M465:PCR and gel electrophoresis
Finding the Wild Wolbachia
We will next attempt to amplify only Wolbachia by using a primer set specific to the bacetrium :a forward primer, wsp_F1: GTCCAATARSTGATGARGAAAC , and a reverse primer, wsp_R1: CYGCACCAAYAGYRCTRTAAA. These primers are short sequences of single stranded DNA that are complementary in sequence to areas of the wsp gene in the Wolbachia genome. The wsp gene encodes for a protein on the surface of Wolbachia. After 30 cycles of polymerase chain reaction in a thermal cycler, the result will be a pcr product containing hundreds, if not thousands, of the wsp gene, but only if your insect was infected with the bacterium.
To review how the polymerase chain reaction works and how it exponentially amplifies specific sequences of DNA, go to the following web site:
PCR animation
http://www.dnalc.org/resources/animations/pcr.html
All PCR reactions require a thermal cycler to elevate and reduce the reaction temperature quickly and keep it at a specific temperature for a prescribed amount of time. There is a basic pattern to these temp. cycles, but there are differences, so you must be sure to program the cycler with the correct time and temperature for your specific amplification. Traditionally, pcr used Taq polymerase, a heat stable DNA polymerase originally found in an extremophilic bacterium, Thermus aquaticus, that lives and reproduces in boiling hot springs. We are not using Taq for our pcr but a different polymerase, Finnzyme's Phusion High-Fidelity Polymerase, a proprietary reagent that uses a novel heat-stable Pyrococcus-like enzyme. Phusion DNA Polymerase generates long templates with a greater accuracy and speed than with Taq. The error rate of Phusion DNA Polymerase in Phusion HF Buffer is determined to be 4.4 x 10-7, which is approximately 50-fold lower than that of Thermus aquaticus DNA polymerase, and 6-fold lower than that of Pyrococcus furiosus, another proof-reading DNA polymerase.
Therefore, our pcr product DNA will have far fewer "mistakes" in the sequences that are replicated from template DNA. Our polymerase will also work much faster so our ~20 cycles will require less time than conventional Taq based pcr.
Protocol for PCR
1. Obtain one 0.2ml pcr tube from your instructor - you will need one for each of your DNA extractions (for each of your insect "types"). All of the ingredients listed below in the table, except the template DNA, have been added together previously and kept on ice for you in these tubes.
2. Label the tube with a fine tipped Sharpie on the side - make sure you keep track of the code name in your lab notebook. Do not use tape!
3. To each tube, you will add 4 μL of the DNA you extracted. Since your pcr tube already has 10μL master mix, 4μL DNAase free water, and 1μL of each of 2 primers, the total reaction volume for everyone will be 20μL.
It is very important to pipet these tiny volumes accurately. Use the P10 or P20 pipettes. Look at the tip after you draw up your measured volume to make sure you have liquid there.
4. Dispense the template DNA into the liquid directly, watching to make sure that the liquid has left the pipette tip.
5. Bring your tube to your instructor; they will show you where the thermal cycler is located in JH 022. Keep track of where in the PCR machine your tubes have been placed (the exact quadrant, row and column). Your instructor will start the reaction when everyone's tubes are loaded.
Component TABLE
| Component | amt. in a 20 μl reaction |
Final Conc. |
|---|---|---|
| Purified DNAase free Water |
4 μL already in tube. Want to achieve total of 20 μl reaction vol. Add from 0 - 3μl |
_ |
| 2x Phusion Master Mix | 10 μl | 1x |
| wspF primer | 1 | 0.5 μMolar |
| wspR primer | 1 | 0.5 μMolar |
| template DNA | 4 μl | optimum is 100ng of DNA/reaction |
The cycling program is shown below.
94°C for 2 min
30 cycles at:
94°C for 30 s
56°C for 45 s
72°C for 1 min 30 s
1 cycle at:
72°C for 10 min
4°C hold
While the wsp genes from all of the species in your DNA are being amplified in the thermal cycler, you will have about an hour to work on dilutions worksheets and making a good figure..
After the PCR reactions are complete, you will run your PCR products on a gel to determine if you have amplified Wolbachia.
