Physics307L:People/McCoy/FFormal
SJK 16:19, 15 December 2008 (EST)

This is a really good report, good work! Due to the end-of-semester time crunch, my comments below are incomplete. I particularly liked your discussion of the initial discovery of electron waves and your references to classic papers. Your data and analysis are very careful and your figures and captions are very nice.
An analysis of the Graphite Crystal Lattice from Electron Diffraction
Experimentalist: Chad A. McCoy
University of New Mexico
Department of Physics and Astronomy
Albuquerque, NM 87131
cmccoy1@unm.eduAbstract
In this experiment, my primary objective was to measure the internal lattice spacing of graphite using the properties of electron diffraction. The secondary objective of the experiment was to demonstrate the validity of the de Broglie hypothesis, thereby proving the existence of the particle-wave duality.
In doing this experiment I measured the diameter of the rings in the diffraction spectra from the electrons passing through the spacings in the polycrystalline graphite lattice, thereby achieving both objectives as the existence of a diffraction spectra itself demonstrates the validity of the de Broglie hypothesis. Having measured the height of the rings, I calculated the internal lattice spacing of graphite to be d=.110(10)nm and d=.208(7)nm, which when compared to the accepted values of d=.123nm and d=.213nm are both smaller than the accepted result but valid to 80% confidence, as that is the interval corresponding to the less accurate result of .110(10)nm.
Introduction
SJK 16:05, 15 December 2008 (EST)

This is a really good introduction to the topic of electron diffraction. I spent way too much time reading the references you found, including the famous Davisson and Germer 1927 paper which I wish I had more time to read! Very good job on these references. Missing is a broader introduction to your paper...For example, just a little bit to introduce what research YOU will present.
The basic fundamental concept behind electron diffraction originated from the doctoral dissertation of Prince Louis-Victor de Broglie in 1924. In his dissertation, de Broglie evaluated the relation between the momentum and energy of a photon, hypothesizing that the energy and momentum of particles was related in the same way. From this hypothesis, the concept of the de Broglie wave was created with the relationship [math]\displaystyle{ h=p\nu }[/math] applied to particles along with its previous application for photons. The relationship that de Broglie based his hypothesis on corresponded with Einstein's assumption that the relationship [math]\displaystyle{ E=hf }[/math] was correct. From this assumption de Broglie was able to formulate his hypothesis, with it's creation being specific for light particles, specifically the electron, and after it was proved the expansion to heavy particles followed.[1]
Because the de Broglie relationship allowed for the determination of a fixed wavelength for a particle, experimentalists determined that according to the de Broglie relationship the wavelength of an electron after accelerating through an electric field would be on order with the crystalline spacing in most elemental solids. Because the wavelength is of the same order as the size of the crystal lattice, experiments using the diffraction of photons using a grating or single/double slit diffraction would be relevant, because objects diffract when they interact with spacing on the same order as the wavelength of the particle. Allowing for the interaction between photons and crystals, W.H. and W.L. Bragg found that it is possible to accurately determine the spacing of the crystals based on the wavelength of X-rays, thereby being the pioneers of X-ray crystallography.[2] Bragg's experiment came out of the experiment of Fredrich, Kipping, and Laue, in which they passed x-rays through a crystal and saw a series of spots on a phosphorescent plate where the X-rays hit. Looking at their results, Bragg noted that it appeared as if the spots were at the corners of adjacent pencils, which he determined was the arrangement of atoms in a crystal, with the reason for the appearance of the dots unknown (until his later experiment).[3]SJK 15:38, 15 December 2008 (EST)

Refs 2 & 3 are actually letters to the editor from Bragg. So, they're not strictly speaking "peer-reviewed" original research papers. But being from Bragg, they're pretty cool references. I'm not sure quite what the pencil terminology means...but I think your rewording may not be accurate.
