Lateral Flow Assays - Alex Debreceni
Introduction
Lateral flow assay is a paper based microfluidic technique used to detect a specific chemical component in a complex mixture. Lateral Flow Assays are cheap, easy to use, and rapid portable detection devices, designed for use for non-laboratory settings. This technology is widely used in medical fields and law enforcement to test bodily fluids such as urine, blood, sweat, and saliva.[1] Biomarkers for organ failure, infection, toxic pathogens, or illicit drugs can be detected with high sensitivity and selectivity due to the fact that each device is specifically designed to interact with the molecule of question.[2] Essentially these devices operate through the flow process called capillary action, which pushes the fluid containing the analyte through various zones, where molecules are bound to interact with and detect only that analyte.[2]
Components

The membrane material of most of these devices is nitrocellulose with various evenly distributed pore sizes.[2] Changing the material and pore size influences the capillary flow time, which influences the reaction time of the analyte in binding area. The first component of most lateral flow assay devices is the sample pad. The sample pad is the spot where the fluid sample is placed. The sample pad treats the sample and modulates any chemical variability. The pad is made of cotton linter, glass fibre, rayon, and filtration materials. Sample pads are pretreated with a pH buffer, surfactants, and blocking reagents to influence the flow rate and viscosity of the sample.[2] The next part of a general lateral flow assay is the conjugate pad. The conjugate pad couples the analyte in the sample with the conjugate and releases the pairing. These conjugates are generally antibodies that specifically bind to the target molecule. These antibodies are bound to the pad using colloidal nanoparticles, such as gold.[2][3] The next part of lateral flow assays devices is the reaction membrane. This membrane works as a capturing mechanism for the conjugate-analyte pair. This part also forms visible bands when the analyte is present. These bands are placed further down the device to improve interaction time. As the fluid flow rate decreases the The final part of the system is the waste reservoir. This component serves as a sink for excess sample and prevents wicking towards the reaction membrane.[2]
Mechanism

A liquid sample, which contains the analyte of interest is placed on the sample pad. Through capillary action, the fluid travels through the various zones of the device. When the fluid reaches the conjugate pad, the analyte will bind to the antibodies specific to the molecule. these antibodies are conjugated to colored or fluorescent particles. The most common conjugate particles are colloidal gold and latex microspheres. The conjugate-analyte pairing travels with the fluid to the reaction membrane This membrane contains antibodies or antigens, immobilized in a nitrocellulose These particles react with the analyte and activates the test line. The control line in the system activates when there is flow through the system. When both lines are present, the analyte is present in the sample. If only the control line is present, there is no analyte in the sample. Once the fluid passes these sections, and absorbing pad at the end of the system collects the excess. This pad also maintains the capillary action throughout the full device.[1]
Pregnancy Tests
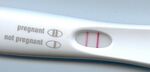
Pregnancy tests are the most common commercial use for lateral flow assay devices. The test fluid for these devices is urine. If a women is pregnant, the hormone, human chronic gonadotropin is present in the women's urine. The antibodies used for pregnancy tests are different for each manufacture. Some use antibodies to detect nonnicked hCG and free-beta hCG, while others detect nicked and nonnicked hCG.[6]
References
[1]Koczula, K. M., & Gallotta, A. (2016). Lateral flow assays. Essays In Biochemistry, 60(1), 111-120. doi:10.1042/ebc20150012
[2]Posthuma-Trumpie, G. A., Korf, J., & Amerongen, A. V. (2008). Lateral flow (immuno)assay: its strengths, weaknesses, opportunities and threats. A literature survey. Analytical and Bioanalytical Chemistry, 393(2), 569-582. doi:10.1007/s00216-008-2287-2
[3]Martin, J. M., P[Acaron]Ques, M., Tiny A. M. Van Der Velden-De Groot, & Beuvery, E. C. (1990). Characterization of Antibody Labelled Colloidal Gold Particles and Their Applicability in a Sol Particle Immuno Assay (SPIA). Journal of Immunoassay, 11(1), 31-47. doi:10.1080/01971529008053256
[4]Yetisen, A. K., Akram, M. S., & Lowe, C. R. (2013). Paper-based microfluidic point-of-care diagnostic devices. Lab on a Chip, 13(12), 2210. doi:10.1039/c3lc50169h
[5]Lateral Flow Assay. (2014, March 29). Retrieved February 17, 2017, from http://www.dxdiscovery.com/lateral-flow-assay.html
[6]Cole, L. A. (1997). Immunoassay of human chorionic gonadotropin, its free subunits, and metabolites. Clinical Chemistry, 43(12), 453. Retrieved February 17, 2017, from https://www.ncbi.nlm.nih.gov/pubmed/9439438.
[7]HCG pregnancy strip test. (2016, November 19). Retrieved February 17, 2017, from https://en.wikipedia.org/wiki/HCG_pregnancy_strip_test