Agarose Gel Electrophoresis of Your wsp PCR product
To see if you successfully amplified the wsp gene - and therefore identified Wolbachia in your samples - you will "run a gel" on your pcr products. To run a gel means that we will perform an electrophoretic separation of the DNA fragments in your pcr product, using a small fraction of the volume of your pcr product applied to a 1% agarose gel stained with Sybr Safe DNA stain. Your instructor will help you to photograph the gel so you can evaluate whether you found the Wild Wolbachia. You should see a single band of ~400 bp if Wolbachia was present.
Quick Q: Can you explain how we know the size of our amplified gene fragment?
Importantly, your instructor has already made an agarose gel for you (thank Lindsay, will ya?). Below are the instructions for making one, in case you are curious.
Protocol for making a 1% agarose gel
In order to run out your PCR products you will first pour a 1% agarose gel. The recipe for the gel is 1.0% agarose (w/v) in 1x TBE buffer (10x=890mM Tris, 890mM Boric Acid, 20mM EDTA) with SybrSafe™ stain. Your instructor has added a SybrSafe stain to your molten agarose in order to allow us to visualize the DNA.
Protocol for making the agarose gel (already done for you!):
1. Using a graduated cylinder, fill a glass, 250 mL flask with 100 mL of 1x TBE buffer.
2. Using the top loading balance, measure out 1 gram of Agarose
3. Pour the agarose into the flask containing the TBE buffer and stir to mix. The agarose won't go into solution here but needs to be heated
4. Place the flask inside the microwave and heat for 1 minute, maximum power. At the end of the minute, swirl the solution to mix and place back in the microwave.
5. Heat for 1 minute again, this time watching closely to make sure it does not boil. At the end, swirl to mix.
6. Add the SyberSafe dye (it is at 10,000x concentration), and pour your gel into the mould. It will take ~ 25 minutes to set.
DNA is uniformly negatively charged and will,therefore, move toward the positive electrode. The separation is determined by the size or mass of the molecule or fragments of DNA.
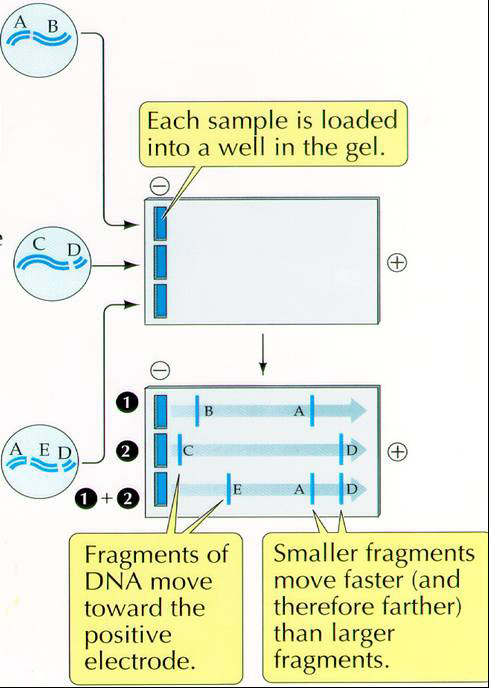
Below is the protocol for running your PCR products on this agarose gel. This you WILL complete during this lab.
Procedure for Agarose Gel Electrophoresis of PCR products
1. Load 5 microliters of your PCR product on your gel by placing the 5 microliters of your pcr product as a spot on a small piece of parafilm and adding 5 microliters of loading dye (0.25% XC, 30% glycerol, 0.1mg/ml RNAase).
2. Mix the loading dye by pipetting up and down on the spot, on the parafilm, before loading all 10 microliters into a lane of the 1% agarose gel.
3. Record which lane is loaded with which product. Be sure to leave the first lane and the last lane empty for the 100 bp ladder. Do not forget to load your positive and negative controls as well!
4. The gel will be run at 120V for approximately 30 minutes.
Clean Up
At the end of this lab remember to:
1) Put your DNA in the -20C freezer
2) Wipe down your lab bench with the 70% ethanol spray bottles and paper towel
3) Dump your ice in the sink
4) Wash your hands!
