Mouse Heart Harvest for DNA Arrays
Overview
List of reagents is not yet complete
Procedure
9/9/00 By Alex Zambon, Nathalie Cotte and Bruce Conklin
1. One week prior to sacrifice, mouse will be housed in a separate cage to minimize the effects of any inter-animal stresses on the days preceding the sacrifice.
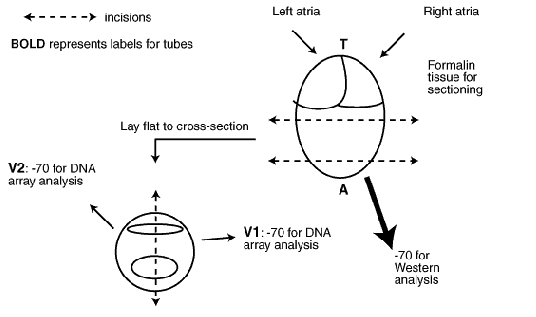
2. On the day of the sacrifice, pre-label all tubes (Mouse #, DOD [data of death], genotype and either: T, V1, V2, A [Top, Ventricle 1, Ventricle 2, Apex]- see picture in section 9) and prepare balance for quick weighing.
3. Mice are to be kept out of sight, smell and sound of the animal sacrifice area (the fume hood in 422 is a good place for this). Also, place enough six well plates containing PBS and 1M KCL in PBS on ice so that each heart can be individually bathed in 1 well of ice cold PBS and then transferred into 1 well of ice cold 1M KCl in PBS (three hearts/ 6 well plate).
4. Administer 0.8 cc of 2.5% Avertin as an IP injection and weigh the unconscious mouse.
5. Lay the mouse on its back, pin down hands and feet and use 70% ethanol to wipe down the entire chest, stomach and ribcage (done to prevent hair from contaminating samples). Make a U shaped incision cutting both skin and peritoneum that begins at navel area and extends up to front legs making sure you cut the rib cage on both sides. Fold up the flap over head and pin.
6. Open the chest cavity by making an incision in the diaphragm and harvest the heart by cutting the great vessels (the heart should be beating at this point) and drop immediately into ice cold PBS. Let the heart beat approx. 4 times to pump all the blood out of atria and ventricles.
7. Transfer the heart into ice cold 1M KCl in PBS for 2-3 seconds and start the timer. At this point the heart should stop beating. You should not take more than 4 minutes to have the heart pieces in liquid nitrogen from this point.
8. Remove the heart from KCl solution, trim away any excess vessels and/or fatty tissue and gently dry the heart on a towel (paper towel is fine) to remove excess liquid. Weigh the heart if you have enough time.
9. Section heart as shown below. At the end there should be 4 samples per heart that are labeled with the mouse number and either T, A, V1 or V2. (These data will be entered into a master spread sheet that is accessible on the intranet that anyone in the lab can access. These data will include birth date, treatment dates, age, sacrifice date, freezer location etc... so that in the future we know exactly where in the freezer all samples are and what the samples are from)
10. Place each piece of tissue (4 pieces/heart) in a prelabled eppendorf tube and flash freeze tissue in liquid nitrogen.
drawing of heart sectioning
Notes
No further notes are available at this time.
References
Relevant papers and books
No further references are available at this time
Contact
- Who has experience with this protocol?
or instead, discuss this protocol.