymer chains [6].

Salt Content
Increasing the concentration of salt in aqueous mixture of Polyelectrolytes decreases the viscosity of the complex, up to the point where the PEC becomes full dissolved in the solvent. Removing the salt in a PEC encourages ionic site pairing, drastically increasing the viscosity of the PEC until it becomes a rigid solid which is no longer soluble in water. This phenomenon has been called "Saloplasticity."[7] Saloplasticity can be useful in creating a liquid that can be molded into a shape and then solidified as salt concentration is decreased.
Solvent Quality
Solvent work seems to be less widespread, but good solvents make the PEC act like it has a high counter ion content. Good solvents for PECs tend to be polar and have high dielectric constants (water, oxolane). Good solvents with high salt content, increase polymer molecular clumping, i.e. nano bead formation, while poor solvents with low salt content increases the interaction between neighboring polymer chains. This means that poor solvents and low salt allow for more smooth and homogenized molecular distribution [10].
"Self Healing"
Exploiting the "saloplastic" properties of PECs a self healing property is created. Self healing usually implies a sense of memory in the material, but in the case of PECs the salt content allows for this property. The addition of salt, as previously explained, shields the ionic sites in the polyelectrolyte which removes the physical crosslinking. This creates a more elastic complex and this elastic behavior allows for self healing in the material. This property also implies a method of adhering PECs; by pressing two PECs layers together with a thin layer of salt between the two layers and then removing the salt, the layers, in theory, will form crosslinks and form a single PEC sample [8].
Use in Microfluidic Devices
Although both the field of microfluidics and use of polyelectrolyte complexes has not yet matured, polyelectrolyte complexes have potential to be used for manufacturing of microfluidic devices. The
Fabrication
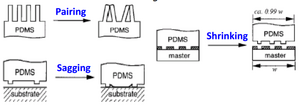
The fabrication of PEC microfluidic devices has yet to be perfected, but the most promising technique combines soft lithography and compression molding. First, a PDMS master mold would be created via soft lithography. PDMS is used as the mold material because of its soft malleable structure that can peel away from the end component without damaging microfluidic design. Then the PEC coacervate can be pressed onto the PDMS mold. Adding pressure (via topical weight) is critical because without an external force the coacervate will not take the shape of the mold and maintain a high resolution. After the liquid coacervate is pressed onto the mold, water can be added. The addition of water pulls ions out of the coacervate, allowing it to solidify. Then the PDMS can be peeled away, and the microfluidic device will be on the PEC. Spin coating can be used instead of compression molding to get thin and consistent PEC layers.
Problems
Due to the novelty of the material, there are many unknown characteristics and hurdles to overcome for the microfabrication process. The PEC coacervates are generally very sticky, which results in extreme difficulty when molding and pressing the devices. The PEC surfaces have also been shown to have a porous structure (figure 5) due to the shift from the complexes formed in the salt rich coacervate phase to the solid low salt concentration PEC material [9]. These pores may interfere with the flow in the channels of the device, which could destroy the laminar flow- the defining and most useful characteristic of microfluidic devices-within the device.

Future Work
In theory, PECs are very promising materials for the fabrication of microfluidic devices. Their impermeable, unreactive, and durable material properties seem ideal for microfludic devices, where the device scaffolding should provide little to no interference with the fluids within the channels. As a result, current and future experimentation should unveil whether or not this material is a feesible material for microfludic devices fabrication. Defining basic characteristics such as swelling properties and resolution will determine whether or not proceeding with microfabrication is feasible. In addition, further analysis is need to determine whether the porous surface will provide limitations, and if so how to evade that problem. These extrapolations combined with existing data on the adhesive properties of PECs will provide insight on the best method for fabrication.
References
1. Whitesides G, et al. Rapid Prototyping of Microfluidic Systems in Poly(dimethylsiloxane). Analytical Chemistry. 1998. 70 (23): 4974-4984. http://dx.doi.org/10.1021/ac980656z
2. Chiang, et al. Quantitative Studies on PDMS-PDMS Interface Bonding with Piranha Solution and its Swelling Effect. Micromachines. 2012. 3. 427-441. http://dx.doi.org/10.3390/mi3020427
3. Xia, et al. Soft Lithography. Angewandte Chemie International Edition. 1998. 37. http://dx.doi.org/10.1002/(SICI)1521-3773(19980316)37:5
4. Lankalapalli S, Kolapalli VRM. Polyelectrolyte Complexes: A Review of their Applicability in Drug Delivery Technology. Indian Journal of Pharmaceutical Sciences. 2009. 71(5): 481-487. http://dx.doi.org/10.4103/0250-474X.58165.
5. Wang Y, Kimura K, Dubin Paul: Effects of Micelle Surface Charge Density, Polymer Molecular Weight, and Polymer/Surfactant Ratio. Macromolecules. 2000; 33(9). 3324-3331. http://dx.doi.org/10.1021/ma991886y.
6. Wang and Schlenoff: The Polyelectrolyte Complex/Coacervate Continuum. Macromolecules. 2014. 47: 3108−3116. http://dx.doi.org/10.1021/ma500500q.
7. C.H. Porcel, J.B. Schlenoff*; Biomacromolecules, 10, 2968-2975 (2009), Compact Polyelectrolyte Complexes: “Saloplastic” Candidates for Biomaterials. http://dx.doi.org/10.1021/bm900373c
8. Reisch, A., Roger, E., Phoeung, T., Antheaume, C., Orthlieb, C., Boulmedais, F., Lavalle, P., Schlenoff, J. B., Frisch, B. and Schaaf, P. (2014), On the Benefits of Rubbing Salt in the Cut: Self-Healing of Saloplastic PAA/PAH Compact Polyelectrolyte Complexes. Adv. Mater., 26: 2547–2551. http://dx.doi.org/10.1002/adma.201304991
9. Schaff and Schlenoff. Saloplastics: Processing Compact Polyelectrolyte Complexes. Advanced Materials. 2015. 27. 2420–2432. http://dx.doi.org/10.1002/adma.201500176
10. Dubas and Schlenoff. Factors Controlling the Growth of Polyelectrolyte Multilayers. Macromelecules. 1999. 32(24): 8153-8160. http://dx.doi.org/10.1021/ma981927a
