Linda Griffith by Adam Murphy
Early Days in Southern Georgia
Born on August 30, 1960, Linda G. Griffith grew up in Valdosta, Georgia. Linda has two brothers, one older and one younger, as well as a sister. While her family was not well off, her mother had a master’s degree in sociology and her father was an engineer, who worked for a local power company. In high school, she received a National Merit Scholarship in writing. Because of her family’s economic situation, she had to pay her way through college. In light of this, she decided to go to the Georgia Institute of Technology to major in chemical engineering, like her older brother before her. She learned to invest her money in the stock market to afford paying for college. Linda was active in college and wrote for the undergraduate newspaper. She eventually became the editor of the Georgia Tech collegiate newspapers, the Technique.[1]

Graduate School and Beyond
Linda Griffith chose to go to the University of California, Berkeley in part because she wanted to a campus social environment where there was as many women as men[1]. More importantly, UC Berkeley was one of the top chemical engineering graduate programs alongside MIT and Minnesota; and according to an interview, she had no desire to move to someplace where there was snow. She received a PhD at Berkeley in 1988. Because of her strong academic background, she quickly received faculty interviews in her third year of graduate school. Due to agreements with her now ex-husband, she chose industry and accepted a position at Dow Chemical two months before completing her graduate thesis[1]. The lab, however, closed down and relocated, leaving her with no job. In the back of a Science magazine, she found a postdoc ad with Robert Langer and Jay Vacanti at Harvard Medical School and the Massachusetts Institute of Technology. During this time, she was an author alongside Langer and Vacanti in over a dozen publications detailing their work in the design and fabrication of degradable polymeric substrates for tissue engineering[2]. After a successful three year period and with the encouragement of her mentor Robert Langer, Griffith was accepted for an open faculty position in the department. In 1996, she become an Associate Professor of the Chemical Engineering department at MIT, eventually reaching tenure in 1998[3]. Griffith led the movement to establish a minor degree in biomedical engineering as part of the chemical engineering curriculum. Launched in 1995, the minor degree was MIT’s first interdepartmental minor degree. Beyond MIT, Linda Griffith is a member of numerous research focus groups and chairs committees relating to biomedical engineering. Dr. Griffith is the scientific director for the Center for Gynepathology Research (CGR) at MIT. Founded in 2009, the CGR takes an engineering approach to determining key biological mechanisms and physiology of the female reproductive system to find novel ways to treat diseases specific to the female body[4]. Dr. Griffith is also a member of the MIT Center for Environmental Health Sciences (CEHS), the NSF Science and Technology Center for Emergent Behaviors of Integrated Cellular Systems (EBICS), and the Koch Institute for Integrative Cancer Research among others. Notably, she chaired the Undergraduate Curriculum Committee for Biological Engineering until 2009, a committee developed out of need to facilitate the creation biological engineering as a new undergraduate major at the Institute.[5]
Honors and Awards
Member of the National Academy of Engineering
Recipient of the MacArthur Foundation Fellowship, 2005
Recipient of the Popular Science Brilliant 10 Award, 2002
Recipient of the National Science Foundation Presidential Young Investigator Award 1991 - 1998
The MIT Class of 1960 Teaching Innovation Award
Radcliffe Fellow at Harvard University, 2009-2010, 2010-2011
Current Research
