Lund:Research
The Lund Lab: The Lymphocyte in Context
In recent years it has become apparent that the tumor microenvironment presents a significant obstacle to the efficacy of clinical cancer therapy. Recently we demonstrated that the lymphatic endothelium plays an active and dynamic role in regulating anti-tumor immunity. Our group explores the hypothesis that the tumor microenvironment directly manipulates local lymphocytes, which in turn, further remodel the microenvironment. This regulatory feedback maintains immune suppression in the context of a growing tumor and prevents newly activated lymphocytes from functioning locally. Ultimately we can leverage these immune suppressive mechanisms into strategies that improve the local immune response against the tumor and drive clinical therapy. To achieve this goal our group focuses on 3 key areas:
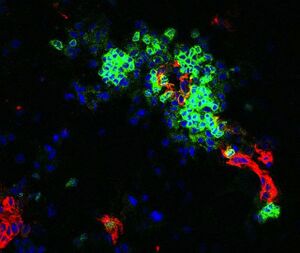
1. Tumor-associated lymphatic vessels and effector cell infiltration, function and retention. Based on our previous work and direct observation of CD3ε+ T cell association with the lymphatic endothelium in the tumor microenvironment, we are interested in exploring the crosstalk between activated T cells and the tumor-associated lymphatic endothelium. How does this cell-cell interaction influence effector T cell function in the context of the tumor microenvironment? How is this interaction regulated and how does it contribute to effector cell exit from the tumor?
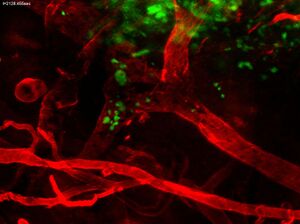
2. Matrix remodeling and immune imprinting on the tumor microenvironment. Cell-matrix adhesions are bidirectional interactions that both influence cell behavior and remodel the local matrix. Upregulation of adhesion molecules coincide with T cell activation and are required for entry into peripheral tissue as well as local retention. We are interested in how lymphocyte-mediated matrix remodeling in turn educates and propagates this immune response locally. How do lymphocyte subsets differentially interact with the aberrant extracellular matrix of the tumor? How do these physical changes influence neighboring and newly infiltrating immune cells, with respect to their subsequent trafficking, function and retention?
3. Microenvironmental targeting for combination immunotherapy. Understanding how the biophysical and stromal context of tumor tissue suppresses immune function provides a unique therapeutic target for the manipulation of tissue specific, anti-tumor immune responses. Ultimately, if the cell-cell and cell-matrix interactions elucidated above are unique points of tumor-mediated immune regulation then we can target these mechanisms to drive T cell function and improve existing immunotherapies.