Lidstrom: Agarose Gel Electrophoresis
From OpenWetWare
Jump to navigationJump to search
Return to Protocols
- handy link: gel electrophoresis
- click the gel electrophoresis button on the left
Imaging
Imaging systems
We have two imaging systems:
(1) ______ on the island
(2) Across from the SDS-PAGE gel pouring room
- Login info is taped to the monitor
- Software = GeneSnap
Instructions:
- Click the green circle on the top left. This turns the camera on
- Set the filter button (button = red/blue/green circles within a circle) to EtBr/UV
- Turn on the light by selecting transillumination (not Et. Br) from the drop-down menu
- Select a time. 500 ms works.
- the buttons below the circle (now red) are:
- aperture (exposure time; donut-looking button)
- zoom (magnifying glass)
- focus (eyeball)
- Click the red circle to take the picture
- There are buttons to invert the colors (yin-yang) and sharpen (knife).
Imaging tips
- Exposures should be < 0.5 seconds to minimize background emissions. If you need longer, you probably should have had more ethidium bromide or other DNA dye.
Tips
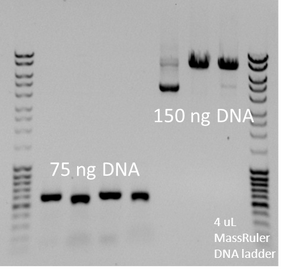
- MassRuler is a great ladder. Use ~4uL.
- Item # SM0403 from Thermo Scientific. Stable at RT for many months.
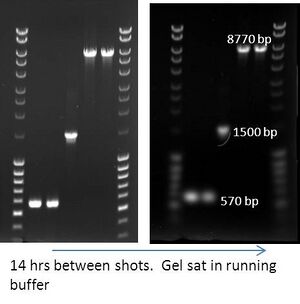
- If you have a voltage source that will stop after a set amount of time, feel free to run your gel as you leave and image it in the morning. Diffusion of bands shouldn't be a big problem unless they are << 500 bp.
- Don't do this if you are going to cut & gel purify the band.
- You don't need much DNA. 50 ng should be plenty.
- TBE buffer can be re-used for months.
- This is nice, because you shouldn't dump ethidium bromide containing solution down the drain.
- Janet usually uses it heavily for 4+ months before replacing it.
- Always add DI water to replace evaporated water, not TBE buffer
- Agarose gels can be "drained" and re-used several times.
- Don't use a previously used gel for DNA you will cut and purify for cloning/sequencing.
- Drain with 100V for ~90 min.
OLD: Ethidium bromide instructions
If starting with an empty box:
- Fill with TBE until it covers the gel holding tray.
- Add 40 uL of ethidium bromide if your gels don't already have it mixed into the liquefied agarose.
- If your buffer already has some ethidium bromide but it needs more, 20 uL is probably good.
- Load sample
- 1.7 uL of a PCR product is usually plenty
- Load ladder
- If using MassRuler, 4 uL is good for skinny lanes.
- Run @ 110 V for 20 min and check the gel.
- 40 min is pretty safe for 0.7 - 1% agarose gels