Micromolding in Capillaries (MIMIC) - Wen-Wei Wong
Capillary action is a phenomenon where fluids travel through channels due to surface forces such as cohesive and adhesive forces (Figure 1).[1] Micromolding in capillaries, or MIMIC, is a technique that leverages this spontaneous fluid travel to deposit materials such as metals, inorganic crystals, polymers, ceramics, and/or suspensions of nanoparticles or organic matter in precise patterns on the microscale.[2],[3],[4] This method leverages capillary action of fluids to disperse a material through a mold of the pattern. After removing the mold, the deposited material is left behind. As the field of microfluidics grows and finds new applications in different industry, it is important to continue to develop different techniques to pattern materials.
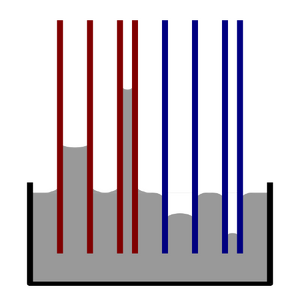
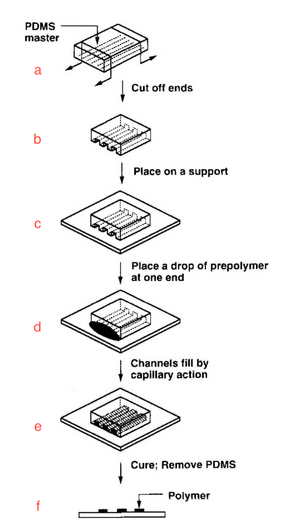
History of Micromolding in Capillaries
Micromolding in capillaries is a method that was developed in the Whitesides Lab at Harvard University in the Department of Chemistry in 1995. Kim et al., published a paper describing a new patterning technology they developed that does not suffer from disadvantages such as destroying the template structure or inconsistent depositions.[5] This method showed great promise in casting organic materials and more, and could be used in any field where microfluidic devices are applicable. Since this publication, MIMIC is a patterning technique that is used in many applications related to electronic, optical, biological, and mechanical devices.
Description of the Process
Figure 2 shows a diagram of the step-by-step process of MIMIC. A mold is created using a material that can be precisely shaped to create the deposition pattern. Polydimethylsiloxane is a popular choice given its physical properties, cost, and ease of use. A mold is fabricated via replica molding techniques, and a master can be fabricated using photolithography techniques. After creating the mold, the ends are cut off to expose the channels on the sides (Figure 2b). It is placed upon a new substrate (Figure 2c), and a small amount of the fluid to be cast is placed at one end of the channels (Figure 2d). Capillary action spontaneously fills the channels, then the fluid is cured via thermal or ultraviolet means (Figure 2e). Finally, the mold is removed to show the patterned material (Figure 2f). Depending on the cast material, the removal of the master should be easy and clean. The resolution of the edges, angles, and surface is limited by the precision of the mold.[2] Kim et al., have shown how poly(methyl methacrylate) could be cast into precise patterns.[5]
Functionality of MIMIC
Flow regime is crucial to the application of MIMIC. At the scales and speeds of capillary travel, the flow regime of the fluid as it fills the mold is laminar and the velocity is constant. At this flow regime, turbulent mixing does not occur; mixing only happens as diffusion across interfaces.[6]
The laminar flow regime can be taken advantage of to produce gradients on the microscale. In biology, materials and substances that can diffuse play an important biological patterning, morphogenesis, and angiogenesis.[7] By designing a network of capillaries, the two components can be introduced to each other at different concentration profiles to form gradients. These can be controlled with respect both space and time gradients.[7] Capillary action can occur which allows the two components to spontaneously travel throughout the system together.
MIMIC can be used to pattern nanoparticles, as in shown in work done by Watanabe et al. (Figure 3),[2] In this work, they fabricated phosphor films made out of rare-earth ion-doped nanoparticles on plastic sheets. By varying the concentration of nanoparticles in the solution, methanol, and the length/width of the channels, different compositions of the film can be obtained. By using capillary action, a constant concentration and even distribution of nanoparticles can be maintained.

