Physics307L F09:People/Dougherty/Photo electric
Photons or Waves? The Photoelectric effect along with Planck's constant
Cary Dougherty, Linh Le
Help from Professor Koch and Professor Hjelm
Junior Lab, University of New Mexico, Albuquerque, New Mexico.
Cary008@unm.edu
Abstract
Classical physicists believed light was a propagating wave, while modern physicists have proved light is made up of photons. With this experiment, we not only proved that light is made up of photons, but we also found Planck's constant which is used in measuring the energy of photons. We measured the the time it took for the stopping voltage to reach a certain percentage of its full potential using different intensity filters for each color in the mercury spectrum. This method proves that light is dependent on frequency not intensity, which means it is made up of photons. Also by measuring the stopping voltage of electrons which escape a cathode tube by the energy of photons, we can find Planck's constant. By using a simple graph and finding the slope, we can discover the value of Planck's constant and the energy of a photon.
After further experimentation after our first day of recording, we were able to come up with consistent values of Planck's Constant. And after combining them with a weighted average, we came up with a value for Planck's Constant along with the Work Force:
h = 7.17(+/- 0.010)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.58(+/- 0.041)*10^-19 J
With the accepted value as 6.626 068 96(33)*10^-34 J s (meaning the uncertainty is 0.000 000 33*10^-34 J s) ( 6)
While relatively close and only a small degree of error, we recorded similar values of this constant over and over, signifying a systematic error in our data.
Introduction
Scientists have long theorized about whether light is a wave or is made up of photons. Ever since it has been proven that light depends only on its frequency, we know that it is made up of photons. The classical theory of light being a wave was tested by the "ultraviolet catastrophe."(1) This catastrophe occurred when a classical physics model, called the Raleigh-Jeans Law [[math]\displaystyle{ f(\lambda) = \frac{2 \pi c k T}{\lambda^4} }[/math]](4), stated that theoretically the amount of light emitted from a blackbody would increase considerably when the wavelength decreased. However, the experimental data showed that the amount of light actually decreased closer to zero. (7) A catastrophe in the making.
Max Planck (1858-1947)(8), a turn of the century German physicist, was very interested in the emission and absorption of light. (1) His widely known work on black body radiation led him to the theory of quantum physics, or the theory of light being made up of photons. (5) In other words, electromagnetic light could be emitted in quantized form following the equation [math]\displaystyle{ E = h\nu }[/math](5), where h is Planck's constant.
This was an enormous step in modern physics. The fact that he theorized that light was directly related to frequency completely disrupted the classical theory of light, which stated it was related to the light's intensity. Not only did it make the assumption that light is directly related to frequency and not intensity, but that in turn proved light was not made up of waves, but of photons, introducing Planck's theory of quantum physics.
Later on, Albert Einstein made the discovery of the photoelectric effect. He theorized that photons making contact with metal, if had enough energy, would eject an electron by a work force and would have as much kinetic energy as the original photon. (2) Setting up the equation:
[math]\displaystyle{ E = KE+\phi }[/math] (2)
Both Einstein and Planck received Nobel Prizes for their work with quantum radiation. Planck for the theory of quantum radiation in 1918, and Einstein after for his work with the photoelectric effect in 1921.
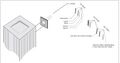
Because of these discoveries, we can use the mercury light and the h/e apparatus to discover Planck's constant using the photoelectric effect, which is the energy emitted as a photon. (see Fig. I - 1 and I - 2)
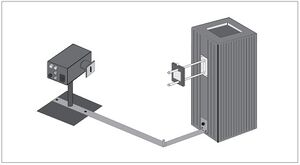
Using filters on the h/e apparatus for pure light, we can discover the stopping potential of an electron, in doing so we can find the kinetic energy and graph it with frequency to find the work function, [math]\displaystyle{ W_0 }[/math], and h, Planck's constant. (see Fig. MM - 3)
-
Fig. I - 2 photograph of entire apparatus
Methods and Materials
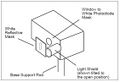
The apparatus is made up of a mercury vapor light source OS-9286, diffraction grating, and an h/e apparatus AP-9368 (made up of a white reflective mask, a light shield, and a photo diode inside) (see Fig. MM - 1 and Fig. MM - 2). The reverse voltage is powered by batteries or a +9 V and -9 V power source. A voltmeter is also necessary for this experiment. We used a Digital voltmeter SE-9589. Also a form of stopwatch is needed for experiment 1. We used my cell phone which is a Samsung t509.
