PET Imaging by Kylee Klinkowski
What is PET Imaging? [1]
Positron emission tomography (PET) imaging is a nuclear medicine imaging technique that uses compounds labeled with positron-emitting radioisotopes to image and observe metabolic processes.
Motivation [2]
PET imaging was introduced as a way to take images of the brain in a non-invasive way (without opening the skull or taking samples of brain tissue). The idea that blood flow is closely related to brain function began as early as the nineteenth century with Italian physicist Angelo Musso. Despite this, interest in the relationship between brain function and blood flow did not become popular until the twentieth century. It was not until the end of World War II that Seymour Kety and his colleagues developed the first quantitative method for measuring whole-brain blood flow metabolism in humans. However, because their measurements were confined to the whole brain, they were not suitable for brain mapping. Instead, their introduction of an in vivo tissue autoradiographic measurement of regional blood flow in animals provided the first account of quantitative regional changes in blood flow in the brain directly related to brain function. Many years later, variations of this technique became important for the measurement of blood flow in humans. PET provided a means of quantifying the spatial distribution of radiotracers in tissue without the need for more painful imaging practices such as pneumoencephalography, a technique used by neurologists and neurosurgeons to localize mass lesions such as tumor and blood clots in and around the brain. The technique consisted of removing spinal fluid and replacing it with air, which would rise in the remaining spinal fluid (like air bubbles in water) into the brain ventricles and over its surface. The patient was then manipulated in space while strapped into a specially mounted chair that facilitated the movement of air bubbles. X-rays were taken in various positions enabling clinicians to map the contours of the brain. Great skill was required to interpret these X-rays, and the patient experienced severe headaches.
History [3]
1878: Italian physiologist Angelo Musso discovers the concept that blood flow relates to brain function by measuring an increase in brain pulsations from the right prefrontal cortex during an arithmetic task performed by a subject with a skull defect.
1911: Hungarian physicist George Hevesy tags a hostel’s food with radioactive lead, and is able to determine that the leftover meat had been reprocessed into two different dinner meals. He later wins a Nobel Peace prize in chemistry in 1934 for producing an artificial isotope.
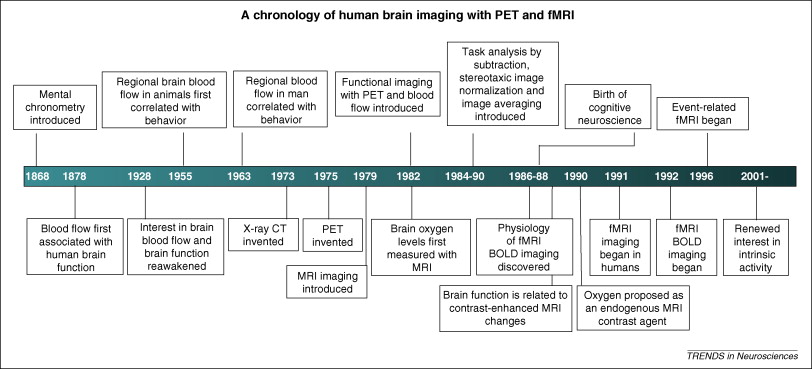
1948: Seymour Kety, physiologist at the University of Pennsylvania, measures cerebral blood flow through exchange of inert gas tracers in local blood flow. The group uses X-ray images of radiolabeled tissue to visualize the distribution and rate of local blood flow.
1950: Benedict Cassen, a physicist at the University of California, develops the first radioisotope imaging system, the scintiscanner. In the same year, the first large-scale use of a human positron imaging device is developed by physicist Gordon Brownwell and neurosurgeon William Sweet at the Massachusetts General Hospital. The machine is used detect brain tumors with sodium iodide.
1956: The photoscanner is developed by David Kuhl at the University of Pennsylvania.