Dr. Linda Griffith is the primary investigator of a graduate research lab at the Massachusetts Institute of Technology and is also the lead scientific director of the MIT Center for Gynepathaology. Dr. Griffith’s lab focuses on the design and fabrication of functionalized biomaterial to target and control receptor-mediated cell processes. In addition, the lab seeks to improve drug development using powerful in vitro tissue models through the lenses of systems biology. Moreover, three dimensional in vitro tissue engineered models are being used to overcome obstacles in drug development and to observe physiological properties of cancerous tissue. An array of 3D tissue models are being developed to mimic various microsystems of the body such as the liver, lung and reproductive systems. In relationship to reproductive systems, the CRG focuses on female reproductive systems diseases – endometriosis, adenomyosis, and infertility – which previously haven’t received enough research attention or funding. The CRG lab applies the previously mentioned biotechnologies and engineering principles to determine the disconnect that occurs in diseased cell communication networks.[6]
Significant Publications
3D Liver Tissue Model-Based Research
Capturing Complex 3D Tissue Physiology in vitro[7]
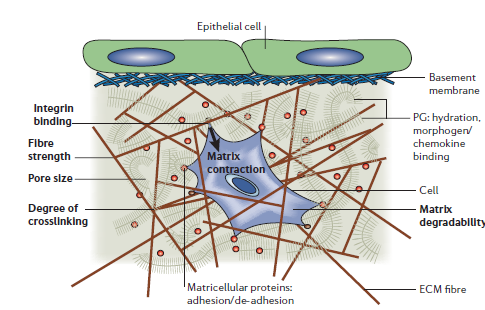
One of Dr. Griffith’s most actively cited publication, “Capturing Complex 3D Tissue Physiology in vitro,” provides guidelines and design principles when determining how to recreate tissue microenvironments using 3D tissue models. 3D tissue models allow researchers to study tissue physiology and pathology in vitro. Some of the salient factors for the adoption of 3D models include the use of human cells over animal cells, pre-clinical drug screening, and superior complexity compared to 2D culture models. Dr. Griffith the third dimension may prove critical in the study of tissue models because of the changed effects of mechanical input and cell adhesion, thereby impacting cell to cell signaling and cell contraction. Three dimensional tissue also creates a potential to observe cell morphology events only possible over large distance scales. Dr. Griffin also cites the importance of matrix stiffness in impacting cell differentiation. One experiment on breast-epithelial-cell culture showed that cells cultured on flat plastic had little cell differentiation whereas cells cultured on ECM showed statistically significant expression of mammary-specific genes. Importantly, the greatest differentiation of the cells occurred when cultured within an ECM gel matrix. When the ratio of collagen and matrigel in the matrix was altered over a range of concentrations, the gel matrix stiffness was proportionally changed and cell differentiation was disrupted. Synthetic ECM is fundamental to the success of 3D tissue models. While natural ECM provides the necessary physiological and chemical properties to facilitate cellular response and activity, it is often impossible to determine the root cause of cell responses given that many of the factors present in the natural ECM are largely unknown. Polyethylene oxide (PEO) peptide-rich gels have been shown to have low tolerance for nonspecific binding of biomolecules, making the material an ideal prospect for creating controlled microenvironments with known growth-factor binding sites and adhesion molecules. To display the power of synthetic ECM in 3D tissue models, it was shown that cell response may be affected by local ligand clustering in addition to ligand concentration. A 3D model system used RGD-containing peptides critical in integrin-driven cell adhesion to observe local ligand clustering and the causation of integrin clustering commonly seen in mammalian cancer tissues. Several studies, highlighted by Dr. Griffith, also stress the important of mechanic-chemical signaling in cell behavior. One study, by Grodzinsky, showed that dynamic compression of cartilage affected how proteoglycans and protease inhibitors were distributed amongst a 3D tissue model. The importance of interstitial fluid flow in the metastasis of tumor cells through the lymphatic system can be quantified through tissue modeling. Through review of several dozens of studies, Dr. Griffith supported the idea that knowledge of cell-receptors and corresponding cell cues isn’t enough to isolate certain cell behaviours. It is necessary to determine how spatial and temporal properties of these cues in the cellular environment impact cell behavior, and this can only be done using three dimensional models with controllable environmental properties.