-
Fig. MM - 1 The specific models of the light source and h/e apparatus
-
Fig. MM - 2 The h/e apparatus with labeled parts
Proving light is a photon
Experiment 1
The photons emitted by the mercury light source pass through the diffraction grating and split into each individual color which we could focus separately towards the h/e apparatus. The light then entered the h/e apparatus through a filter with different intensities labeled on it, 100% intensity to only 20% intensity (We also used green and yellow filters on the matching light to filter out any excess light from other sources). The light passed the filters and entered through the slit in the white shield and hit a cathode tube ejecting photoelectrons onto an anode. The photoelectrons escaped the cathode with a minimum energy or work force [math]\displaystyle{ W_0 }[/math], leaving the cathode with a max kinetic energy, KE. The photoelectrons escaping create what is called a "photoelectric current," which charged a capacitance. Eventually the capacitance reached the stopping potential of the photoelectrons, and the photoelectric current stabilized between the cathode and anode, and we could record the maximum voltage off the voltmeter. This stopping voltage or potential is directly related to the kinetic energy of the electron. We measured how long it took for the stopping potential to go from zero to the full stopping voltage. However, we only measured how long it took to reach about 90% of the recorded stopping voltage because in some cases it took a very long time to reach the max voltage, therefore we still achieved the same results but recorded much smaller time frames. We passed yellow, green, violet, and UV light through each filter and measured how long it took to rise.
Finding Planck's Constant
Experiment 2
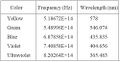
Now without the intensity filter, we measured the stopping voltage of each color and recorded it. The diffraction grating separates the colors into different orders of maxima (see MM- 4). We recorded the stopping voltage twice for two sets of maxima. Using the voltage and the equation KEmax = qVstopping, we found the KE, kinetic energy. Plotting KE vs. frequency we found the slope using a least-square fit program off excel which we called h, Planck's constant, and the work force, [math]\displaystyle{ W_0 }[/math]. (see Fig. MM-3)
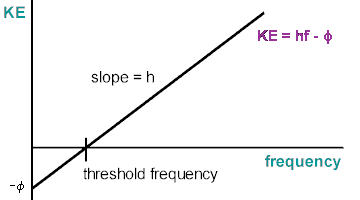 [Fig. MM- 3 Slope, h, Planck's Constant 3]
[Fig. MM- 3 Slope, h, Planck's Constant 3]
[KE = kinetic energy, h = Planck's constant, q = charge of an electron, V = voltage to stop the electron, Φ = work function, [math]\displaystyle{ W_0 }[/math] (min. energy), f = frequency (found in the lab manual.1)(also see Fig. MM - 5)]
-
Fig. MM - 4 The different orders of maxima refracted by the grating
-
Fig. MM - 5 The different frequencies for each color found in the lab manual
Later on two other classmates and I went back and brainstormed a while on how we could achieve better results in our experiment on finding Planck's constant. Our best ideas were darkening the room as much as possible, and also a better filter than what we were using. we ended up finding a lightly smoked poly-carbonate piece of glass that we placed in front of the h/e apparatus blocking out any form of UV light that was obscuring our other data for the other colors. This proved to be very affective considering our green color calculations seemed to be off by a great deal. (New Data can be found here. ND)
Results
Experiment 1
After Linh and I recorded the rise time of each color in the spectrum shown along with each intensity on the filter (100%, 80%, 60%, 40%, 20%), we used excel to plot intensity vs. time (in seconds) to determine if in fact light is directly related to its intensity or if light only depends on frequency.
-
Fig. R - 1 The UV plot of time vs. voltage
-
Fig. R - 2 The blue/violet plot of time vs. voltage
-
Fig. R - 3 The green plot of time vs. voltage
-
Fig. R - 4 The yellow plot of time vs. voltage
After looking at our yellow light graph, we decided it wasn't in sync with the other graphs. We took another measurement, and this time the results looked much better. (see Fig. R - 5)
After viewing our graphs, we can determine that the intensity filter does slow the time to reach the maximum stopping voltage. However, it does not truly affect the maximum voltage itself. We can therefore conclude that the intensity filter does slow down the photons entering the h/e apparatus, but does not affect the maximum energy of the electrons that escape the cathode. This observation supports the quantum light theory. (Raw data for this experiment can be found here.)
