OhioMod2013:Design/NuclearPore
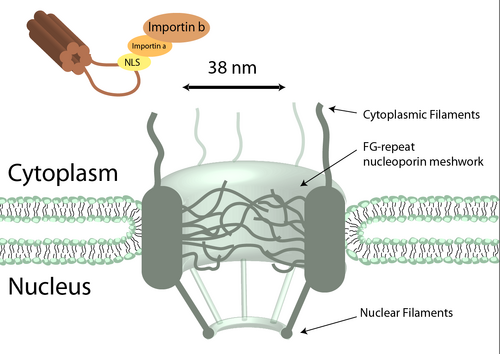
The Nuclear Pore
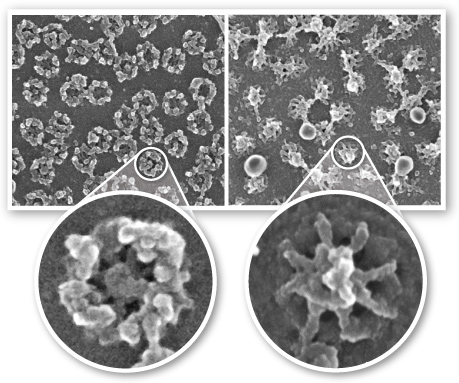
- Image courtesy of Martin Goldberg
- Left image: Cytosol side of the membrane. Composed of the cytoplasmic ring, of eight 50 nm long cytoplasmic filaments, with a gap of 70 nm. These filaments are assymetrical and connect the core structure to the cytoskeleton and protein synthesis assembly lines.
- Right image: Nucleus side of the membrane. Composed of nuclear filaments joining to create a basket-like ring with a gapa of 30-50 nm. This basket is involved with a dynamic molecular platform for nuclear activity such as transcriptional regulation .
- The central pore itself has a gap of 45-50 nm.
The nucleoporin is composed of around 50 different nucleoporins, which occur in multiples of eight.
The nuclear pore complex(NPC) is necessary for both the importing of nuclear proteins into the nucleus and the exporting of mRNA. We are soley focused on importation of genes into the nucleus, and so will be focusing on that single direction. The NPC is a selective barrier in order to prevent foreign objects from entering the nucleus that may have a malicious effect on the chromosomes. There are various proposals as to how the NPC translocates proteins across. Together, they suggest that the rapid translocation is observed can be contributed by sequential interactions with the nucleoporins that coat the central pore, the affinity transport proteins have for the nucleoporins causes their accumulation towards the NPC gate. We also know that the central channel and periphery forms a meshwork of unstructured nucleoporins with phenylalanine-glycine (FG) repeats which only allow passage of transport proteins. We know that without this meshwork the central channel is aroudn 38 nm, which matches our known maximum size for transported particles.
The NLS peptide is a tag that is recognized by karyopherins including Importin-α, which is then bound by Importin-β, the actual protein responsible for importing. The classical NLS has a stretch of basic, charged amino acids. The importin α/ importin β/ NLS-DNA complex binds to the phenylalanine of the FG repeat nucleoporin meshwork, which triggers translocation across. Other groups have shown that forming hyrodphobic regions to their nanoparticle will cross the nuclear pore complex. Another group had disrupted the hydrophobic regions of the FG-repeat nucleoporin central channel.
Zanta's group had shown a 10-1,000 fold increase in transfection by attaching a single NLS signal to a 3.3 kb luciferase gene [1]. Attempts to replicate by Tanimoto or by
Covalent Binding of NLS peptides to DNA
- NLS peptides can be conjugated directly to DNA, but must be careful not to affect the encoding regions of the DNA. Groups that try this approach often see a drop in transfection efficiency because the DNA itself can no longer work in the nucleus. Some groups avoid this inhibition effect by targeting only certain regions of the plasmid DNA by covalent triple-helix formation and photoactivation. We have an advantage in that if we keep the important domains of the plasmid within the interior of the structure, then they'll be relatively safer. If we can keep the number of NLS tags covalently tagged to the structure then this should be an effective design.
Non-covalent Binding of NLS peptides to DNA
- We can take advantage of the cationic peptides and simply
- Zanta MA, Belguise-Valladier P, and Behr JP. Gene delivery: a single nuclear localization signal peptide is sufficient to carry DNA to the cell nucleus. Proc Natl Acad Sci U S A. 1999 Jan 5;96(1):91-6. DOI:10.1073/pnas.96.1.91 |
- van der Aa MA, Mastrobattista E, Oosting RS, Hennink WE, Koning GA, and Crommelin DJ. The nuclear pore complex: the gateway to successful nonviral gene delivery. Pharm Res. 2006 Mar;23(3):447-59. DOI:10.1007/s11095-005-9445-4 |
- Strambio-De-Castillia C, Niepel M, and Rout MP. The nuclear pore complex: bridging nuclear transport and gene regulation. Nat Rev Mol Cell Biol. 2010 Jul;11(7):490-501. DOI:10.1038/nrm2928 |