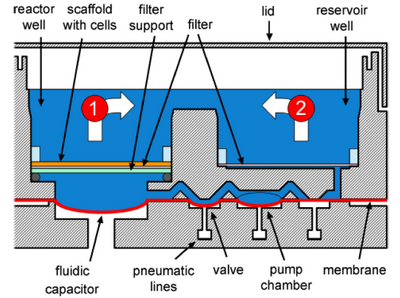
Perfused Wellplate for Liver Tissue Engineering[8]
To accurately model the complexity of in vivo tissue using in vitro tissue models, devices and designs that have large scalability or high throughput are desirable. Dr. Griffith and colleagues developed an array of open well bioreactors with perfusion flow capabilities. The multiwell plate contains 12 individually isolated bioreactors that can grow between 400,00 and 600,000 cells. 3D tissue is cultured within the multiwell system by pipetting cell suspension directly onto ECM scaffolds present within each well. To ensure successful maintenance of the tissue, a transport model for oxygen was established to determine the effect of system parameters on oxygen concentrations gradients across each well. Conceptually, each bioreactor contains a reactor well, where the ECM scaffold and tissue is, a reservoir well to hold perfusion liquid, and a micropump to facilitate perfusion. The bioreactors of the multiwell array can be manually or automatically loaded with cell cultured and then individually sealed to reduce airborne contamination risks. The ECM scaffold itself contains 769 microchannels with a depth of 0.24 mm and 0.34 mm in diameter. A 5-micron pore filter under the scaffold captures cells within the channels. The inclusion of a filter in the reservoir prevents cells from clogging the perfusion micropumps. The tissue is perfused through the use of pneumatic micropump actuation, illustrated in Figure 4. This design for an array of microwell bioreactors successfully maintain 3D liver tissue with constant microperfusion. Predictive oxygen transport models for gradients through the bioreactor corresponded to quantitative data taken from experiments that measured the oxygen concentrations in rat hepatocytes cultures as a function of time and flow. LSECs, which struggle to maintain cell phenotype differentiation in static conditions, were proven maintain cell differentiation over two weeks due to the constant microperfusion. The benefits of this system include the scalability of the system, the controlled and stable oxygen gradients across the bioreactor, and the pneumatic pump system for vacuum-driven fluid flow.
Spontaneous Dormancy of Metastatic Breast Cancer Cells in an All Human Liver Microphysiologic System[9]
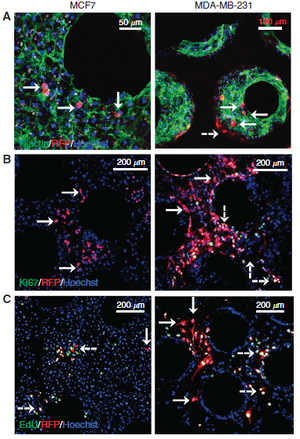
Breast cancer has the ability to enter dormancy behavior that makes future reoccurrences of metastatic growth possible. To address the shortcomings of preexisting cancer dormancy models, Griffith et al propose a human ex vivo hepatic microphysiologic system, where human hepatocytes and non-parenchymals cells are used in combination with breast cancer cells to form the tumor microenvironment. The created hepatic tissue is functional for 15 days, as confirmed by performing liver-specific protein production and drug metabolism assays. Specifically, ASR and ALT markers were monitored to determine the amount of hepatocyte injury; a steady decrease in these markers was observed as tissue formed within the reactor. Glucose consumption levels and urea levels were observed to be mostly constant, with urea decreasing by the end of the 15-day trial. Upon cell seeding of breast cancer cells into the liver tissue, all the same protein and metabolite assays showed no difference between control and tissue with cancer cell seeding, indicating that the breast cancer cells did not impact liver cell functionality. NPC’s include endothelial, stromal, and immune cells. Furthermore, the NPCs are proven to be key for the successful tissue model due to their role in cell signaling. The breast cells were able to penetrate the liver tissue without impacting liver function. Furthermore, the cancer cells were shown to enter quiescent dormancy, proven by appreciably decrease in cell cycling. The results of this experiment support the application of this ex vivo liver tissue for research relating to breast cancer metastases.