The first such experiment was carried out by Davisson and Germer, where after an initial experiment by Davisson and Kunsman in 1921, in which they were doing an experiment similar to the Rutherford experiment and scattering electrons due to the charge distribution in nickel. In that experiment, they saw multiple scattering angles, not all of which could be explained by the Rutherford model of charge distribution, be the difference because of the electron shells in the nickel or the de Broglie relationship.[4] Because of this, Davisson and Germer went into a deeper investigation starting in 1925, especially after an "Accident" in the lab where they observed that the electrons interacted with the nickel crystal as if they were normal to the surface, and calculating the wavelength from Bragg's Law, they found that the wavelength corresponded to the de Broglie relationship of [math]\displaystyle{ \lambda=\frac{h}{mv} }[/math].[5]
Along with the use of high-energy electron diffraction as discovered by Davisson and Germer, the principle of Low-Energy Electron Diffraction (LEED) wasn't fully developed until the mid-late 1960s with the work of McRae, Beeby, Holland and others. Van Hove describes the use of LEED for the diffraction off the surface of crystals, as it has a higher surface sensitivity than HEED. Using this principle, researchers have been able to map the surface features of crystals, such as unit cells, present at reconstruction sites on the surface, or incommensurate and disordered overlayers that do not have the same structure as the substrate and the core of the crystal.[6]
From its discovery on to today, the concept of electron diffraction has seen many uses in crystallography, be it through high or low energy diffraction, as the two functions can work together to allow for a more detailed image of crystalline spacing, both on the surface and in the core of a crystal, than what can be resolved using other methods.
Materials and Procedure
Data Procedure

Image Courtesy Physics307L[7]
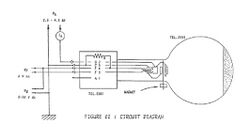
Image Courtesy Professor Gold's Lab manual[8]
To set up the experiment, I first got the electron diffraction apparatus (consisting of a Teltron 2555 electron diffraction tube and a Teltron 2501 universal stand) out of the storage closet. Then, going from the picture seen in Figure 1, I got the Teltron Limited 813 kV power unit and the Hewlett Packard model 6212B power supply so as to connect the power supplies to the apparatus. Using 4mm Banana cables, I hooked the apparatus up to the two power supplies, wiring a WaveTek Meterman 85XT digital multimeter in series between the high voltage ports on the power supply and the anode, so as to monitor the current. I had the multimeter wired in series so that i could monitor the current going to the anode, as it stated to not let it exceed .25mA, because the thin coating of graphite making up the diffracting grating would heat and puncture if the current running to it is too high.
With the apparatus connected according to Figures 1 and 2, I turned the Teltron power supply on, with the slider controlling the anode voltage set to zero, allowing the heater to warm up and begin emitting electrons. After waiting for approximately 1 minute, I slowly turned the anode voltage up using the slider, stopping at the maximum value, which corresponds to having the high voltage set at 5kV. After turning on the HP power supply, I began taking data points at the 5kV setting, with the odd numbered points corresponding to a bias voltage at 10V and the even numbered points corresponding to the bias voltage at 5V. To take the data points, I measured the ring height using analog calipers accurate to .001 inches and then converted the values to metric. (Note: As the bias voltage affects the overall voltage, I needed to slightly change the voltage on the high voltage supply to have the overall voltage remain constant.) Having done this, I repeated the data sets at 4.5kV, using bias of 5V and 2.5V, at 4kV, using bias of 2.5V and 0V, at 3.5kV, using bias of 1V and 0V, and at 3kV, using bias of 0V for all measurements, because it was too dim to see the ring without this bias.
On the day for taking additional data, I took five data values for each set corresponding voltage settings of 4.75kV, 4.25kV, 3.75kV, 3.25kV, and 2.75kV. With these five data sets, I used Carrera Precision digital calipers to measure the ring heights for both the inner and outer rings, and followed the same procedure in changing the bias between measurements as in the first day of the experiment, with the bias corresponding to the 4.5kV, 4.0kV, 3.5kV, 3.0kV, and 3.0kV respectively.
