Modules:Best Disease
Mutations in the Best1 gene (encoding the protein of the same name) is known to cause a variety of retinal degenerative diseases in man (Hartzell, 2008; Marmorstein, 2007). The gene was originally called VMD2. Bestrophins are found throughout the animal kingdom, including prokaryotes and eukaryotes (Hagen, 2005; Hartzell, 2008; Milenkovic 2008). Was originally discovered by two different groups independently (Marquardt, 1998; Petrukhin, 1998).
Structure of Best1 Protein
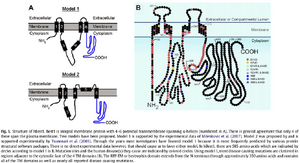
Structure of hBest1. Best1 is integral membrane protein with 4–6 potential transmembrane spanning a-helices (numbered in A). There is general agreement that only 4 of these span the plasma membrane. Two models have been proposed. Model 1 is supported by the experimental data of Milenkovic et al. (2007). Model 2 was proposed by and is supported experimentally by Tsunenari et al. (2003). Through the years most investigators have favored model 1 because it is most frequently predicted by various protein structural software packages. There is no direct experimental data however, that should cause us to favor either model. In hBest1, there are 585 amino acids which are indicated by circles according to model 1 in B. Mutation sites and the human disease(s) they cause are indicated by colored circles. Using model 1, most disease causing mutations are clustered in regions adjacent to the cytosolic face of the 4 TM domains (B). The RFP-TM or bestrophin domain extends from the N-terminus through approximately 350 amino acids and contains all of the TM domains as well as nearly all reported disease causing mutations.
Mechanism and Function of Best1
The mechanism and function of bestrophins is a controversial subject. Bestrophins have been thought to be Ca2+ activated Cl- channels, regulators of ion transport, or both (Hartzell, 2005, 2008; Marmorstein, 2007; Sun 2002). A lot of evidence supports their function as a Cl- channel. However, evidence of this in tissue is lacking. Based on animal data from animal models and tissue culture, it is suggested that bestrophins work as ion transport regulators rather than actual ion channels (Burgess, 2008; Hartzell, 2008; Marmorstein, 2007; Marmorstein, 2006; Rosenthal, 2006).
The Light peak (LP) is thought to represent a depolarization of the basolateral membrane in the RPE due to activation of Cl- conductance (Fujii, 1992; Gallemore, 1989, 1993). The LP is thought to be triggered by a yet discovered "LP substance" secreted by the photoreceptors and requiring activation of a second messenger cascade in the RPE (Gallemore, 1998). A diminished LP is the hallmark of BVMD.
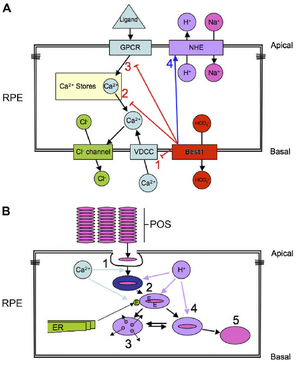
Hypothetical model of Best1 function in the RPE. We propose that Best1 functions to set the gain on GPCR signaling via Ca2þ and to maintain intracellular pH in the face of a changing gradient of CO2/HCO3- flowing across the RPE in response to changes in photoreceptor respiration. Combined data from several labs indicate that Best1 interacts physically and functionally with VDCCs (panel A, 1). The kinetic effects on VDCCs are to accelerate opening and closing times, resulting in a diminished entry of Ca2þ. Other effects on Ca2þ which we have identified in both Best1W93C knock-in, and Best1-/- mice as well as fhRPE cultures are the ability to modulate the release of Ca2þ stores in response to binding of a ligand to a GPCR (panel A, 2 and 3). It is not clear yet whether this results from a block of store release (panel A, 2) or by interference with GPCR signaling (panel A, 3). Best1 may be a channel protein. Although most investigators have examined its ability to conduct Cl-, it was recently found that Best1 was as or more efficient at conducting HCO3-. We have found that Best1 can alter pHi by promoting NHE activity (4). These effects appear to be dependent on HCO3-. It is likely that the changes in pH and Ca2þ due to Best1 activity are also interdependent. Should these functions be disrupted, we propose that the level of dysfunction dictates the disease phenotype (panel B). The common denominator in the bestrophinopathies is accumulation of lipofuscin, which could result from altered kinetics of phagocytosis uptake (panel B, 1), maturation (panel B, 2, 4), due to changes in acidification and/or delivery of lysosomal enzymes (E) (panel B, 2). Under normal circumstances (panel B, 3) the phagocytosed photoreceptor outer segment (POS) is properly degraded, however, a delay or acceleration in uptake, acidification, or degradation could promote the formation of A2E or other lipofuscin components from precursors already present in the POS. Liposufscin is non-degradable (panel B, 4) and eventually accumulates in lipofuscin granules (5). The rate and level of lipofuscin accumulation would play a major role in the severity of the disease.
