Liquid-Liquid Extraction in Microchemical Channels - Jacob Brandner
Liquid-Liquid Extraction
Fluid separation is a necessary process in almost all chemical and biochemical processes. Liquid-liquid extraction is the process of separating two (or more) different fluids based on the ability of a liquid to be miscible in another. On the macroscopic scale, it requires that the two fluids are either immiscible or only partially miscible in each other (or by addition of a solvent that will allow these two conditions to hold true). This is due to the separation efficiency that can be achieved at that scale. To achieve an efficient separation, turbulent mixers are placed in vats to allow the liquid phases to partition themselves. Macroscopic scale extraction becomes more difficult when the solvents are not in high purity, the mixing forms large emulsion layers, or when miscible fluids cannot be separated with the use of a solvent. In addition the environment of a macroscale liquid-liquid extraction is not very tuneable. Conditions such as temperature, separation efficiency, and pressure are not nearly as fine tuned as in a microfluidic device.
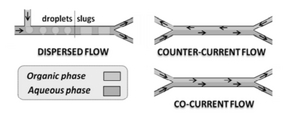
Operating at the microscale offers distinct advantages that cannot be offered on the macro scale, such as small diffusion lengths, high surface area to volume ratios and precise temperature control. With these characteristics, phase separation requires significantly less time and energy consumption. Mass transport in microchannels is mostly characterized by diffusion-based separation. This is due to the restriction of cocurrent flow in most applications. The only bypass for this operation would be to operate at low flow conditions as to not disrupt the liquid-liquid interface. Factors that affect a separation are diffusion distance, surface modifications (material that is present), the miscibility of the fluids, flow rate, the geometry of the channels, and the effect of outside forces (for example: gravitational force). In most instances, it is difficult to model the separation kinetics to have high throughput separation. This is why in most cases continuous separation is used over dispersive.
The most widely used liquid-liquid extraction techniques used in microchemical systems are dispersed (such as slug or emulsion formation) or continuous cocurrent extraction. These flow patterns can be characterized by the following dimensionless numbers:
[math]Ca = \frac{\mu \nu }{\sigma }[/math] [math]We = \frac{\rho \nu^2{} }{\sigma }[/math] [math]Bo = \frac{\rho g d}{\sigma }[/math] [math]E = \frac{\Delta \rho gd}{\sigma }[/math] [math]Re = \frac{We}{Ca} = \frac{\rho \nu d}{\mu }[/math]
Where the Weber number describes the ratio between the inertial force and the surface tension force (hence why it is used to describe interfaces of at least two different fluids). The Reynolds Number which is in terms of the Weber number and the Capillary Number. The Bond number measures the forces of gravity as fluids will differ in density inducing high dimensional flows.
Dispersive Flow
One phase is embedded in a continuous phase. Dispersive flow is characterized by distinct emulsions that take the form of microdroplets formed by shear forces. In some cases, it is advantageous to generate stable emulsions in a microfluidic environment. However, it is often useful to be able to separate two immiscible components and emulsions formed between the liquid interfaces often make it difficult for separation. On the macro scale, it is a simple procedure of shaking and removing the separated layers. On the micro scale, these will appear as emulsions within a larger bulk fluid. As they are moving continuously, the same methods cannot be employed to separate these emulsions as it is possible for them to be thermodynamically stable. The separation of these emulsions is typically done using surfactants or an electric field gradient. Between distinct slugs in slug flow, there are microdroplets dispersed in the continuous fluid. All in all, to effectively separate these emulsions, modifications to the liquid interface, polarity, or charge are needed to effectively separate immiscible components in a microfluidic environment.
