Physics307L F09:People/Barron/labsum~Planck
Balmer Series Lab Summary
Here is the lab manual page.
Here are my lab notes.
Partner: Justin Muehlmeyer
Introduction
Most people will immediately invoke "[math]\displaystyle{ E=mc^2 }[/math]" when asked for what Einstein received his Nobel Prize. As scientists in training, we know that the Prize was instead for his work on the Photoelectric Effect, and this famous relation:
[math]\displaystyle{ E = h\nu + \omega_0 }[/math]
...which describes the energy of an electron ejected from a material due to an atom's interaction with a photon of energy [math]\displaystyle{ h\nu }[/math].
[math]\displaystyle{ h }[/math] is Planck's constant, and [math]\displaystyle{ \nu }[/math] is the frequency of the photon. [math]\displaystyle{ omega_0 }[/math] is the work function, which is a material-dependant value of energy the energized electron has to overcome in order to eject. This experiment is designed to ascertain [math]\displaystyle{ h }[/math].
Setup
A detailed, illustrated discussion of the experimental setup can be found in Dr. Gold's manual.
Final Results
Experiment 1 - Preliminaries
This experiment is used to show the physical realities of Planck's constant and the photoelectric effect qualitatively. We take data on the stopping potential for a frequency, then see how much time the capacitor in the apparatus takes to charge to the stopping potential. Rinse and repeat for another frequency.SJK 18:31, 18 December 2008 (EST)

Very nice graphs.
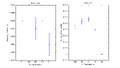
| Vstop vs. Transmission | Charge Time vs. Transmission | ||
|
|
Error bars represent 68% confidence intervals.
As one can see, the charge time increases with decreased transmission. This doesn't make sense at all using the photon theory of light. If this experiment strictly obeyed the theory of photons, the stopping potential of the apparatus would be the same regardless of how intense the incident light was. Hence, the charge time would be the same for any transmission rate. If the experiment obeyed the classical theory, charge time should increase with decreased transmission - the supposed amplitude of the waves should have decreased, so the "charged" photoelectrons ejected should have been less energetic. The observed trend correlates with the classical theory of light. SJK 18:33, 18 December 2008 (EST)

??? I think charging time increasing for decreasing amplitude is consistent with either theory. Whereas the merely slight change in stopping potential is supported by photon theory only.
The voltage reading trend to drop with decreased transmission is at odds with the photon theory as well, via the above argument. The most plausible explanation for the drop I can think of involves the current leak from the capacitor in the apparatus. The manual creates the analogy of the capacitor being a bathtub being filled with "different flow rates while the drain is partly open." If the classical waves can't charge enough electrons quickly enough, the current leak could overcome the charging under low transmission filters.
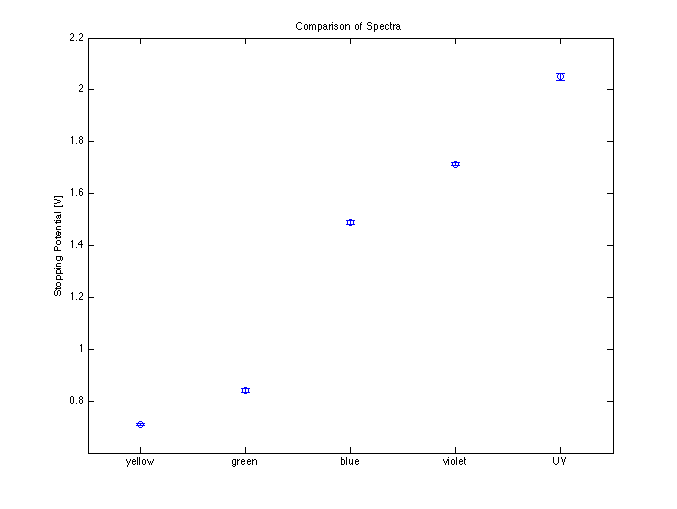
The next preliminary experiment compares stopping potentials for differing frequencies.
Clearly, the frequency axis increases to the right, as does the stopping potential. This supports the photon theory of light, since the intensity should be fairly constant over the spectra, indicating that intensity doesn't alter the energy of excited photoelectrons.
Experiment 2: Planck's Constant
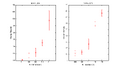
Here we measure the stopping potential for all spectra in both the first order and second order series.
Via this illustration of measured spectra from the lab manual:
From Einstein's famous relation:
[math]\displaystyle{ E = eV + \omega_0 \longrightarrow eV = h\nu - \omega_0 }[/math]
I can find h via linear regression.
| Numerical Representations | |
|
1st-Order Spectra:
|
2nd-Order Spectra
|
| accepted h = 4.14e-15 [eV s][1] | |
| accepted ω0 = 1.36 ± 0.08 [eV][2] | |
| Graphic Representations | |||
| |||
The accepted values of ω0 and h do not coincide with my 68% confidence interval, but qualitatively are not far off. If one looks at my lab notes, there seems to be significant systematic error in ascertaining the correct stopping potential for either experiment. The most obvious anomaly is the ever-rising trend of voltage measurements in the 2nd-order part of experiment 2. This could result from an electronics design flaw, or perhaps have something to do with the steady rise in temperature of the Hg lamp. By the end of the experiment, the lamp was too hot to touch. It is not obvious how that would change the stopping potential reading, however. The diffraction grating would still separate the component frequencies of light into individual spectra, but the intensity of light would probably increase. An increase in stopping potential as a result of higher intensity would correspond to a classical wave theory of light, which is troubling.