M465:Gram Staining and Slants
Bacterial Morphology and the Gram Stain
The morphological characteristics of bacteria, including size, shape, and arrangement, can be seen by staining a bacterial smear so that individual bacterial cells are distinguished. Generally, bacteria range in size from approximately 0.2 µm to 3.0 µm. The basic shapes are spherical (coccus), rod-shaped (bacillus), curved (vibrio), or helical (spirillum, spirochete). However, there are some species of bacteria that show considerable within species variation (termed pleomorphism). For example, both Mycoplasma (bacteria lacking the rigid cell wall of most bacteria) and Arthrobacter (a type of soil bacteria) show forms ranging from coccoid (round) to rodlike to filamentous. Some pathogenic species such as Mycobacterium tuberculosis and Corynebacterium diptheriae are also pleomorphic.
Bacteria are often individual but some species take on a group arrangement based on the way cell division and subsequent separation of the daughter cells occur. Most of the Gram-negative bacilli (rods) are found singly (Escherichia coli) or, sometimes, characteristically in pairs (Klebsiella pneumoniae). The cocci, Streptococcus pneumoniae and Neisseria are called diplococci because they tend to pair. Bacilli found in chains include some Bacillus species. The coccus genera that most often form chains are named Streptococcus for this defining arrangement. A few cocci make regular packets of four or eight (Micrococcus) and some are seen in irregular clumps that resemble bunches of grapes (Staphylococcus). Short rods that form parallel lines, called palisades, include the species that causes diptheria, Corynebacterium diptheriae. Chinese character formation describes a sharply angled bacterial arrangement. Because of their waxy cell walls, Mycobacterium species are difficult to emulsify and tend to stick together in clumps. The pathogen in this group, Mycobacterium tuberculosis, may form long cords of cells.
Keep in mind that individual cells may show deviations from these standard forms. For example, cocci of Neisseria show flattened sides, making them bean-shaped. The rods of Corynebacterium and Mycobacterium often appear club-shaped, with swollen ends or knobs. Both groups may show irregular staining. The diplococci of Streptococcus pneumoniae often appear slightly elongated and lancet-shaped (with one flattened end and one tapered end).
Background on Using Stains in Bacteriology
The first of the dyes most useful to bacteriologists was a reddish violet dye, mauvein, synthesized in England by William. H. Perkin, and patented by him in 1856. This synthetic dye and others were immediately appreciated by histologists, but were not applied to bacterial cells until Carl Weigert (a cousin of Paul Ehrlich) used methyl violet to stain cocci in preparations of diseased tissue in 1875. Subsequently, the use of various synthetic dyes for bacteriological preparations developed rapidly when they were promoted through the publications of Robert Koch and Paul Ehrlich.
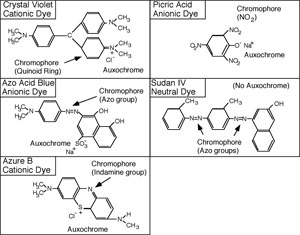
The synthetic dyes are classified as acid dyes, or basic dyes, depending on whether the molecule is a cation or an anion. The introduction of the terms acidic and basic was unfortunate because it would be more revealing to refer to them as cationic or anionic dyes. A look at the structural formula reveals the nature of the dye.
There are many stains that can reveal the morphology of the cell, and some simple stains, such as methylene blue, are quite good for viewing bacteria. The Gram stain is especially useful because it not only reveals bacterial morphology, but also is a differential stain. A differential stain differentiates organisms. (A differential stain shows a visible difference between different groups of organisms based on some characteristic they do not share, even though the procedure to stain the different looking organisms is the same). The Gram stain relies on cell wall differences between groups of bacteria.
The Gram staining procedure as it is done today, involves: a) primary staining of all cells with crystal violet, b) precipitating the primary stain dye within the cells with iodine (a mordant), c) removing the dye-iodine precipitate from some cells (the Gram-negative) with a decolorizer such as 95% ethanol, acetone, or n-propyl alcohol, and d) counter-staining of the decolorized cells with safranin. Organisms that retain the crystal violet primary dye are termed Gram-positive, while those which lose the primary stain and show the red safranin counter-stain are termed Gram-negative. This differentiation is not absolute, because it is based on the differences in the rate at which the primary dye is lost from the cells. If you over decolorize for too long or with too harsh a decolorizer, Gram-positive organisms will appear Gram-negative. Truly Gram-positive cells, such as Bacillus subtilis or Staphylococcus aureus, will not retain the primary dye if the iodine step is omitted.
Lab Activities
Activity 1: Prepare a Bacterial Smear Slide
0. Obtain your bacterial isolates (a single colony from your restreaked plates).
1. Label a clean, glass slide with a graphite pencil on the far left of the slide with the number of the culture you will me smearing (The decolorizer in the Gram stain can remove your labels if you use pen or wax pencil.)