1961: Danish scientist Niels Lassen and Swedish scientist David Ingvar use 133Xe to localize sensory, motor, and mental functions of the brain. Color-coded patterns track brain blood flow as related to its function. This became the standard visual representation of cerebral PET images.
1973: PC-I, one of the first PET imaging devices, is unveiled at the Meeting on Tomographic Imaging in Nuclear Medicine.
1976: American scientists Louis Sokoloff, Alfred Wolf, and Joanna Fowler develop 2-[18F]fluoro-2-deoxy-D-glucose (FDG). This compound is the most widely used radiotracer today.
1985: Ring-shaped and cylindrical shaped detectors are implemented in PET scanners, providing better resolution and sensitivity.
Modern era: PET scanners improve diagnosis, management, and treatment of neurological disorders including Parkinson’s disease, dementia, and epilepsy. In addition, PET scanners are widely used in cardiology and clinical oncology.
Physics Behind PET Imaging
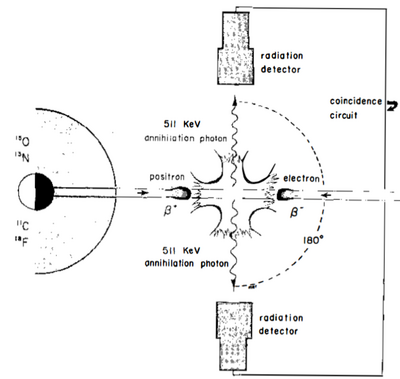
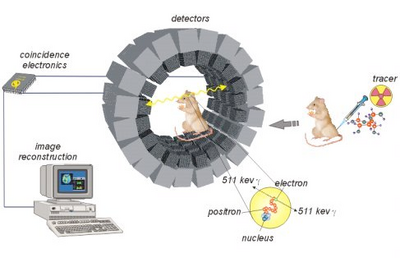
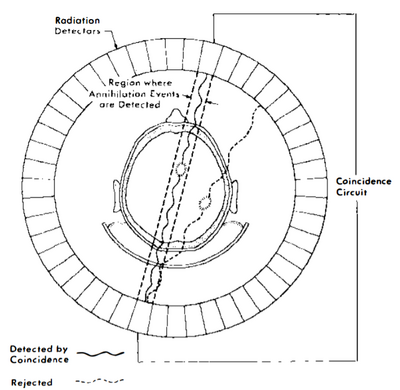
PET imaging uses the principle of positron emission to create images [1]. Positron emitters have nuclei rich in protons and stabilize themselves by getting rid of the excess protons either by positron emission (common in lower atomic weight nuclei) or electron capture (common in higher atomic weight nuclei). When a positron is emitted, it travels a short distance before it is slowed down by the electron clouds of surrounding materials in the patient’s tissue. As the positron travels through the patient’s tissue, it continuously loses energy until it combines with an electron to annihilate completely, emitting a pair of 511 keV photons at opposite 180 degree angles (Figure 3). The PET detectors register the photons as an event if they are detected within a narrow 3 to 15 nanosecond time frame. Detecting both the photons within a certain time window is the basis of coincidence detection and is termed electronic collimation. The interaction is assumed to have taken place somewhere on the straight line drawn between the two PET detectors deemed the coincidence line. Millions of recorded coincidence events, forming a large number of intersecting coincidence lines, give information about the quantity and location of positrons in the body. Modern PET scanner detectors use small crystals that couple to large photodetectors to create images [1]. The subject is surrounded by a cylindrical ring of detectors with a diameter of 80 to 100 cm, and an axial extend of 10 to 20 cm [4]. The detectors are shielded from radiation on their outer surface. When a photon interacts with the crystal, electrons are moved from the valence band to the conduction band. These electrons return to the valence band at impurities in the crystal, simultaneously emitting light. These crystals are coupled to photo-multiplier tubes which are then used to create the PET image. The two most used crystals are lutetium oxyorthosilicate (LSO) and lutetium yttrium oxyorthosilicate (LYSO) due to their very high light output, high sensitivity, and very fast decay times [4].