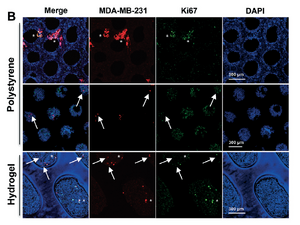
A Liver Microphysiological System of Tumor Cell Dormancy and Inflammatory Responsiveness is Affected by Scaffold Properties[10]

Griffith and collaborators revisited this ex vivo liver tissue model to address a phenomena that was observed during previous work. Dormant breast cancer cells were mainly located within the 3D tissue itself, while there existed a population of proliferating breast cancer cells that were in contact with the polystyrene scaffold. To address the stiffness and mechanical properties of the scaffold, a polyethyleneglycol- based hydrogel modified with a fibronection-dervied adhesion peptide mimic to promote cell-scaffold binding(PEGDa-SynKRGD) was proposed. Through procedures similarly reported in the previous publication, ATL, AST, Urea, and Glucose were all measured during 15 day culture process. No differences were found between polystyrene and the hydrogel for the four assays. A large analyte assay targeting pro-inflammation showed that newly cultured hepatic tissue secreted lower levels of inflammatory analytes and was generally more sensitive to inflammatory stimuli. This is hypothesized to be attributed to the permeability and flexibility of the hydrogel. Importantly a significant increase in dormant cells was found in the interface tissue proximal to the new hydrogel scaffold material. A lack of Ki67 staining (indicating cell proliferation) implies that the cancer breast cells of the hydrogel scaffold are dormant. To generalize, Griffith also analyzed 35 cancer markers to monitor cell activity of sub-populations in the hydrogel and polystyrene respectively. A high baseline of cancer markers in the polystyrene indicated that these cells were metastatic and proliferative rather than the ideal dormant state.
Other Famous Collaborations
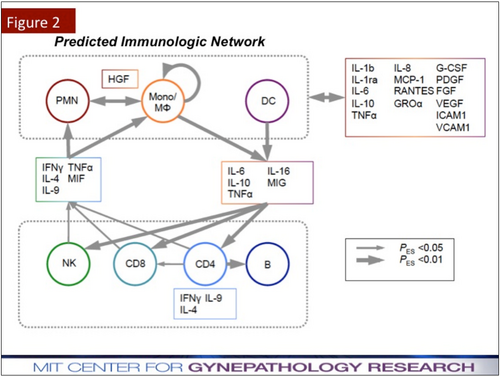
In addition to Dr. Griffith's groundbreaking lab research at MIT and several research centers, she also received significant fame from non-scientists for her collaboration with Dr. Vacanti in the assistance of growing human ear tissue on the back of mice[1]. Also, Dr. Griffith collaborated with Emanuel Sachs, Michael Cima, and others from MIT in the first patent for three dimensional printing. This patent has been commercialized and is currently used in the production of FDA-approved scaffolds for bone regeneration[5]. She has also done extensive research on female-specific reproductive system diseases, such as endometriosis and adenomyosis. Griffith and her research team implement systems engineering and multiplex computational modeling to tackle complex cell communication networks and monitor protein activity, uncovering key genes for therapeutics.
References
[1] Linda G. Griffith, an interview conducted by Infinite History at the Massachusetts Institute of Technology. [1]
[2] The Practical Activist, an MIT Technology Review on Linda Griffith [2]
[3] MacArthur Fellows Program, Linda Griffith Bioengineer [3]
[4] The MIT Center for Gynepathology, Linda G. Griffith[4]
[5]MIT, Griffith Lab Home Page[5]
[6] MIT Biological Engineering, Linda G. Griffith:Research-at-a-glance[6]
[7] Capturing complex 3D tissue physiology in vitro. PUBMED[7]
[8]Perfused Multiwell Plate for 3D Liver Tissue Engineering[8]
[9]Spontaneous Dormancy of Metastatic Breast Cancer Cells in an All Human Liver Microphysiologic System, British Journal of Cancer. [9]
[10]A Liver Microphysiological System of Tumor Cell Dormancy and Inflammatory Responsiveness is Affected by Scaffold Properties, Lab on a Chip 2017.[10]
[11]Multiplexed Protease Activity Assay for Low-Volume Clinical Samples Using Droplet-Based Microfluidics and Its Application to Endometriosis, Journal of the American Chemical Society. [11]