2. In the hoods, sterilize the loop in the incinerator. Then place one very small loopful of deionized water on the slide. (Using the sterile loop, you can use the deionized water in the 15 mL falcon in your white tray).
3. Sterilize the loop in the incinerator, allowing it to cool for a few seconds and then touch the cooled loop to the patch of your isolate, picking up a TINY bit of growth from the patch. An invisible amount of growth obtained from just touching the cooled loop to the colony is fine. Too much picked up, such that the culture is a visible plug on the loop, is not fine.
4. Place the loop with the bacterial growth into the drop of water on the slide. Use a circular motion to make a smooth suspension of the bacteria in the water. Stop when there is a circle of emulsified bacteria about the size of a nickle on the slide.
5. Resterilize the loop the loop.
6. Repeat steps 1-4 with the other cultures you'd like to stain (your 5 isolates at minimum).
7. Allow the slide to air dry completely! Be sure all the water on the slide has evaporated before proceeding to heat fixation!!! This drying step is crucially important. If you are impatient, you will "explode" the cells in the next step. Protip: use the slide warmers to allow the slides to dry more quickly
8. Heat fix (to kill and attach organisms to the slide) by quickly passing the slide (smear side up) through a flame 3 times. Your instructors will provide butane lamps to use on your benches for this purpose. Use a clothes pin or slide holder and avoid contact with hot glass.
An example of a multiple smear labeled slide:

Activity 2: Preparing a Gram Stain
The Gram stain is a standard staining technique useful for the identification of culturable bacterial organisms and you will perform it now.
Use the slides prepared in Activity 1 and follow the Gram Stain Protocol found below. Your instructors will demonstrate how to perform a gram stain at the instructors bench.
To Gram stain the bacterial smear slide that you prepared in Activity 1, you must be careful to apply the staining reagents liberally so all the smears are evenly and completely covered and you must be sure to expose each smear to each reagent for the same amount of time.
1. Place your smear on the two pipettes over the sink at your bench. It is important that the slide be level during staining so use paper towels under the tray to get it leveled. If you do, it is much easier to be sure that your smears will be covered evenly with each reagent.
2. Dispense just enough Crystal Violet solution (0.5% crystal violet, 12% ethanol, 0.1% phenol) to completely cover each smear and stain for 1 minute. (Crystal violet is the primary stain.)
3. Rinse the slide by lifting it at a 45 degree angle (using gloves or a clothes pin or slide holder) and use a squirt bottle to direct a very gentle stream of water slightly above the top smear. Rinse until the waste water coming off at the bottom is relatively clear; drain off excess water by touching the edge of the slide to a paper towel.
4. Dispense just enough Gram's Iodine (mordant)to completely cover each smear. Let stand for 1 minute. Rinse thoroughly with a gentle stream of water as in Step 1.
5. Lift the slide at a 45 degree angle and drip Decolorizing Reagent (80% isopropyl alcohol, 20% acetone) down the length of the slide making sure it comes in contact with all three smears. This step is tricky as it is easy to over- or under-decolorize. Do this for 10 seconds and IMMEDIATELY rinse, as in step 3, with a gentle stream of water.
6. Place the slide flat and dispense just enough Counterstain (0.6% safranin in 20% ethanol) to cover each smear. Let stand for 2 minutes; rinse with water as in step 3.
7. Blot dry using paper towels. Insert your slide between two paper towels and pat it dry.
8. Clean up your area. Store the positive/negative controls in your drawer; you'll need them for the next lab.
9. Observe your stained microbes microscopically following the correct procedure for using the the oil immersion objective on your compound brightfield microscope. The directions for using the microscope are found below.
Activity 3: View your stained bacteria.
Today you will use only the 10x and 100x objectives. Remember also to read and follow the directions for care of this precision instrument (particularly on how to avoid getting immersion oil on any objective other than the 100x oil immersion lens). Be aware that there would be no field of microbiology if there weren't good, functioning microscopes to view this unseen world. The compound light microscope can magnify to about 1000 times the actual size of the specimen and can resolve details as fine as 0.2µm.
CARE OF THE MICROSCOPES
A compound microscope is available for each student's use. Remember at all times that your microscope is a precision optical instrument and must be handled carefully. When removing the microscope from its cabinet, do not jar or drop it, always carry it upright with one hand below the base and the other hand on the arm of the microscope. Place the microscope at least 6 inches from the edge of the bench. When returning the microscope to storage, check that:
1. the microscope light is turned off before the microscope is unplugged;
2. all lenses have been cleaned with lens paper, especially the oil immersion lens;
3. the lowest objective lens is near the stage and the stage itself is lowered; and
4. the microscope is covered (if there is a cover available).