PET Tracers and Radiochemistry [5]
The four most common positron-emitting radioisotopes used for PET are:
- Fluorine-18 (F-18)
- Carbon-11 (C-11)
- Nitrogen-13 (N-13)
- Oxygen-15 (O-15)
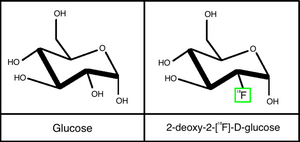
These radioisotopes are used because they can be directly substituted into biomolecules. When these short-lived PET-radioisotopes are used to make PET radiotracers, they must be synthesized and imaged within a time frame that is compatible with the half-life of the isotope. For example, for C-11, this amounts to about 10 minutes for isotope production, 40 minutes for radiotracer synthesis, and about 90 minutes for PET imaging (2.5 hours total). The synthesis of almost any PET radiotracer cannot take more than two half-lives, or else there will be a significant loss of specific activity that is critical for PET studies. Today, the most commonly-used radiotracer is FDG (Figure 4) [5].
Procedure
PET imaging begins with the use of a cyclotron, a device that smashes high-speed particles and ordinary atoms together, transforming them into radioactive atoms [1]. A biological molecule synthesizer is then used to attach the radioactive atom to a biological molecule that is tailored to a specific function of the human body. The biological molecule may be a sugar, a protein, or a hormone, depending on the part of the body being imaged. The biological molecule in complex with the radioactive atom is called a tracer. The tracer must then pass quality control. It is purified and checked to see if it will function properly. The tracer is then injected into the blood stream of the patient. The tracer will accumulate in its corresponding part of the body. The radioactive atom on the tracer will begin to lose its radioactivity by emitting a positron. When this positron collides with an electron at the right speed, they are annihilated and their energy is released as two gamma rays that travel out of the body in opposite directions. The PET scanner detects the two gamma rays on opposite sides of the ring, and the tracer's location in the body can be determined. Thousands of these events occur every second and are used to detect activity in three-dimensions [1]. The overall PET imaging process can be seen in Figure 5. Because the doses of radiotracer administered are small, the patient experiences relatively low radiation exposure. Such low dosage exposure to radiation has not been known to cause any long-term side effects. Additionally, allergic reactions to the injected radiotracer are extremely rare and usually mild, making PET imaging a very safe process [15].
Applications of PET Imaging
Cancer [5]
- Detecting cancer
- Determining whether cancer has spread in the body
- Assessing the effectiveness of a treatment plan
Nervous System Disorders [3]
- Evaluating abnormalities such as memory disorders, seizures, and other central nervous system disorders such as epilepsy, Parkinson's disease, and Alzheimer's disease
Cardiology [15]
- Determining blood flow to the heart
- Determining the effects of a heart attack
- Identifying areas of the heart that would benefit from angioplasty or coronary artery bypass surgery
Other [15]
- Mapping normal brain and heart function
Applications in Clinical Oncology
FDG-PET Imaging
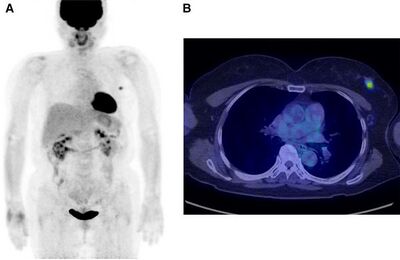
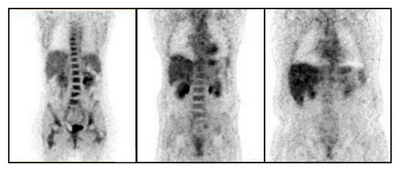
Fludeoxyglucose (18F) is a glucose analog with the positron-emitting radionuclide fluorine-18 substituted for the normal hydroxyl group at the C2 position in the glucose molecule (FDG) [5]. This is the most common radiotracer for PET imaging, especially in clinical oncology. PET imaging is significant in clinical oncology because it can show all the detectable disease in the body with high accuracy. Anaerobic glycolysis is an early indicator of malignant transformation of cells. Using PET imaging with FDG enables analysis of the glucose metabolism throughout the entire body in a single examination. Similarly, PET imaging can be used to assess the therapeutic response of cancer treatments [5]. Uptake of FDG by the tumor can usually be detected with a PET scanner about 45 to 90 minutes after injection [7]. FDG-PET imaging is helpful for primary tumor detection and diagnosis, assessing metastases, and monitoring response to therapy (Figure 6). In pre-operative testing, high uptake of FDG by tumors is observed, especially in ductal carcinomas, but FDG itself is not tumor specific and cannot detect malignant from benign cells. For this reason, false positives can occur when activated inflammatory cells exist [7].