Experiment 2
After measuring the stopping voltage of a number of electrons, we were able to find the kinetic energy with the equation from methods and material (KEmax = qVstopping). Using the kinetic energy and the frequency of light found in the lab manual (see Fig. MM- 5), we were able to graph it and find a slope, h. The diffraction grating by the light source refracted two sets of maxima. The first, brighter maxima was the subject of two of our data sets, while the second, dimmer maxima was the subject of two more of our data sets. (see Fig. MM - 3). (Raw data for graphs of Experiment 2 can be found here) As you can see in Fig. R-6 to R-9, we recorded a stopping voltage for the colors yellow, green, violet, and UV (found in raw data above), and after we found the KEmax, we plotted them on an graph with one axis KE, kinetic energy, and one axis frequency. We used excel to form a least-square fit line and to find the slope of that line. It gave us the slope of the line and the y-intercept which is the work function mentioned in methods and materials above. (see Fig. MM - 3)
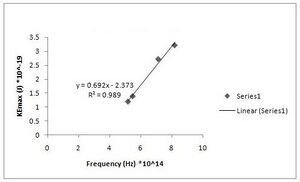
h = 6.92(+/- 0.051)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.373(+/- 0.337)*10^-19 J (Fig. R - 6)
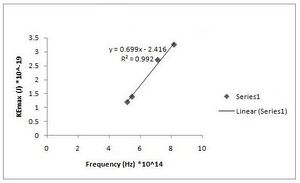
Fig. R - 6 is the 1st order of maxima and our first data set.
h = 6.99(+/- 0.061)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.416(+/- 0.401)*10^-19 J (Fig. R - 7)
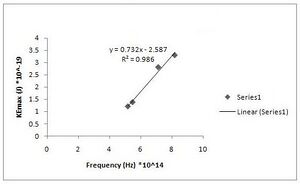
Fig. R - 7 is the 2nd order of maxima and our first data set.
h = 7.32(+/- 0.042)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.587(+/- 0.281)*10^-19 J (Fig. R - 8)
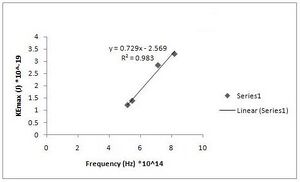
Fig. R - 8 is the 1st order of maxima again but our second data set.
h = 7.29(+/- 0.067)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.569(0.442)*10^-19 J (Fig. R - 9)
Fig. R - 9 is the 2nd order of maxima again but our second data set.
New Data
New Data for Planck's constant:
lightly smoked poly-carbonate glass
| Color (1st Order) | Test 1 (V) | Test 2 (V) right | |||
|---|---|---|---|---|---|
| Yellow | .725 | .726 | |||
| Green | .858 | .858 | |||
| Blue | 1.510 | 1.501 | |||
| Violet 1 | 1.717 | 1.698 | |||
| Violet 2 | 2.112 | 2.075 | |||
| Color (2nd Order) | |||||
| Yellow | .753 | .741 | |||
| Green | .849 | .866 | |||
| Blue | 1.498 | 1.508 | |||
| Violet 1 | 1.691 | 1.696 | |||
| Violet 2 | 2.106 | 2.098 |
Frequency
yellow - 5.18672*10^14 Hz
green - 5.48996*10^14 Hz
blue - 6.87858*10^14 HZ
violet 1 - 7.40858*10^14 Hz
violet 2 - 8.20264*10^14 Hz
New Data Results
1st order, test 1
yellow - KEmax = 1.16*10^-19 J
green - KEmax = 1.3728*10^-19 J
blue - KEmax = 2.416*10^-19 J
violet 1 - KEmax = 2.7472*10^-19 J
violet 2 - KEmax = 3.3792*10^-19 J
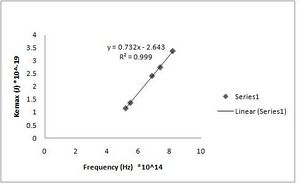
h = 7.32(+/- 0.010)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.64(+/- 0.068)*10^-19 J (Fig. ND - 1)
2nd order, test 1
yellow - KEmax = 1.2048*10^-19 J
green - KEmax = 1.3584*10^-19 J
blue - KEmax = 2.3968*10^-19 J
violet 1 - KEmax = 2.7056*10^-19 J
violet 2 - KEmax = 3.3696*10^-19 J
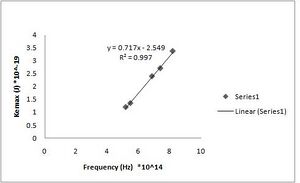
h = 7.17(+/- 0.019)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.55(+/- 0.127)*10^-19 J (Fig. ND - 2)