PARTS OF THE MICROSCOPES
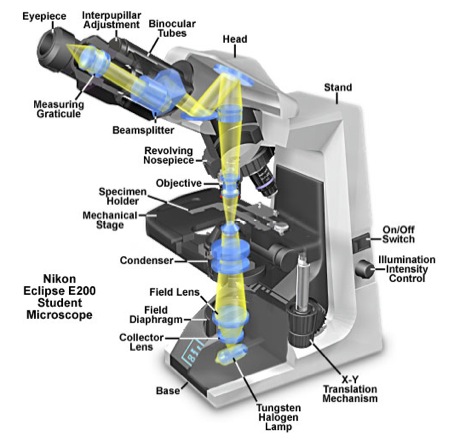
Figure 1 contains a diagram of a compound microscope and may help you locate some of the parts referred to in the following explanation. The compound microscope derives its name from the two sets of lenses it uses to magnify objects. These lenses are the objective lens, which can be found on the rotating nosepiece near the stage of the microscope, and the ocular lens, which is in the eyepiece. Your microscopes are equipped with several objective lenses, ranging from low to high magnification, including one oil immersion lens. The microscope magnifies by shining light from the light source through the iris diaphragm that limits the diameter of the light beam. The condenser lens focuses the light through the specimen that is on the stage. The stage is movable in order to view different parts of the specimen. The image we see is formed under the ocular lens by the objective lens and is a mirror image of the actual specimen.
REGULATION OF ILLUMINATION
1. The illumination intensity can be adjusted with a slider or with a knob.
2. Another way of adjusting illumination is by changing the position of the condenser lens. The condenser lens adjustment knob is located below the specimen stage and on the left side. It allows the user to move the condenser lens assembly up or down. As you move the condenser lens up, closer to the specimen, it concentrates (condenses) more light on your specimen. You will need to make this adjustment as you go up in magnification, so that you will have sufficient illumination.
3. The condenser aperture diaphragm is located below the specimen stage on the condenser lens assembly. It is an adjustable opening, which allows you to make fine adjustments in illumination. The lever, which adjusts the size of the aperture, faces the user. By sliding the lever to the left or right, you may adjust the illumination to the correct level for your specimen. Changing the size of this aperture also affects the amount of contrast in the image. Thus, adjusting the condenser aperture involves finding the brightness level, which gives you the best combination of illumination and contrast. This is the method used most often in adjusting illumination in the light microscope.
HOW TO LOCATE SPECIMENS USING A COMPOUND LIGHT MICROSCOPE
1. Place specimen slide on microscope stage and secure with clamping arm.
2. Use the 10x objective lens to find the specimen and focus it using first the coarse and then the fine adjustment knobs.
3. We will NOT use the 40x objective (high dry) because of the risk of getting immersion oil on that lens. Since bacteria are so small you should go directly to oil after focusing in low power by rotating the 10x objective away from the slide, putting a drop of oil on the center of the field of view, and then rotating the nosepiece so that the 100X objective lens comes in contact with the oil. Focus using the fine adjustment knob ONLY.
4. Never use the course adjustment when focusing a specimen with the oil objective because doing so could result in damage to the 100X objective lens or to the slide. All traces of oil must be removed from the lens before putting away the microscope. Only lens paper should be used to remove oil from the 100X objective.
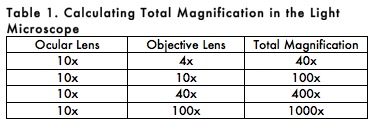
CALCULATION OF TOTAL MAGNIFICATION
Total magnification of the specimen is determined by multiplying the magnifying power of the ocular and objective lenses. For example, a 10X ocular and a 100X objective together give a total magnification of 1,000X (Table 1). This means that the specimen appears 1000 times larger when viewed with a microscope than its actual size.
MEASUREMENT OF SIZE
Cell size can be measured using an ocular micrometer. A micrometer is sometimes found in one of the ocular lenses of each microscope. It looks like a small ruler with both large and small units. The large units are numbered 1, 2, 3, etc. The small units are subdivisions of the large units and are not numbered. There are 10 small units per large unit. The small units represent different lengths depending on the objective lens in use. You measure cellular structures in small units only, and then convert to metric units (µm = micrometers) using the conversion values below.
Therefore, if you are observing a cell with the 100X objective, and this cell spans 2.5 small units on the ocular micrometer scale, then the size of the cell is calculated by multiplying 2.5 small units x 1µm/small unit = 2.5µm.
Figure 1. A Cutaway Diagram showing the beam path of the Nikon Eclipse E200 Compound Light Microscope. [SOURCE: http://micro.magnet.fsu.edu/primer/anatomy/nikone200cutaway.html]