M465:Fly genetic screen part 2
The fruit fly is an excellent model for molecular genetics. Not only have thousands of genes been characterized in the fly, with regards to their function, we also have resources online that integrate all of the various studies that have been completed in each of these loci. For example, let us say you are interested in immune gene function in Drosophila. You can click on this link here: http://flybase.org/ and search for immune. The result will be 517 hits with that query phrase. Click on “genes” and at the top of the list is the gene imd, or immune deficiency. If we click on the imd symbol it takes us to the gene page, where we can find out lots of information about it, as in “what chromosome is it on?” and “when is it expressed during fly development” or “is it an essential gene?”.
Today we will be asking “Does gene dosage play a role in infection?” by investigating flies from a collection called the Deficiency Kit. These flies each contain a genomic ablation covering a large segment in the chromosome. We will see if these flies have the same number of associated microbes (CFUs) and if they harbor Wolbachia (checked by PCR). As you have already performed these protocols earlier in the semester, this should be a straightforward week!
Agarose Gel Electrophoresis of Your wsp PCR product
To see if you successfully amplified the wsp gene - and therefore identified Wolbachia in your samples - you will "run a gel" on your pcr products. To run a gel means that we will perform an electrophoretic separation of the DNA fragments in your pcr product, using a small fraction of the volume of your pcr product applied to a 1% agarose gel stained with Sybr Safe DNA stain. Your instructor will help you to photograph the gel so you can evaluate whether you found the Wild Wolbachia. You should see a single band of ~400 bp if Wolbachia was present.
Importantly, this time we will be making two agarose gels for the entire class. We need two volunteers to perform the protocol below:
Protocol for making a 1% agarose gel
In order to run out your PCR products you will first pour a 1% agarose gel. The recipe for the gel is 1.0% agarose (w/v) in 1x TBE buffer (10x=890mM Tris, 890mM Boric Acid, 20mM EDTA) with SybrSafe™ stain. Your instructor will then add a SybrSafe stain to your molten agarose in order to allow us to visualize the DNA.
1. Using a graduated cylinder, fill a glass, 250 mL flask with 100 mL of 1x TBE buffer.
2. Using the top loading balance, measure out 1 gram of Agarose
3. Pour the agarose into the flask containing the TBE buffer and stir to mix. The agarose won't go into solution here but needs to be heated
4. Place the flask inside the microwave and heat for 1 minute, maximum power. At the end of the minute, swirl the solution to mix and place back in the microwave.
5. Heat for 1 minute again, this time watching closely to make sure it does not boil. At the end, swirl to mix.
6. Add the SyberSafe dye (it is at 10,000x concentration), and pour your gel into the mould. It will take ~ 25 minutes to set.
DNA is uniformly negatively charged and will,therefore, move toward the positive electrode. The separation is determined by the size or mass of the molecule or fragments of DNA.
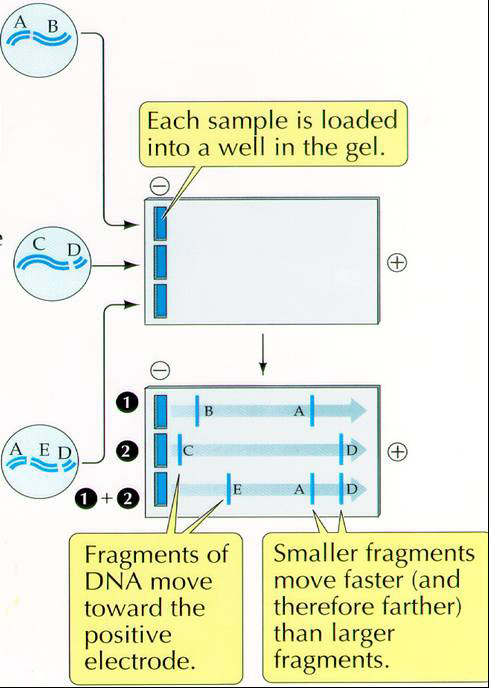
While your DNA is setting, you will count your CFUs.
Procedure for Agarose Gel Electrophoresis of PCR products
1. Load 5 microliters of your PCR product on your gel by placing the 5 microliters of your pcr product as a spot on a small piece of parafilm and adding 5 microliters of loading dye (0.25% XC, 30% glycerol, 0.1mg/ml RNAase).
2. Mix the loading dye by pipetting up and down on the spot, on the parafilm, before loading all 10 microliters into a lane of the 1% agarose gel.
3. Record which lane is loaded with which product. Be sure to leave the first lane and the last lane empty for the 100 bp ladder. Do not forget to load your positive and negative controls as well!
4. The gel will be run at 120V for approximately 30 minutes.
Clean Up
At the end of this lab remember to:
1) Put your DNA in the -20C freezer
2) Wipe down your lab bench with the 70% ethanol spray bottles and paper towel
3) Dump your ice in the sink
4) Wash your hands!
