Lidstrom:Chemical Transformation
From OpenWetWare
Jump to navigationJump to search
Back to Protocols
You can chose between chemically competent cells and electrocompetent cells. Andrew & Nicole make chemically competent cells for the lab to use. Several strains are kept in stock in the -80oC freezer.
Using Chemically Competent Cells
- Thaw frozen (-80oC) competent cells on ice.
- You REALLY dont want it to get too warm; add plasmids while it is still slushy.
- Add 1-10 uL DNA
- 10 uL if ligated plasmid. Use only 1 uL if regular plasmid from miniprep
- Incubate @ 42oC for 45 sec - 1 min
- Incubate on ice for 2 min
- Add 1 mL LB
- Sandy, Ceci, & Janet use 500 uL
- Incubate at 37oC for 45 min - 1 hr in eppendorf tubes
- Ideally shaking though it may not matter. You can tape your tubes to a rack in the shaker. Tape them well if you do -- they fly off!
- Pellet cells by centrifugation
- keep the ~100 uL droplet after you pour it off (Andrew)
- Plate 50-100 uL cells on LB (+antibiotic(s)) agar plate
- If you are worried about having a lawn, do one plate with more cells and dilute a fraction of the cells and plate a diluted aliquot.
Note: electroporation is much more efficient than chemical transformation
- Transformations done with 1uL of 0.1 ng/uL DNA (pGA 3K3 RFP from Rob Egbert). 0.1 ng is ~10^8 plasmid copies. Tami's electrocomp cells yielded several hundred colonies, which suggests the transformation efficiency is on the order of 100/10^8 = 0.000001 = 0.0001% efficiency. The chemically competent cells yielded < 10 colonies, which is much lower efficiency.
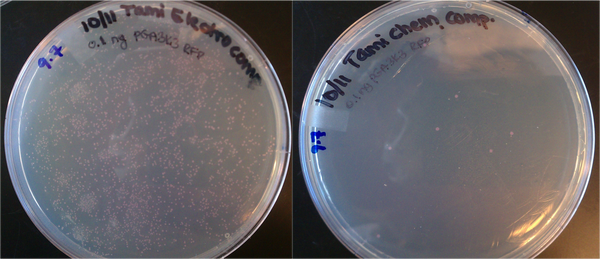
Top10 cells prepared for electroporation and chemical transformation. Both were transformed with ~10^8 copies of DNA (0.1 ng of plasmid.)