Analysis Procedure
Having taken all the data I extrapolated it to fit a flat surface, rather than a curved surface like the inside of the diffraction tube. In order to do this, I used the symbolic mathematics program Maple 11, and evaluated the extrapolation using the curvature of the tube, the length from the graphite to the end of the tube and the height of the rings on the curved surface. Doing this I came up with the equation: [math]\displaystyle{ D=2{L} \mathrm{tan}(\frac{ {\mathrm{arcsin}}(\frac{h_{0}}{C})}{2}) }[/math] where L is the length of the tube, h_0 is the height of the ring on the curved surface, measured from the center of the tube, and C is the curvature of the surface of the tube.
With the formula developed, I was able to do the extrapolation of the heights using both Matlab 7.0.4 and Microsoft Excel 2007, and with the extrapolated height, I was able to calculate the lattice spacing and its error. To do so I used the formula that: [math]\displaystyle{ d=\frac{2{L}{h}}{D\sqrt{2{m_{e}}{e}{V_{a}}}} }[/math] where d is the spacing, L is the length of the tube, h is Planck's constant, D is the extrapolated height, m_e is the mass of the electron, e is the fundamental charge, and V_a is the anode voltage.
To calculate the error, I calculated propagated the error in my measurements through the equation and added in quadrature the error in the length of the tube, also propagated through the calculations to determine the total error in the measurement from the different data values.
Figures
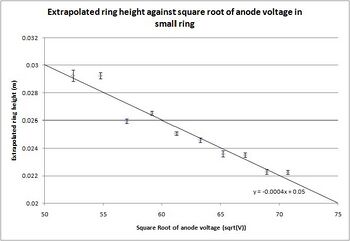
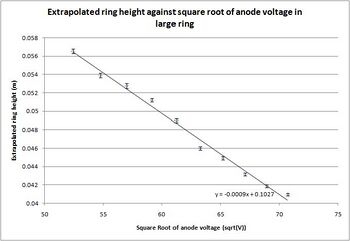
Steve Koch 16:10, 15 December 2008 (EST): Very nice data!
Results and Errors
SJK 16:12, 15 December 2008 (EST)

you have very good data and this table would be very valuable to other researchers. I just want to point out that in a "real" publication, you would very likely be forced to bury this information in the "supplementary online material" and would not be likely to post a table of your raw data.
Data
| 5 kiloVolts | 4.5 kiloVolts | 4 kiloVolts | 3.5 kiloVolts | 3 kiloVolts | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Trial # | Ring 1 | Ring 2 | Ring 1 | Ring 2 | Ring 1 | Ring 2 | Ring 1 | Ring 2 | Ring 1 | Ring 2 |
| 1 | 0.839 | 1.576 | 0.945 | 1.662 | 0.966 | 1.780 | 1.023 | 1.944 | 1.181 | 2.064 |
| 2 | 0.856 | 1.559 | 0.909 | 1.651 | 0.976 | 1.753 | 1.044 | 1.960 | 1.133 | 2.022 |
| 3 | 0.882 | 1.580 | 0.922 | 1.681 | 0.946 | 1.778 | 1.059 | 1.963 | 1.173 | 2.021 |
| 4 | 0.895 | 1.575 | 0.928 | 1.652 | 0.942 | 1.784 | 1.063 | 1.976 | 1.127 | 2.031 |
| 5 | 0.898 | 1.586 | 0.960 | 1.655 | 0.973 | 1.736 | 1.024 | 1.941 | 1.156 | 2.051 |
| 6 | 0.859 | 1.595 | 0.913 | 1.659 | 0.984 | 1.748 | 1.021 | 1.938 | 1.128 | 2.061 |
| 7 | 0.863 | 1.572 | 0.891 | 1.664 | 0.988 | 1.759 | 1.022 | 1.956 | 1.161 | 2.054 |
| 8 | 0.886 | 1.582 | 0.915 | 1.662 | 0.942 | 1.781 | 1.039 | 1.941 | 1.131 | 2.064 |
| 9 | 0.884 | 1.580 | 0.922 | 1.661 | 0.981 | 1.755 | 1.052 | 1.936 | 1.121 | 2.072 |
| 10 | 0.871 | 1.599 | 0.914 | 1.662 | 0.939 | 1.766 | 1.026 | 1.947 | 1.101 | 2.003 |
Table 1: Original Data - The data in this table corresponds to the 10 individual data points taken for each data set using the analog calipers accurate to .001 inches during the first two days of the experiment. With this data, I first converted it to metric values than used its mean and standard error to calculate the lattice spacings listed in Table 3.
