Mechanobiology and Microscale devices - Robert Sterian
Introduction
Mechanobiology is the field of science involving the responsiveness and signaling of tissues and cells by altering physical and mechanical properties [1]. By integrating mechanobiology, changes of cellular mechanics can lead to treatments of diseases, organ development and homeostasis [2]. In the field of mechanobiology, microfluidics is needed for research in order to fully comprehend the relationship between biological responses from cellular compounds and engineering from a mechanical device. Many experiments involving in vivo and in vitro methods have many flaws. For in vitro experiments, many involve large scale procedures that will experiment with many different cell types and tissues inside many different types of cultures. will be involved where important biological factors and functions will not be fully emulated. Where as for in vivo experiments, there is difficulty in observing animal studies on a daily basis. By studying mechanobiology of microfluidics, errors for both methodologies can be mitigated with enhanced imaging in a microscopic level and developing simulations of environments for microorganisms undergoing different scenarios [3].
Types of Mechanical Stimuli
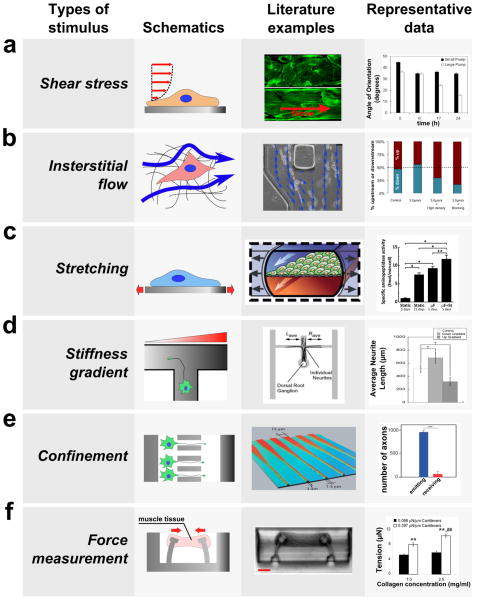
In order to develop more accurate experiments using mechanobiology, microbiology will be used to answer mechanobiology questions by developing different microenvironments for cells and tissues to undergo different situations using mechanical devices. The use of simulating these environments are to gain more knowledge on the interaction of cells and develop more realistic models for in vivo models. The development of realistic models will lead to more efficient methodologies on how to develop experiments with the end goal of achieving organ equilibrium and/or treatment of diseases. The capability of a 3D view of cellular compound interactions between a compound and a surface. For many examples, PDMS is used as a material for many devices. These interactions from different mechanical stimuli will also lead to more accurate observations rather than viewing cell at a two dimensional plane[3].
Shear Stress
Endothelial shear stress is the friction of blood flow through an endothelial surface[4]. If a blood vessel were to have a high endothelial shear stress, then there is very high friction leading to decrease of blood flow. Where as a blood vessel with a low endothelial shear stress has a low friction leading to an increase of blood flow. With the use of mechanobiology, scientists are able to study the effects of fluid flow by using a constricted pathway to emulate a blood vessel with shear stress. Some responses as a result of this observation are cell elongation, increased cell contractibility, and decreased permeability of blood vessels[3].
Interstitial Flow
Interstitial flow is the motion of fluids through an extracellular matrix such as tissues. This type of flow is encouraged in order to help transport large proteins and induce physiological responses from interstitial cells[5]. By implementing a pressure fluid gradient on a hydrogel, motion and alignment of cells can be mimicked as interstitial flow. According to figure 1 (right), the blue arrows represent a culture of cancer cells migrating at an upstream motion [3].
Stretching and Stiffness Gradient
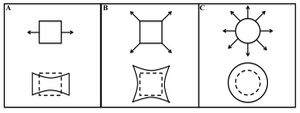
Stretching is a form of mechanical stimulation that engineers the physical lining of the membrane and modifies it. This technique is primarily used for promoting the process of myogenesis, the formation of muscular tissue. Stretching can also play a major role in the growth and functionality of muscle [6]. There are multiple methods for how a membrane can be stretched depending on using one more more axels (Figure 2). Studies have shown that muscle tissue under Embryonic Chick Cardiomyocyte and Neonatal Rat Cardiomyocyte culture have been studied for approximately a week in order observe the effects while under mechanical stimulation. The results presented evidence of the muscle tissue becoming more enlarged, improved functionality of contracting, and increased tensile strength [7][8]. Similar to stretching, stiffness gradient has an influence in myogenesis [3]. In addition, with an in vitro model, stiffness has the ability to control a range of cellular functionality such as motility, morphology, and response signalling [9].
References
[1]“Mechanobiology.” Wikipedia, Wikimedia Foundation, 7 Feb. 2020, en.wikipedia.org/wiki/Mechanobiology.
[2]Kim, Anna A, et al. “Microfluidics for Mechanobiology of Model Organisms.” Methods in Cell Biology, U.S. National Library of Medicine, 2018, www.ncbi.nlm.nih.gov/pmc/articles/PMC6418080/.
[3]Polacheck, William J, et al. “Microfluidic Platforms for Mechanobiology.” National Center for Biotechnology Information, U.S. National Library of Medicine, 21 June 2013, www.ncbi.nlm.nih.gov/pmc/articles/PMC3714214/.
[4]Chatzizisis, Yiannis S., et al. “Role of Endothelial Shear Stress in the Natural History of Coronary Atherosclerosis and Vascular Remodeling: Molecular, Cellular, and Vascular Behavior.” Journal of the American College of Cardiology, Elsevier, 8 June 2007, www.sciencedirect.com/science/article/pii/S073510970701220X.
[5]Yao, et al. “Interstitial Fluid Flow: The Mechanical Environment of Cells and Foundation of Meridians.” Hindawi, Hindawi, 17 Dec. 2012, www.hindawi.com/journals/ecam/2012/853516/.
[6]Rangarajan, S., Madden, L., & Bursac, N. (2013). Use of Flow, Electrical, and Mechanical Stimulation to Promote Engineering of Striated Muscles. Annals of Biomedical Engineering, 42(7), 1391–1405. doi: 10.1007/s10439-013-0966-4
[7]Fink, C., Ergün, S., Kralisch, D., Remmers, U., Weil, J., & Eschenhagen, T. (2000). Chronic stretch of engineered heart tissue induces hypertrophy and functional improvement. The FASEB Journal, 14(5), 669–679. doi: 10.1096/fasebj.14.5.669
[8]Boublik, J., Park, H., Radisic, M., Tognana, E., Chen, F., Pei, M., … Freed, L. E. (2005). Mechanical Properties and Remodeling of Hybrid Cardiac Constructs Made from Heart Cells, Fibrin, and Biodegradable, Elastomeric Knitted Fabric. Tissue Engineering, 11(7-8), 1122–1132. doi: 10.1089/ten.2005.11.1122
[9]Isenberg, B. C., Dimilla, P. A., Walker, M., Kim, S., & Wong, J. Y. (2009). Vascular Smooth Muscle Cell Durotaxis Depends on Substrate Stiffness Gradient Strength. Biophysical Journal, 97(5), 1313–1322. doi: 10.1016/j.bpj.2009.06.021