Slug Flow
This flow is highly dependent on the wettability of the constituents. The nonwetting phase will not have favorable interactions with the walls of the channel and if flow and geometrical parameters are optimized, then the nonwetting phase will form a phase separated droplet. This droplet is referred to as a “slug” and is more frequently used in microfluidics due to the low flowrate environment offering the interfacial stability necessary to maintain it. Streamlines are formed around the droplets creating vortices. This internal circulation allows the droplet to remain intact and the bulk fluid to stay separated from the nonwetting phase. Not only that, the internal circulation increases mixing which allows for additional separation. To separate the droplet from the bulk fluid a change in material is usually necessary. For example, the separation of acetic acid from benzene might require the channel to have one outlet made of steel and the other made of teflon as illustrated by Kashid et al5. This provides a difference in polarity to separate the immiscible constituents. Slug flow has recently been used in the separation of ethyl esters from N-3 polyunsaturated fatty acids (PUFA-Ets). Certain ethyl esters are used in maintaining the integrity of the nervous system (for example: docosahexaenoic acid and eicosapentaenoic acid) and thus are very important for use in the pharmaceutical industry. The separation of these ethyl esters from PUFAs is often very time intensive and exhaustive. To overcome this challenge, scaling out microreactors could be a promising solution. Kamio et al. has been able to show the separation of a bulk organic phase from the aqueous slug droplet phase using a union tee. This is done by using a Y-mixer to generate internal circulation and force the creation of a slug flow of the aqueous phase. Silver ions are used on the macro-scale to improve separation efficiency and thus were used to help create the aqueous slugs. After slug formation, the two phases are separated based on their residence time within the union tee. As the union tee has a small diameter, it allows for the organic phase (between slugs) to pass into the union tee and allow for partial phase separation. Although, not all of the organic phase is separated, it has been proved (using gas chromatography) that the organic phase is clean4.
Electric Field Separation
Using an electric field to actively coalesce the fluids causes them to agglomerate and then split respectively allow for simple separation. The electric field strength is therefore an important parameter to consider to allow for complete separation without excessive heating causing a change in phase. If this is being done in an aqueous environment, it is very important to use an alternating current to avoid the electrolysis of water into hydrogen and oxygen gas. Using an alternating current does not allow for efficient electrolysis as the anode would continually switch places with the cathode (hence alternating) and not allow for gas evolution.
Reverse Micelle Extraction
The second means of separation involves increasing the concentration of surfactant above the critical micelle concentration. It is already a common procedure to decrease the energy required to form an emulsion by use of a surfactant. This stabilization of the interface between immiscible compounds allows for emulsions to form easier, however it is also possible to use surfactants as a separations technique. This is done through a technique called reverse micelle extraction which as the name suggests, relies on a dilute polar medium dominated by a organic/nonpolar liquid phase. Once critical micelle concentration is reached, it is more stable to form a reverse micelle as there are more favorable interactions on the large surface area of the outer shell2. This allows for the encapsulation of polar compounds for separation from the emulsion. It is then possible to use electric field separation or a centrifugal microfluidic device for separation. In most contexts, reverse micelle separation is used to aid the separation efficiency of difficult to extract biological compounds.
Cocurrent Flow
Pressure-driven fluid flow is one of the more common means of microfluidic separation. Short diffusion distances and a large surface area contact between component phases allows for high degrees of separation in an otherwise inefficient process. As suggested, due to laminar flow conditions, multiple fluid phases are distinct and will remain separated. A stable interface forms between the phases and allows for active diffusion. This allows for distinct continuous phase separation of multi-components7. One example of this is extracting radionuclides1. A radionuclide is an atom that has been shown to have a tremendous amount of nuclear energy. Radionuclides that are analyzed are often in small amounts and are highly expensive materials. These radioactive materials have the potential to be analyzed using microfluidics or to be extracted in small amounts. The purpose of the study by Hellé et al, was to both extract and analyze the kinetic and thermodynamic of the extraction in order to further optimize the system. The device was made out of glass as it is resistant to organic solvents and acids for various extractions. The choice extractants were the neutral dimethyl-N1,N3-dibutyl-2-tetradecyl malonamide (DMDBTDMA), the acidic di-2-ethylhexylphosphoric acid (HDEHP), and cupferron as it can be used for ion pair extraction1. With a cocurrent flow design, the phases will remain separated and allow for an easy extraction.
