Luckau Protocols:Agarose Gel
From OpenWetWare
Jump to navigationJump to search
| Agarose Gel Protocol |
Purpose
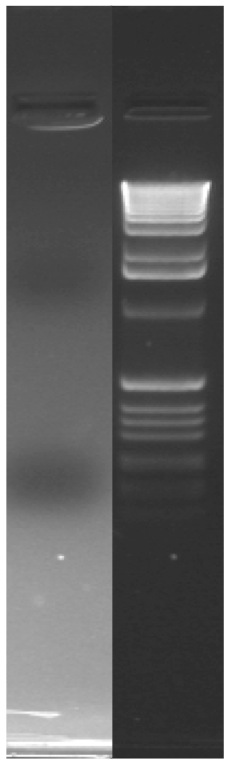
Agarose gels are used to verify the presence and estimate the size of DNA (both genomic and amplified). When loaded into a gel and subjected to an electric current, DNA fragments migrate to the positive terminal. Small DNA fragments migrate more rapidly than large ones. By running a commercially available ladder of fragments of known size, we can estimate the DNA fragment size in our sample of interest.
To visualize the DNA after electrophoresis, a dye called GelRed is added during the agarose-making process. The dye binds to the DNA and flouresces when exposed to UV light.
Protocol
Use 1.5% for genomic DNA; use 2% for amplified DNA
| Gel Rig Size | 1x TAE (mL) | 1.5% agarose (g) | 2% agarose (g) | Gel Red (µL) | Sample volume (µL) | max voltage (V) |
|---|---|---|---|---|---|---|
| small (10cm x 10cm) | 50 | _ | 1 | 5 | 4-6 | 80 |
| medium (x x) | 130 | _ | 2.6 | 13 | 24-tooth comb: 6-10 | 120 |
| 36-tooth comb: 3-5 | ||||||
| large (20cm x 27cm) | 270 | _ | 5.4 | 27 | 24-tooth comb: 6-10 | 170 |
| 36-tooth comb: 3-5 |
Pour Agarose Gel
- According to the table above, mix agarose powder and 1x TAE in Erlenmeyer flask; swirl
- Microwave on 1/2 power until the mixture starts to boil
- Note: be careful not to let the mixture boil over, or you'll lose much of your volume and will have a mess to clean up
- Note: make sure all the agarose has dissolved into solution
- Add 10,000X GelRed when agarose mixture is warm; swirl
- Allow to cool - do the baby milk test (it should be at a warm temperature, but not too warm for a baby to touch)
- Pour into caster; set combs and cover with cardboard; allow to harden (10-20 minutes) (will appear cloudy)
- Remove combs
- Pre-poured gels may be stored covered in the fridge for up to 1 week
Load Agarose Gel
- On parafilm, mix sample with gel load dye in 1:6 ratio
- if loading 6µL into gel, mix 1µL gel load dye with 5µL sample in parafilm
- Transfer from parafilm into gel wells
- Load same volume (6µL) of ladder into wells on either side of your samples
Run Agarose Gel
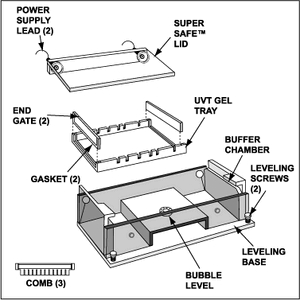
- Attach rig's safe lid and ensure power supply leads fit snugly
- Plug loose ends into power supply, as marked
- black gets plugged into the ground
- red gets plugged into the red
- Turn power supply to 'on'
- Adjust voltage according to table
- use the right-side dial to adjust the right-side meter (red numbers)
- Check for the curtain of small bubbles to ensure everything is working properly
- Cover with cardboard; takes about 30 min
Image Agarose Gel
- Assemble cafeteria tray:
- Gel (keep on caster)
- Kim Wipes
- USB stick
- gloves
- room key (in drawer)
- Gel Imager (UV transilluminator) is in room 215
- CAUTION! Ethidium bromide (EtBr) is a carcinogen, mutagen and teratogen, and it's used (instead of GelRed) by other users of this room.
- Keep one hand gloved to handle EtBr stuff, one hand ungloved to handle clean stuff
- The gel imager is EtBr-contaminated; the computer is CLEAN!
- Plug in USB drive; open the program "AlphaImager Mini"
- Place gel on glass top; center and zoom
- Close door; turn on UV light (bottom right)
- Adjust zoom, focus, aperture and exposure as needed
- Zoom: middle knob on camera (which is mounted on top of the box)
- Focus: bottom knob on camera
- Aperture: top knob on camera
- Exposure: arrow buttons in computer program
- Click 'Acquire' then 'Save'
- save as .jpg (not .tif) to the USB stick
- feel free to take multiple images of different settings or zooms
FYI
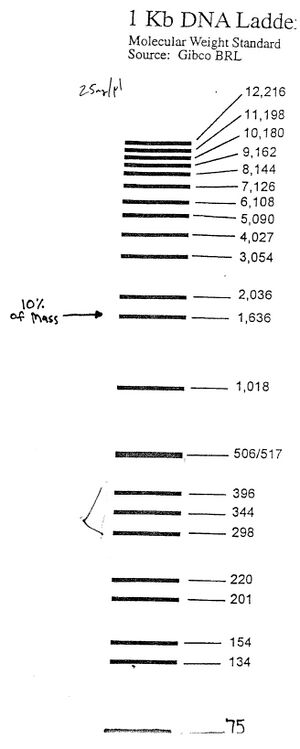
Migration of gel load dye & Ladder Map
- Bromophenol Blue (175bp) and Xylene Cyanol FF (1500bp)
To make 1L of 1x TAE from 50x TAE
20mL 50x TAE + 980 NanoPure H2O
Materials To Be Familiar With
- Gel Rig, caster tray, combs