Hormone Receptor PET Imaging (FES-PET Imaging) [7]
Up to 70 percent of breast cancer patients have tumors that test positive for hormone receptors. The majority of them are positive for estrogen receptor (ER), and more than 95 percent of tumors positive for progesterone receptor (PR) are also ER-positive. Anti-hormonal treatment strategies are based on hormone receptor expression, which is predictive of the efficacy of the treatment given. Therefore, monitoring hormone-receptor expression in tumor sites and metastatic sites can help physicians determine if anti-hormonal treatment should be used during different phases of treatment. The PET tracer 16α-,18F-fluoro-17β-estradiol (FES) was developed as a receptor ligand for ER. Several studies in breast cancer patients have shown a correlation between FES tumor uptake with ER tumor density. When comparing FES and FDG tumor uptakes, as well as ER status, in a sample of 43 patients, there showed an 88 percent overall agreement between FES uptake and ER status, while there was no correlation between FDG and ER status or between FES and FDG uptake. In 47 patients with ER-positive tumors, FES-PET was performed either prior to or shortly after the initiation of anti-hormonal therapy. This baseline response was then compared to the response after six months of treatment with anti-hormonal therapy. In patients with a low baseline FES uptake, no response was shown, while in patients with a high FES tumor uptake at baseline, 34 percent of the patients responded to anti-hormonal therapy. Therefore, although FES-PET cannot be used as a routine imaging technique, it can be useful for predicting response to anti-hormonal therapy.
HER2 PET Imaging [7]
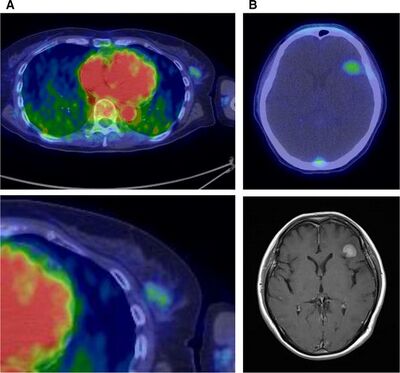
HER2, which is overexpressed in 25 to 30 percent of breast cancer patients, is involved in tumor cell survival, proliferation, maturation, metastasis, and angiogenesis. Trastuzumab, an antibody against HER2, is widely used in targeting HER2. HER2 expression is determined using immunohistochemistry (selectively imaging antigens by exploiting antibodies binding specifically to antigens in tissue), or fluorescence. However, these techniques cannot be used when the tumor cannot be easily accessed for a biopsy. Additionally, HER2 expression can vary during the course of the disease. In order to avoid these complications, PET imaging can be used to non-invasively determine HER2 expression. Currently, HER2 antibodies have been labeled with 124I, 86Y, 76Br, 89Zr, and 64Cu for PET imaging. Use of 64Cu-trastuzumab allowed for primary tumor lesions larger than 2 centimeters in diameter, and metastatic brain lesions larger than 1 centimeter in diameter to be visualized (Figure 7).
