Physics307L F07:People/DePaula/Milikan Oil Drop
Theory
Because mineral oil is conductive it has free electrons. This key property of mineral oil allows for the following experiment. We will use an atomizer to spray droplets of mineral oil between two brass plates. The two plates have a high voltage, or electrical potential field, between them, which manipulates the fall time of the droplets. We must assume that the charge of an electron is constant, and therefore any measurements we receive for the charge will be a multiple of that intrinsic value.
Set Up
We set up the experiment according to the directions in the Milikan Pasco Manual. At one point we were wondering what we needed in order to clean the metal plates in the chamber assmbly. Anne called the Pasco support line, and they informed us that a soft cloth would do the trick. We used the inside of a soft sweater. The following image from Wikipedia gives a good description of the aparatus: 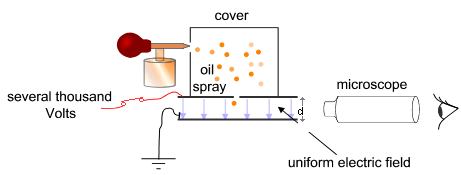
We had to place the entire experiment set up on a stack of books so that our necks would not be too strained during the data collection process. We leveled the experiment so that our droplets fall parallel to the grid lines in the eye piece, otherwise we would have incorrect velocity values. We take the chamber assembly apart, and measure the plastic separator, so we know the exact distance between charged plates. We focus the eyepiece and reticle by placing a focusing wire in the chamber, and turning knobs on the eyepiece until the side-lit wire is in sharp focus. After total reassembly we connect the High Voltage to the plates, and set the plate charge to "grounded". A multi-meter is now connected to the internal thermistor so we can monitor the internal temperature throughout data collection. The experiment is now ready to go.
Procedures and Methods
We are looking to isolate the charge of an electron, when this experiment was originally conducted by Millikan himself, the quantification of charge on a subatomic particle was only theory. The idea behind the actual experiment is to spray an oil into a chamber equipped with an extremely high voltage, in an attempt to manipulate any free electrons in the oil. Mineral Oil is used because it can resist evaporation against the heat lamp in small droplet form. This forces the droplets to remain constant in mass, and therefore the perfect bodies to experiment on. The charged plates determine whether or not the drops fall/rise. By reversing the charge on the plates the droplets should change their velocity, and head off in a different direction. We have limited the possible directions of movement to essentially up and down. These particles are not in a vacuum, so there is some intrinsic air resistance. Fortunately these droplets are small enough that they reach terminal velocity in a few milliseconds. It is incredible that the attractive charge between a few electrons and the high voltage field is enough to resist the fundamental pull of gravity. This gives a good example of how important it is to understand the properties of electrons. These sub-atomic particles resist gravity at all times, if it was not for this resistance matter would have no cohesiveness.
We choose a droplet, of medium size, whose fall time between large grid separations is sufficient to accurately measure. We measure the time it takes to move between one separation or .5mm, then we charge the metal plates, in an attempt to accelerate the particle upwards. If we charge the top plate, and the particle begins to fall faster, then we know that the particle itself is of the same charge as the plate, so a simple reversal of plate charge causes the particle to rise against gravity. We take multiple measurements of one particle, rising and falling, in an attempt to obtain an accurate average fall and rise time. After taking about 10-20 measurements for one droplet, we expose it to an Alpha particle source in an attempt to reverse the charge on that single particle. In this case the source is Thorium-232. Theoretically we use alpha particles to strip electrons from the droplets, not affecting the fall time, but instead affecting the charge attraction that gives way to the particle's rise. There is not enough mass lost in the electron theft to influence the fall time. We did not have great success with this step, it did not seem to affect our droplets greatly.
Important Constants and Formulas
*[math]\displaystyle{ d=7.59\times 10^{-3} m }[/math] (Plastic spacer *[math]\displaystyle{ \rho=8.86\times 10^2 \frac{kg}{m^3} }[/math] (Oil Density) *[math]\displaystyle{ g=9.8 \frac{m}{s^2} }[/math] (Accepted value for Gravity) *[math]\displaystyle{ l=1.0\times10^{-3} m }[/math](Length of one droplet pass)
* η=(Air Viscosity) *[math]\displaystyle{ v_f=\frac{l}{t_f} }[/math] (average falling velocity of oil droplet ,without electric field) *[math]\displaystyle{ v_r=\frac{l}{t_r} }[/math] (average rising velocity of oil droplet, with electric field) *[math]\displaystyle{ a=\sqrt{\left(\frac{b}{2p}\right)^2+\frac{9\eta v_f}{2g\rho}}-\frac{b}{2p} }[/math] (droplet radius) *[math]\displaystyle{ q=\frac{4}{3}\pi\rho g d\frac{a^3}{V}\frac{\left(v_r+v_f\right)}{v_f} }[/math] ( oil droplet charge {Coulombs})
Results
We have data for four droplets, these four seemed to have the best, and most consistent data.
The charge for each droplet is as follows:
A 8.27827E-10
B 9.76101E-10
C 1.25709E-09
D 1.76178E-09
We can assume that C, and D are smaller droplets with fewer electrons, and A and B are larger droplets, so dividing the larger by the smaller, we should come to a reasonable value for the fundamental charge.
B/A=1.17
C/A=1.51
D/A=2.13
Anne suggests that we use Kyle's system to determine the best value of q.
The value obtained by using Kyle's method according to Anne's Summary is 4.14E-10 e.s.u. The accepted value is 4.83E-10 e.s.u. Anne's relative error is 13.75%
Doing my own calculations I find that my numbers are in agreement with Anne. I received a value of 4.16E-10 e.s.u., which is off by 13.98% in relative error.
Improvments
It did not seem that our Thorium-232 really had an effect on the droplets, so next time it would be nice to use a more potent source of alpha particles. The potency may not be the problem, it could be that our technique was not correct. This part of the lab is missing from this summary because it did not make any real usable difference in our data. It would be nice to follow a few more than just 4 droplets, but only 4 trials were worthwhile. It would help to have multiple droplets of similar charge, to try and reduce random error effects. More frequent temperature readings of the chamber may lead to a better data calibration system, accounting for heat influence.