| 4.75 kiloVolts | 4.25 kiloVolts | 3.75 kiloVolts | 3.25 kiloVolts | 2.75 kiloVolts | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Trial # | Ring 1 | Ring 2 | Ring 1 | Ring 2 | Ring 1 | Ring 2 | Ring 1 | Ring 2 | Ring 1 | Ring 2 |
| 1 | 22.59 | 41.16 | 24.01 | 44.06 | 25.31 | 48.01 | 25.43 | 51.23 | 28.08 | 53.79 |
| 2 | 21.71 | 41.11 | 23.19 | 43.49 | 24.82 | 46.99 | 25.98 | 50.11 | 28.78 | 54.98 |
| 3 | 22.24 | 41.03 | 23.64 | 43.76 | 24.76 | 47.45 | 26.13 | 51.43 | 28.36 | 54.43 |
| 4 | 21.95 | 40.79 | 22.86 | 44.30 | 24.90 | 47.70 | 25.87 | 50.78 | 30.23 | 53.93 |
| 5 | 22.57 | 40.87 | 23.79 | 43.48 | 24.91 | 47.62 | 25.66 | 51.06 | 29.56 | 54.07 |
Table 2: Additional Data - The data in this table corresponds to the 5 data points that I took using the Carrera Precision digital calipers for each of the 5 values of the voltage so as to form an interpolant to my original data set, allowing me to have a more continuous range of data rather than 5 data sets with spacing of .5kV between each set.
Final Results
| 2.75kV | 3.0kV | 3.25kV | 3.5kV | 3.75kV | 4.0kV | 4.25kV | 4.5kV | 4.75kV | 5kV | |
|---|---|---|---|---|---|---|---|---|---|---|
| d (inner) | .2052(65) | .2103(68) | .2048(65) | .2099(69) | .2076(66) | .2104(65) | .2059(64) | .2182(68) | .2017(64) | .2106(75) |
| d (outer) | .112(10) | .112(10) | .112(10) | .110(10) | .111(10) | .1076(99) | .1066(97) | .1074(99) | .109(10) | .109(10) |
Table 3: The results that I generated for each of the 10 used data sets along with the errors. The final results had a relatively symmetric appearance in the error, especially for the measurements off the large ring where the error calculated to be approximately .01nm for all the data values. The calculated error in the measurements from the small ring are all relatively close, with the exception being that from the 2.75kV data set, which was the hardest to see, due to the very faint ring.
From the individual values corresponding to each of the voltages, I was able to calculate the overall mean and standard error of my value for the lattice spacing. By taking the weighted average of the spacing values and the total standard error, I came out with final values of:
[math]\displaystyle{ d_{inner}^{exp}=.208(7)\mathrm{nm} }[/math] and [math]\displaystyle{ d_{outer}^{exp}=.110(10)\mathrm{nm} }[/math]
The known spacing in the graphite crystal lattice is [math]\displaystyle{ d_{inner}^{act}=.213\mathrm{nm} }[/math] and [math]\displaystyle{ d_{outer}^{act}=.123\mathrm{nm} }[/math]
With the known and my experimental spacings, I was able to generate a confidence interval about my experimental result, demonstrating that for measurements based off the inner ring, the known value lies well within a 68% confidence interval of my answer. For the measurements based off the large ring, the accepted value is 1.3 times the standard error of the mean from my experimental value, meaning that a confidence interval stretching to the accepted value would be an 80% confidence interval, as from the Z-chart of normal probabilities[9] the interval corresponding to 1.3 standard deviations on either side of the mean has a likelihood of 80.64%.
Errors
In my calculation of the error, I used the error in the manufacture of the diffraction tube, and the error generated from having multiple data measurements. By propagating those errors, I came up with my final error in calculation and the weighted average of the variance gave me the overall error in the lattice spacing.