Applications in Nervous System Disorders
Alzheimer's Disease [8]
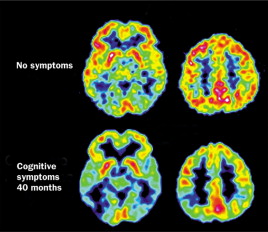
Studies in families with Alzheimer’s disorder caused by mutations in the APP (amyloid precursor protein) and presenilin genes have shown evidence of structural changes, in addition to impairment of brain glucose metabolism. These changes in glucose metabolism can often be detected before symptoms are experienced. For example, impairment in cerebral glucose metabolism has been observed in patients with a PS1 mutation 10 to 15 years before the onset of Alzheimer’s disease (Figure 8). Progressive impairment of cerebral glucose metabolism in people who have the Swedish mutation can be visualized with PET imaging, and people who have the APOE E4 (apolipoprotein) allele experience low rates of glucose in their brain decades before the possible onset of Alzheimer’s. In addition to FDG, the compounds fluorine-18-labelled FDDNP and carbon-11-labelled PIB have both been administered to patients with Alzheimer's disease.
Advantages and Disadvantages of PET Imaging
Advantages
- Non-invasive [1]
- Unlike most other imaging techniques that only image anatomical structures, PET imaging shows physiologic function [1]
- Penetrates deep into tissues [1]
- Live imaging [1]
- Low-risk for patient [9]
Disadvantages
- Relatively long imaging times: The injected radiotracer takes 45 to 60 minutes to be absorbed into tissues and organs, and the imaging itself takes an additional 30 to 60 minutes [9].
- Expensive: A full-body can cost up to $1,500 for a patient with Medicare, and $4,900 on average for a patient without health insurance [10].
- Heavy patient problem: The quality of the PET images decreases with increasing weight of the patient due to increased attenuation (Figure 10) [11].
- Low resolution: The low resolution of PET imaging is the result of positron range, noncollinearity of annihilation photons, and the detector size.
Positron range: Since the PET image is a map of the distribution of annihilation points within the object being scanned, the image resolution will be dependent upon the distance traveled by the positron within in the surrounding material before it combines with an electron and emits photons. This leads to a limit on the spatial resolution of the reconstructed PET image. The maximum range for common PET isotopes such as 18F, 11C, 15O, and 82Rb is between 2.6 and 16.5 mm. The effect on image spatial resolution is lower, falling between 0.2 and 2.6 mm [1].
Noncollinearity of annihilation photons: Some residual momentum of the positron just before annihilation will lead to the emission of two annihilation photons that are not exactly 180 degrees from each other (Figure 9). However, the reconstruction algorithm will always assume the two photons to be collinear, which results in a slightly misplaced coincidence line (usually less than 5 degrees). This will lead to a blurry image [1].
Detector size: The intrinsic detector spatial resolution in modern PET scanner detectors use small crystals that couple to large photodetectors. The detector spatial resolution is usually similar to the size of the crystals used. This can therefore determine spatial resolution of the reconstructed PET image [1].
References
[1] Basu, Sandip, Thomas C. Kwee, Suleman Surti, Esma A. Akin, Don Yoo, and Abass Alavi. 2011. "Fundamentals of PET and PET/CT Imaging." Annals of the New York Academy of Sciences 1228 (1): 1-18. doi:10.1111/j.1749-6632.2011.06077.x. http://onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.2011.06077.x/abstract.
[2] Raichle, Marcus E. 2009. "A Brief History of Human Brain Mapping." Trends in Neurosciences 32 (2): 118-126. doi:10.1016/j.tins.2008.11.001. http://www.sciencedirect.com/science/article/pii/S0166223608002658.
[3] Portnow, Leah H., David E. Vaillancourt, and Michael S. Okun. 2013. "The History of Cerebral PET Scanning: From Physiology to Cutting-Edge Technology." Neurology 80 (10): 952-956. doi:10.1212/WNL.0b013e318285c135. http://www.ncbi.nlm.nih.gov/pubmed/23460618.