Aqueous Two-Phase Systems (ATPs)
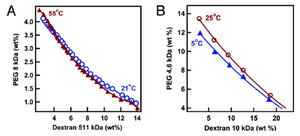
Polymeric systems (especially protein systems) can be more difficult to separate, especially when there are multiple polymer-rich phases. Miscible polymer systems are used to aid in the separation of macromolecules as well. Other components such as alcohol concentration and salt concentration can “salt out” a protein into a different phase. This effect, while not completely understood, minimizes the interactions of the protein with the aqueous phase and allows it to congregate into the polymer-rich phase. As one might imagine, this type of phase separation is very delicate and requires a lot of optimization to use well, but becomes easier in a microfluidic device6. With laminar flow and a stable interface, it is possible especially when restricting the aqueous phase to allow for the shortest diffusion distances. The most cited example of the aqueous two-phase system is the PEG-dextran system. As with all aqueous two-phase systems the miscibility of the two components in each other dependents on the relative concentrations of both7. In one study composed by Long et al., a PEG-dextran ATPs was used for the creation of a synthetic cell/vesicle. This system was used to help mimic compartmentalization within cells8. This approach was done by encapsulating soluble PEG and dextran within a lipid bilayer. By varying concentrations of the ATPs allowed for the two solution to become immiscible in one another. Proteins that favor a more hydrophilic environment would migrate to the aqueous dextran portion of the artificial cell. This study also shows how membraneless organelles can partition themselves within a cell. Refer to Figure 3 for the phase diagram associated with this system8.
References
1. Hellé, Gwendolyne, et al. “Microfluidic Tools for the Liquid-Liquid Extraction of Radionuclides in Analytical Procedures.” Procedia Chemistry, vol. 7, 2012, pp. 679–684., doi:10.1016/j.proche.2012.10.103.
2. Kralj, Jason G., et al. “Surfactant-Enhanced Liquid–Liquid Extraction in Microfluidic Channels with Inline Electric-Field Enhanced Coalescence.” Lab on a Chip, vol. 5, no. 5, 2005, p. 531., doi:10.1039/b418815b.
3. Datta, Subhra, and Sandip Ghosal. “Characterizing Dispersion in Microfluidic Channels.” Lab on a Chip, vol. 9, no. 17, 2009, p. 2537., doi:10.1039/b822948c.
4. Kamio, Eiji, et al. “Microfluidic Extraction of Docosahexaenoic Acid Ethyl Ester: Comparison between Slug Flow and Emulsion.” Industrial & Engineering Chemistry Research, vol. 50, no. 11, 2011, pp. 6915–6924., doi:10.1021/ie102207c.
5. Gu, Hao, et al. “Droplets Formation and Merging in Two-Phase Flow Microfluidics.” International Journal of Molecular Sciences, vol. 12, no. 4, 2011, pp. 2572–2597., doi:10.3390/ijms12042572.
6. Iqbal, Mujahid et al. “Aqueous two-phase system (ATPS): an overview and advances in its applications” Biological procedures online vol. 18 18. 28 Oct. 2016, doi:10.1186/s12575-016-0048-8
7. Huang, Yushi, et al. “Aqueous Two-Phase Extraction for Bovine Serum Albumin (BSA) with Co-Laminar Flow in a Simple Coaxial Capillary Microfluidic Device.” Microfluidics and Nanofluidics, vol. 16, no. 3, 2013, pp. 483–491., doi:10.1007/s10404-013-1245-2.
8. Long, M. S., Jones, C. D., Helfrich, M. R., Mangeney-Slavin, L. K., & Keating, C. D. (2005). Dynamic microcompartmentation in synthetic cells. Proceedings of the National Academy of Sciences, 102(17), 5920. https://doi.org/10.1073/pnas.0409333102
9. Gu, H., Duits, M. H., & Mugele, F. (2011). Droplets formation and merging in two-phase flow microfluidics. International journal of molecular sciences, 12(4), 2572-2597.