1st order, test 2
yellow - KEmax = 1.1616*10^-19 J
green - KEmax = 1.3728*10^-19 J
blue - KEmax = 2.4016*10^-19 J
violet 1 - KEmax = 2.7168*10^-19 J
violet 2 - KEmax = 3.32*10^-19 J
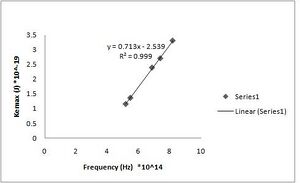
h = 7.13(+/- 0.010)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.54(+/- 0.069)*10^-19 J (Fig. ND - 3)
2nd order, test 2
yellow - KEmax = 1.1856*10^-19 J
green - KEmax = 1.3856*10^-19 J
blue - KEmax = 2.4128*10^-19 J
violet 1 - KEmax = 2.7136*10^-19 J
violet 2 - KEmax = 3.3568*10^-19 J
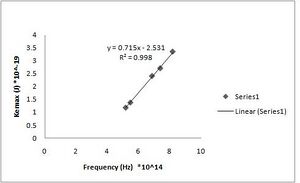
h = 7.15(+/- 0.014)*10-34 J s [math]\displaystyle{ W_0 }[/math] = 2.53(+/- 0.097)*10^-19 J (Fig. ND - 4)
After viewing the uncertainties of each new value we found, I concluded it was a good idea to combine them using a weighted average. In this way i could average the values together instead of just taking the average. The weighted average is:
- [math]\displaystyle{ \bar{x} = \frac{ \sum_{i=1}^n w_i x_i}{\sum_{i=1}^n w_i}, }[/math]
where [math]\displaystyle{ w_i = \frac{1}{\sigma_i^2}. }[/math]
and the uncertainty is [math]\displaystyle{ \sigma_{\bar{x}}^2 = \frac{ 1 }{\sum_{i=1}^n 1/{\sigma_i}^2}, }[/math]
Using these equations, I was able to come up with a single value for Planck's Constant and the Work Force:
h = 7.17(+/- 0.010)*10^-34 J s [math]\displaystyle{ W_0 }[/math] = 2.58(+/- 0.041)*10^-19 J
Conclusion
With the accepted value as 6.626 068 96(33)*10^-34 J s, (meaning the uncertainty is 0.000 000 33*10^-34 J s) ( 6) I felt we were very close. And since our each of our values were very close together I can also conclude there was some sort of systematic error involved.
I also wanted to see how far away I actually was from the actual values, so I used:
[math]\displaystyle{ %error= \frac{|Actual-Experimental|}{|Actual|}x100 }[/math]
Our values was had only 8.21% error.
The systematic error could've risen from bad filters in my opinion. We discovered the green filter we were using did not really affect the data we were taking. Which is the reason we decided to use the poly-carbonate glass. This helped filter out a lot of the UV light. In any case it didn't work perfectly. I think with state of the art filters and a very dark room, this experiment could wield excellent results for the students.
Acknowledgments
I would like to thank Linh, of course, for helping with these experiments and data collection.
Also I would like to thank Professor Koch and Professor Hjelm for answering any questions we might have had and assisting in any way they could.
Thanks to Openwetware for the platform for presentation and storing data.
Thanks to the past lab manual for keeping the experiment clear and making it less confusing.
And lastly i want to thank Max Planck for starting this entire experiment and discovering one of the greatest constants in physics.
References
2.Einstein, Albert (1905a), "On a Heuristic Viewpoint Concerning the Production and Transformation of Light", Annalen der Physik 17: 132–148
3.Colwell H. Catharine (1997) Physics Lab online
4.Rayleigh-Jeans Law Wikipedia Article (2007)
5.Planck, Max (1901) On the Law of Distribution of Energy in the Normal Spectrum