[4] Ollinger, J. M. and J. A. Fessler. 1997. "Positron-Emission Tomography." IEEE Signal Processing Magazine 14 (1): 43-55. doi:10.1109/79.560323. http://ieeexplore.ieee.org/document/560323.
[5] Schlyer, D. J. 2004. "PET Tracers and Radiochemistry." Annals of the Academy of Medicine, Singapore 33 (2): 146. http://www.ncbi.nlm.nih.gov/pubmed/15098627.
[6] Yasuda, Seiei and Michiru Ide. 2005. "PET and Cancer Screening." Annals of Nuclear Medicine 19 (3): 167-177. doi:10.1007/BF02984601. http://www.ncbi.nlm.nih.gov/pubmed/15981668.
[7] Kurihara, Hiroaki, Chikako Shimizu, Yasuji Miyakita, Masayuki Yoshida, Akinobu Hamada, Yousuke Kanayama, Kan Yonemori, Jun Hashimoto, Hitomi Tani, Makoto Kodaira, Mayu Yunokawa, Harukaze Yamamoto, Yasuyoshi Watanabe, Yasuhiro Fujiwara, and Kenji Tamura. "Molecular imaging using PET for breast cancer." Breast Cancer 23.1 (2015): 24-32. Web. https://link.springer.com/article/10.1007/s12282-015-0613-z.
[8] Haq, Kaiser, David Firth, Debadrita Chakraborty, Ruksana Abdul-Majid, W. B. Gooderham, Bill Schwarz, Keith Jardim, et al. 2017. "PET imaging of amyloid in Alzheimer's disease." Wasafiri 32 (1): 77. doi:10.1080/02690055.2017.1252131. http://www.tandfonline.com/doi/abs/10.1080/02690055.2017.1252131.
[9] Cleveland Clinic. "Positron Emission Tomography (PET) Scan." Cleveland Clinic. N.p., n.d. Web. 08 Apr. 2017. https://my.clevelandclinic.org/health/articles/pet-scan.
[10] "How Much Does a PET Scan Cost? - CostHelper.com." CostHelper. N.p., n.d. Web. 08 Apr. 2017. http://health.costhelper.com/pet-scans.html
[11] Raichle, Marcus E. "PositronEmission Tomography." Annual Reviews Neuroscience 67th ser. 6.249 (1983): n. pag. Annual Reviews. Web. 8 Apr. 2017. http://www.annualreviews.org/doi/pdf/10.1146/annurev.ne.06.030183.001341.
[12] Josef Pacák, Zdeněk Točík, Miloslav Černý: "Synthesis of 2-Deoxy-2-fluoro-D-glucose"; Journal of the Chemical Society D: Chemical Communications, 1969, p. 77–77; doi:10.1039/C29690000077. http://pubs.rsc.org/en/Content/ArticleLanding/1969/C2/c29690000077#!divAbstract.
[13] West, Liz. "PET scan." Flickr. Yahoo!, 21 Aug. 2006. Web. 08 Apr. 2017. https://www.flickr.com/photos/calliope/221238656
[14] Muehllehner, Gerd and Joel S. Karp. 2006. "Positron Emission Tomography." Physics in Medicine and Biology 51 (13): R137. doi:10.1088/0031-9155/51/13/R08. http://iopscience.iop.org/0031-9155/51/13/R08.
[15] Radiological Society of North America (RSNA) and American College of Radiology (ACR). "PET/CT (Positron Emission Tomography - Computed Tomography )." RadiologyInfo.org. N.p., n.d. Web. 12 Apr. 2017. <https://www.radiologyinfo.org/en/info.cfm?pg=pet>.
[16] Professor Peyton. Bioimaging PowerPoint (lecture 15). April 10, 2014.