The data that I took gives a belief in a possible systematic error in the measurement of the large ring. Measuring the rings, I took my measurement from the inside of the left side of the ring to the outside of the right, because the accurate measurement is a center-to-center measurement, but in doing the inside-outside, I was able to approximate the center-to-center measurement. An error that could have resulted from this method is a bias towards either the inside or outside of the ring, resulting in my measurements being either larger or smaller than the proper diameter, which would result in a different calculation of the spacing. The second primary source of error is that the lower voltages had the appearance of the rings become more and more faint, and as my ability to distinguish the rings, the possibility of error increased in my measurements. The only other source of error that could have influenced my results is a problem with the power supply that resulted in the voltage output not corresponding to the value shown on the supply.
In order to make this experiment better, I would use a larger apparatus with a more powerful power supply, so as to make measurements on a larger surface, which would allow for the measurements to be more accurate as the rings would be larger and easier to distinguish from the ambient light produced bu electrons scattering at angles not corresponding to the first-order maxima.
Conclusion
Because my results from the small ring had the accepted value within the 68% confidence interval centered on my experimental value it is reasonable to accept my value as calculated form the smaller ring. As my calculation based off the large ring is outside the 68% confidence interval, accepting that value as reasonable is less valid, because from a normal distribution, a value that is 1.3 standard deviations away from the mean will only be found 20% of the time, so assuming that there is no systematic error, a value as extreme as my calculated value would only be found 1/10 times.
Because of this, I feel that there was some form of systematic error related to the exterior ring, be it from the width of the ring and measurement, or from factors such as the power supply or the lack of intensity in the light, that altered my measurement of the exterior ring.
Ignoring sources of error, the appearance of scattered rings proves the validity of the de Broglie hypothesis, that proof being one of the objectives of this experiment. The existence of the rings proves the hypothesis, because as an electron travels, it will only diffract when interacting with a diffraction grating if it has a finite wavelength on the order of the size of the spacing in the grating, meaning that, by seeing a diffraction pattern, the de Broglie hypothesis is verified because it proves the existence of the particle wavelength.
Acknowledgements
I would like to thank my lab professor, Dr. Steven Koch, and the lab assistant, Aram Gragossian, for all their help fixing the different apparatus if I was getting incorrect data. I would also like to thank the UNM Physics Department for allowing us to use the lab and providing the apparatus so that we can operate.
References
SJK 15:30, 15 December 2008 (EST)

Page numbers are very important for journal article citations. Most journals allow you to retrieve an article by volume and page number. Sorry I didn't notice this when you asked me the question over the weekend.
[1] L. de Broglie. "The Wave Nature of the Electron." Nobel Lecture. 1929. http://nobelprize.org/nobel_prizes/physics/laureates/1929/broglie-lecture.pdf
[2] W.H. Bragg & W.L. Bragg, "The Reflection of X-rays by Crystals," Nature, vol. 91, no. 2280, (1913).
[3] W.H. Bragg, "X-Rays and Crystals," Nature, vol. 90, no. 2243, (1912).
[4] C. Davisson & H. Kunsman, "The Scattering of Electrons by Nickel," The Physical Review, vol. 19, no. 3, (1922).
[5] C. Davisson & L. Germer, "Diffraction of Electrons by a Crystal of Nickel," The Physical Review, vol. 30, no. 6, (1927).
[6] M. A. Van Hove, "Surface Crystallography with Low-Energy Electron Diffraction," Proceedings: Mathematical and Physical Sciences, vol. 442, no. 1914, (1993).
[7] S. Koch. Physics 307L. OpenWetWare. (2007). http://www.openwetware.org/wiki/Image:Physics_307L_e_diffraction2.JPG
[8] M. Gold. Physics 307L: Junior Laboratory. UNM Dept. of Physics and Astronomy. (2006).
[9] Taylor, John R. An Introduction to Error Analysis - 2nd Ed. University Science Books. Sausalito, CA. 1997.