An application MIMIC combining polymer patterning and biology was performed by Suh et al. They patterned poly(ethylene glycol) microstructures onto a substrate then coated it with P3 and P9 antibodies. After this, they introduced E. coli cells, some of which were healthy and others which were infected with M13 virus. By leveraging virus-antibody interactions, they were able to trap just the infected cells in the microstrucutres.[9]
Rate of Fluid Travel
Surface and interfacial energies are the main driving forces of MIMIC to be an effective method. Different liquids The rate of liquid traveling through the mold via capillary action is defined in the following equation:[8]
- [math]\displaystyle{ \mathrm{\frac{dl}{dt}} = \frac{\gamma r cos(\theta)}{4 \eta l} }[/math]
Where,
- [math]\displaystyle{ \mathrm{\frac{dl} {dt}} }[/math] is the rate of liquid traveling through the channels
- [math]\displaystyle{ \gamma }[/math] is interfacial free energy of the liquid
- r is the radius of the capillary
- [math]\displaystyle{ \theta }[/math] is the contact angle between the liquid and surface of the channel
- [math]\displaystyle{ \eta }[/math] is the viscosity of the liquid
- l is the distance the fluid has traveled through the capillary
An important consideration for using MIMIC is that the fluid is given sufficient time to penetrate through the entirety of the master, and that the fluid does not get stuck in the capillaries of the mold.
Comparison to Microtransfer Molding and Solvent Assisted Micromolding
Advancement in soft lithography have led to the development of several methods capable of patterning materials, such as polymers, on the microscale, including microtransfer molding (µTM) and solvent assisted micromolding (SAMIM). It is important to note these methods are distinct from MIMIC.
In µTM, a stamp is manually filled with the material to be patterned, such as a prepolymer solution. The system is then inverted onto a substrate, then the material is cured. Finally, the stamp is peeled away.
In SAMIM, a stamp is coated on the patterning side with a good solvent for the material to be patterned. Then, the coated stamp is placed on top of a substrate with a layer of the material to be patterned on top. The stamp with solvent dissolves the polymer and sinks into the layer of polymer to create the pattern until the desired thickness is reached, at which point the stamp is removed.
In both cases, the patterning material was inside the stamp, however, there was no capillary action taking place in either µTM or SAMIM. By prefilling the stamp with material or leveraging solubility, capillary action is not the driving force which allows the lithography technique to work.
An example that leverages the capabilities of MIMIC such as the mechanism of capillary fluid flow and the laminar mixing properties of the fluid flow is highlighted by the work of Kenis et al., A PDMS mold featuring a zigzag channel and a Y-junction at one end was fabricated. The inside walls were coated with a thin layer of gold. Then, the Y-junction introduced a stream of water and a stream of gold etchant. Due to the capillary action and laminar flow of the two streams, they both traveled together through the mold with no mixing, allowing the gold etchant to remove the gold layer from one side while the water preserved the gold layer on the other side. This selective treatment to the device would be much more difficult or impossible using methods like µTM or SAMIM.[4]
To learn more about µTM or SAMIM, refer to their specific Wiki pages.
References
1. Peiris, A.; Stein, B. Capillary Action. Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Physical_Properties_of_Matter/States_of_Matter/Properties_of_Liquids/Capillary_Action
2. Kim, E.; Xia, Y.; Whitesides, G. M. Micromolding in Capillaries: Applications in Materials Science. Journal of the American Chemical Society 1996, 118 (24), 5722–5731. https://doi.org/10.1021/ja960151v.
3. Watanabe, S.; Takeo Asanuma; Hiroshi Hyodo; Soga, K.; Matsumoto, M. Micromolding in Capillaries for Calcination-Free Fabrication of Flexible Inorganic Phosphor Films Consisting of Rare-Earth-Ion-Doped Nanoparticles. Langmuir 2013, 29 (35), 11185–11191. https://doi.org/10.1021/la401810x.
4. Kenis, P. J. Microfabrication inside Capillaries Using Multiphase Laminar Flow Patterning. Science 1999, 285 (5424), 83–85. https://doi.org/10.1126/science.285.5424.83.
5. Kim, E.; Xia, Y.; Whitesides, G. M. Polymer Microstructures Formed by Moulding in Capillaries. Nature 1995, 376 (6541), 581–584. https://doi.org/10.1038/376581a0.
6. Weibel, D. B.; DiLuzio, W. R.; Whitesides, G. M. Microfabrication Meets Microbiology. Nature Reviews Microbiology 2007, 5 (3), 209–218. https://doi.org/10.1038/nrmicro1616.
7. Dertinger, S. K. W.; Chiu, D. T.; Jeon, N. L.; Whitesides, G. M. Generation of Gradients Having Complex Shapes Using Microfluidic Networks. Analytical Chemistry 2001, 73 (6), 1240–1246. https://doi.org/10.1021/ac001132d.
8. Radha, B.; Kulkarni, G. Micromolding—a Soft Lithography Technique. Micromanufacturing Processes 2012, 329–347. https://doi.org/10.1201/b13020-25.
9. Suh, K. Y.; Khademhosseini, A.; Yoo, P. J.; Langer, R. Patterning and Separating Infected Bacteria Using Host–Parasite and Virus–Antibody Interactions. Biomedical Microdevices 2004, 6 (3), 223–229. https://doi.org/10.1023/b:bmmd.0000042052.47444.9a.