RPE Phagocytosis - Phagocytosis is regulated by illumination and hypoosmotic cell swelling or application of the Cl- channel blocker tamoxifen decreases RPE phagocytosis (Irschick, 2006; Mannerstrom, 2001). Both Ca2+ and pH play important roles in RPE phagocytosis and fluid transport processes (Deguchi, 1994; Fenney-Burns, 1983; Hall, 1991; Liu, 2008; Peters, 2006). Lipofuscin accumulation is a by-product of phagocytosis (Feeney-Burns, 1983; Rakoczy, 1996), and pH and Ca2+ are important mediators of purinergic and adrenergic signaling pathways that appear to play a role in RPE fluid transport (Joseph & Miller, 1992; Quinn, 2001). Defects in RPE phagocytosis are known to cause retinal degeneration (LaVail, 1983), and impaired fluid transport can cause serious retinal detachment (Marmor, 1990; Negi & marmor, 1983).
Ca2+ is an important regulator of phagocytosis. Best1 alters intracellular Ca2+ and interacts physically and functionally with voltage-dependent calcium channels (VDCCs). VDCCs regulate the uptake of outer segments by RPE cells (Karl, 2008).
Pathophysiology
hBest1 was found only in the retina/RPE and the human derived RPE cell line ARPE-19 (Marquardt, 1998). Since then, it has been found in a number of different tissues using rtPCR (Marmostein, 2009). Using antibody labeling, Best1 has been found to be localized to the basolateral plasma membrane of the RPE in macaque, pig, human, canine, and mouse (Marmostein, 2000; Guziewicz, 2007; Mullins, 2005; Bakall, 2003). It is still not entirely clear whether Best1 is expressed outside the eye (Marmostein, 2009).
The clinical features in patients with mutations in Best1 seem to cluster into at least 4 different categories: 1) Classic Best Vitelliform Macular Dystrophy, 2) autosomal-dominant vitreoretinochoroidopathy, 3) autosomal recessive bestrophinopathy, and 4) adult-onset vitelliform macular degeneration. Glaucoma frequently accompanies bestrophinopathies, especially later in life. The major functional impairment is reduced visual acuity, resulting from the macular/retinal disease.
Best Vitelliform Macular Dystrophy (BVMD). Until the best1 gene was discovered, BVMD was identified by a fundus lesion, family history of the disease, and a subnormal electrooculogram (EOG) Arden ratio (iight peak (LP/dark trough) with an otherwise normal ERG (Cross, 1974; Marmor, 1979; Mohler, 1981). the gross appearance of the fundus among individuals with subnormal EOGs may be completely normal in some patients while in others the macula is severely damaged with gliosis and dense pigmentary scarring (Mohler, 1981). Even in patients aged 60-84, mean visual acuity has been reported as 20/40 and acuity is rarely worse than 20/400. During the disease progression, there is substantial lipofuscin accumulation (a common manifestation of AMD, Alzheimer's, and Parkinson's Disease. There are 5 major stages of BVMD as defined by Mohler & Fine:
-
Phase I – Pre-vitelliform disease, manifests with only granularity and window defects in the RPE
-
Phase II – characterized by classical vitelliform lesion located in the fovea and can be subdivided into stages IIa and IIb. Here, vision is often considerably better than would be expected based on the fundus.
-
Phase IIa - is the classical vitelliform cyst; a yellow well demarcated round lesion that is often compared to the yolk of an egg in appearance.
-
Phase IIb - the borders of the yellow color of the lesion become irregular.
-
Phase III - pseudohypopyon stage, presents as a partial reabsorption of the lesion yolk. An apparent fluid line can be resolved.
-
Phase IV - typically associated with severe vision loss
-
Phase IVa - atrophic and hypopigmented
-
Phase IVb - scarred with fibrous tissue in the macula, often with hyperpigmentation
-
Phase IVc - neovascularization is present around or within the gliotic scar
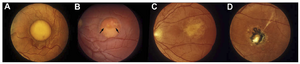
Clinical progression of the fundus in BVMD. Classical vitelliform or stage IIa lesion is shown in A. Stage IIb is characterized by a visible fluid line (arrows) within the lesion (B). Partial resorption of the fluid within the lesion gives the appearance of a scrambled egg and is characteristic of stage III or pseudohypopyon (C). Stage IV (D) is characterized by a gliotic scar accompanied by regions of hypo (stage IVa) or hyperpigmentation (stage IVb) and occasionally neovascularization (stage IVc).
Animal Models of Best Disease
A noninvasive way to measure LP responses in mice has been developed (Peachey, 2002; Wu 2004).
Mouse Model (BVMD-Associated W93C mutation). Shows similar changes in LP response to that in human Best Disease. Also, there is a significant build up of lipofuscin compared to age-matched litter mates. Another phenotypic response of BVMD. (Marmorstein, 2006; Wu, 2007).
Canine Model (cBest1, canine multifocal retinopathy). Found in multiple breeds and has vitelliform lesions, RPE hypertrophy, and enhanced RPE autofluorescence.
Best1 Promoter
The upstream region of Best1 (-154 to +38 bp) is sufficient to target expression of a transgene to the RPE.
References